Peer Reviewed
Erythema Nodosum in an Adolescent Following Acute Streptococcal Pharyngitis
Introduction. A 15-year-old adolescent girl with Fitzpatrick Skin Type III presented to dermatology with a 3-week history of tender, brownish-red, poorly demarcated, subcutaneous nodules on the bilateral anterior shins.
History. Three weeks prior, the patient had presented to an urgent care facility with subjective complaints of chills, rhinorrhea, and sore throat. She had a fever of 101.2°F with posterior oropharyngeal erythema and tonsillar exudate and was diagnosed with acute pharyngitis. A rapid streptococcal antigen detection test done at that time was negative, and it was supposed that her symptoms were likely secondary to a viral infection. She was sent home with supportive care, including recommendation to rest, stay hydrated, and use over-the-counter analgesics as needed for symptoms relief, and instructions to return if her symptoms persisted or worsened.
At her dermatology presentation, she reported that her throat was still slightly sore, but she denied any history of “sandpaper” rash, strawberry tongue, desquamation of the hands or groin areas, or dark-colored urine. On physical examination, she was afebrile with a normal respiration rate. Present on her bilateral anterior shins were scattered, exquisitely tender, brownish-red, poorly demarcated, subcutaneous nodules that were non-suppurative. She had no cervical, popliteal, or inguinal lymphadenopathy (Figure 1).
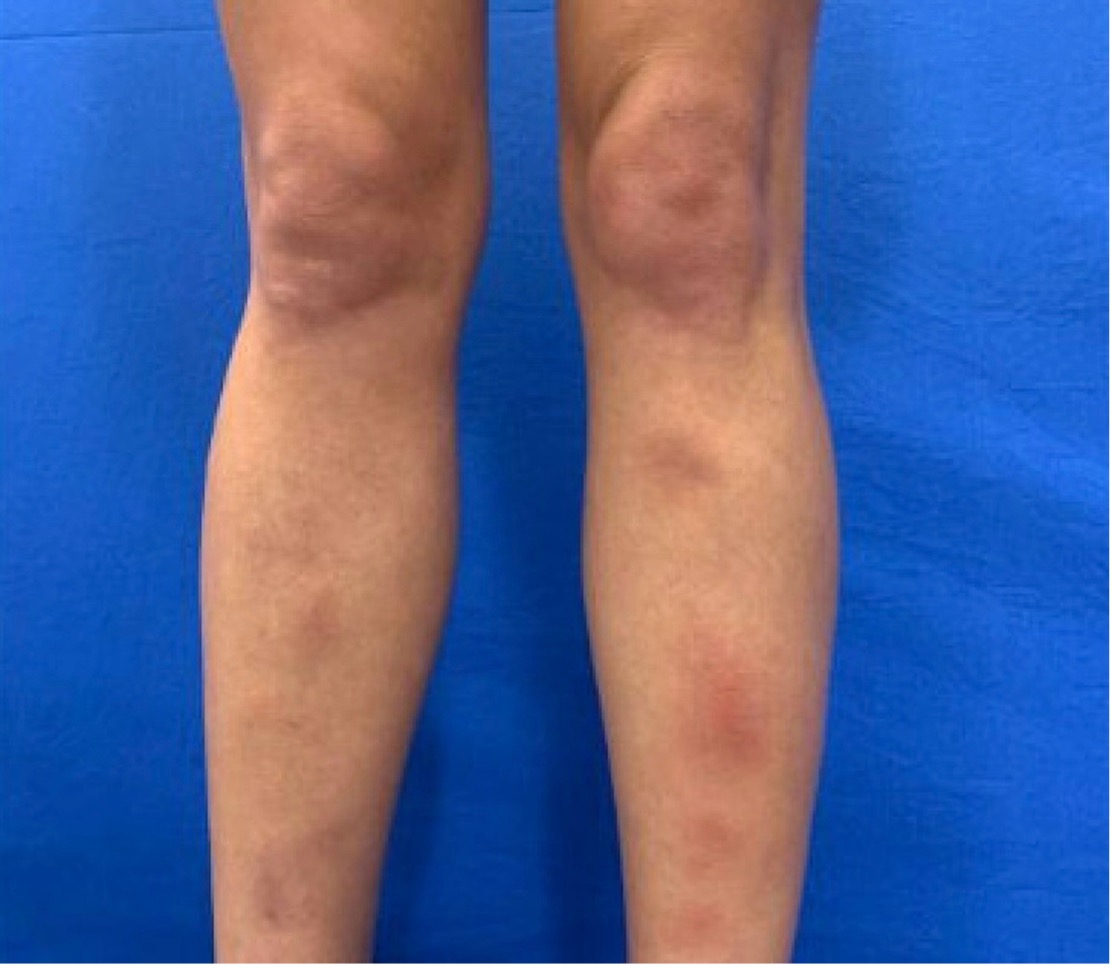
Figure 1. Fitzpatrick Skin Type III female with exquisitely tender, brownish-red, poorly demarcated, subcutaneous nodules on the bilateral anterior shins.
