Daily Procalcitonin-Guided Protocol Safely Reduces Antibiotic Use in Critically Ill Adults With Sepsis
Key Highlights
- Daily procalcitonin (PCT)-guided protocol safely reduced total antibiotic duration in critically ill adults with sepsis.
- The PCT-guided group demonstrated noninferior 28-day all-cause mortality compared with standard care.
- No significant reduction in antibiotic duration or conclusive safety outcome was found with daily C-reactive protein-guided protocols.
- These results support the use of PCT-guided discontinuation protocols in routine sepsis care.
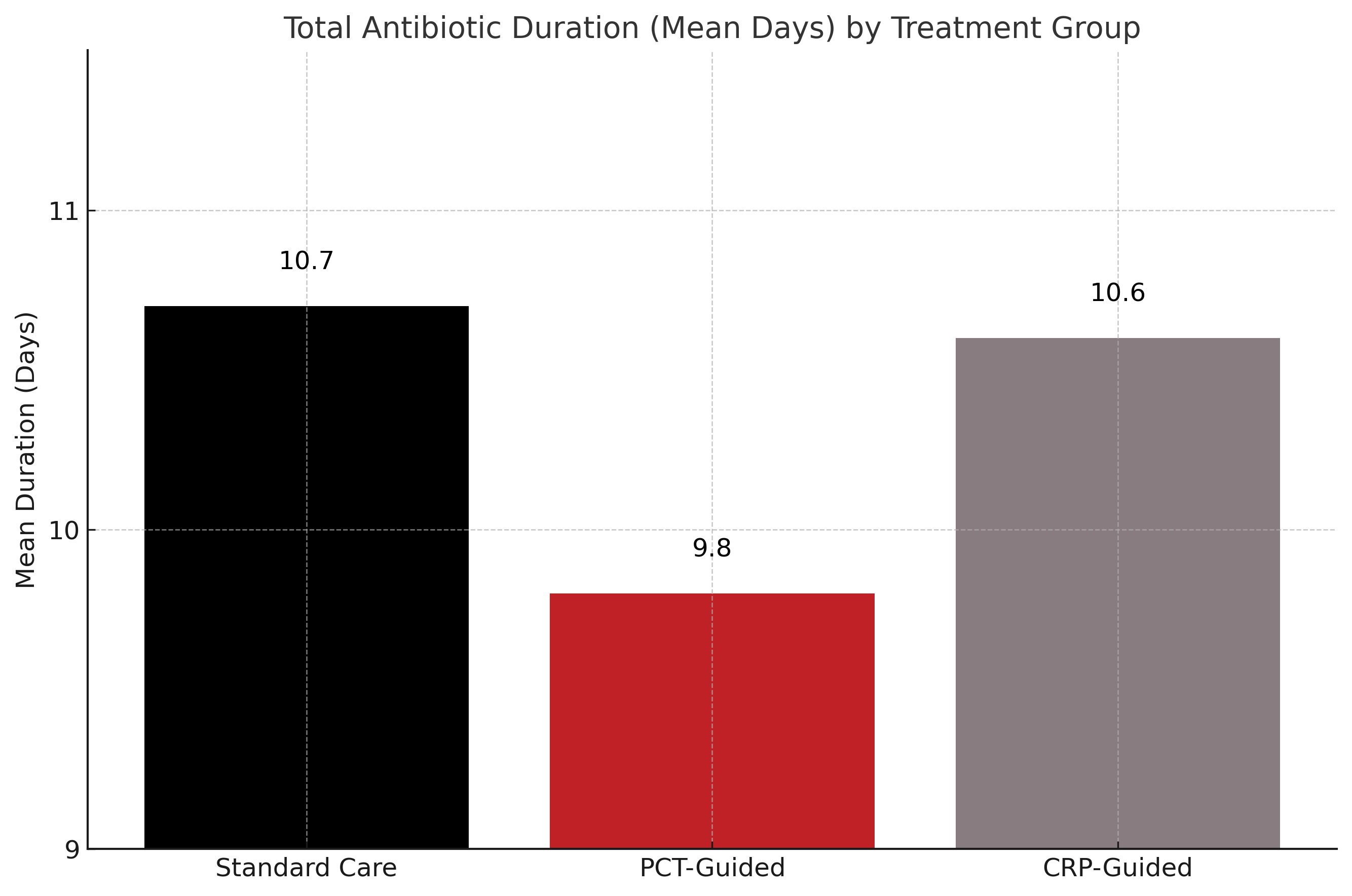
In this multicenter randomized clinical trial of 2760 critically ill adult patients with suspected sepsis, a daily procalcitonin (PCT)-guided antibiotic protocol led to a statistically significant reduction in total antibiotic duration compared with standard care, with a mean reduction of 0.88 days. This reduction was achieved without compromising patient safety, as 28-day all-cause mortality in the PCT group was noninferior to standard care. In contrast, a daily C-reactive protein (CRP)-guided protocol did not reduce total antibiotic duration and failed to demonstrate conclusive noninferiority in mortality outcomes.
The study was prompted by a need to optimize the duration of antibiotic therapy in sepsis management. While early antibiotic initiation is a cornerstone of sepsis care, prolonged antibiotic exposure can lead to adverse events, antimicrobial resistance, and unnecessary resource use. According to the authors, biomarkers such as PCT and CRP have been proposed as tools to guide antibiotic discontinuation, but prior evidence has been limited and of low quality, resulting in only weak clinical guideline recommendations, particularly for PCT and none for CRP.
The ADAPT-Sepsis trial was an intervention-concealed, multicenter randomized clinical trial conducted in 41 intensive care units across the United Kingdom. Eligible adults were enrolled within 24 hours of intravenous antibiotic initiation for suspected sepsis. Participants were randomized 1:1:1 to receive either standard care, daily PCT-guided antibiotic discontinuation advice, or daily CRP-guided advice. The intervention involved daily biomarker testing and centralized computer-generated guidance to local clinical teams, while maintaining allocation concealment.
Among the 2760 enrolled patients, the mean age was 60.2 years and 60.3% were men. PCT-guided therapy was associated with a reduction in total antibiotic duration over 28 days (mean 9.8 vs 10.7 days in standard care; mean difference, 0.88 days; 95% CI, 0.19–1.58; P = .01). The CRP-guided group had no significant difference in duration compared with standard care (mean duration, 10.6 vs 10,7 days in standard care; mean difference, 0.09 days; 95% CI, −0.60 to 0.79; P = .79). For safety, 28-day mortality was 20.9% in the PCT group versus 19.4% in standard care (absolute difference, 1.57%; 95% CI, −2.18 to 5.32; P = .02), meeting the predefined noninferiority margin. Mortality in the CRP group was 21.1%, yielding inconclusive noninferiority (absolute difference, 1.69%; 95% CI, −2.07 to 5.45; P = .03). Secondary outcomes—including hospital length of stay, readmissions, and adverse events—were not significantly different across groups.
Study limitations include potential bias due to the concealment strategy possibly delaying antibiotic discontinuation in the standard care arm, and the possibility of protocol contamination due to shared environments in ICU settings. Furthermore, the study did not assess the role of these biomarkers in initiating antibiotic therapy and may have limited generalizability to low-resource settings.
“The ADAPT-Sepsis trial strengthens substantially international recommendations for the routine use of protocolized daily PCT-guided antibiotic discontinuation in critically ill adults with sepsis,” the study authors concluded.
Reference:
Dark P, Hossain A, McAuley DF, et al. Biomarker-guided antibiotic duration for hospitalized patients with suspected sepsis: the ADAPT-sepsis randomized clinical trial. JAMA. 2025;333(8):682-693. doi:10.1001/jama.2024.26458