Diagnostic testing. The patient’s leg lesions were identified clinically by the pediatric dermatology team as erythema nodosum (EN). Recognizing that EN is not a specific diagnosis but, rather, a “reaction pattern” to a myriad of potential underlying disorders, the team sought to elucidate a more precise cause of her skin findings. Throat culture was negative for beta-hemolytic Streptococcus; however, the patient’s anti-streptolysin O (ASO) titer was elevated (> 200 IU/mL), suggesting a current or recent Streptococcal infection. The patient’s urine human chorionic gonadotropin was negative. QuantiFERON gold assay was negative for tuberculosis, and her chest X-ray was normal as well. Given her constellation of signs and symptoms and investigative workup, the patient was diagnosed with EN secondary to Streptococcal infection.
Differential diagnosis. Although Streptococcal infection is the most common identified cause of EN,1 we considered other common causes including infections (e.g., tuberculosis, COVID-19), pregnancy, or oral contraceptive pills. 1-3 The patient had no risk factors for tuberculosis, and her QuantiFERON gold test was negative. She was negative for COVID-19, and she denied any recent vaccinations. Asked with her parents out of the room, the patient denied having ever been sexually active. She was not taking any medications including oral contraceptive pills, vitamins, or herbal supplements. Ultimately, post-streptococcal EN was strongly supported by the patient’s constellation of signs and symptoms and her elevated ASO titer.
Less common causes of EN were also considered, including inflammatory bowel disease (IBD), sarcoidosis, autoimmune disease, rarer infections (including bacterial, fungal, viral, and parasitic), and underlying malignancy. The patient denied a known personal and family history of IBD. She also denied abdominal tenderness, recent diarrhea, or hematochezia. Her chest X-ray showed no signs of bilateral hilar lymphadenopathy, making active sarcoidosis unlikely. She denied a known personal or family history of autoimmune disease, and a screening antinuclear antibody (ANA) was negative.
Rarely, blastomycosis and histoplasmosis have been associated with EN.1 In this case, the patient specifically denied spelunking, international travel, and any known interaction with birds, bats, or other non-domesticated animals. Malignancy was also not likely in this otherwise healthy patient with no history of unintentional weight loss and otherwise normal bloodwork.
Treatment and management. To ensure definitive treatment of her suspected Streptococcal infection, the patient was prescribed doxycycline monohydrate 100 mg tablets twice a day for 14 days. She was counseled on good sun protection practices, and she was instructed to take her medication with a full glass of water and to avoid laying down for at least 1 hour after taking her medication to mitigate doxycycline-associated esophagitis. Due to the painful nature of her EN lesions, the patient was prescribed oral potassium iodide 300 mg three times a day for 28 days. She was also advised to manage her symptoms with rest, elevation, and non-steroidal anti-inflammatory medications. A 2-week follow-up call was made to the patient and her family to assess the patient’s progress.
Outcome and follow-up. The patient and her family were contacted again two weeks later and confirmed that the patient’s symptoms completely resolved within 4 weeks of treatment. Given the challenges of accessing the clinic, the family agreed that a follow-up appointment was not necessary.
Discussion. EN is a hypersensitivity reaction to an antigen resulting in inflammation of the subcutaneous fat. Clinically, it is characterized by the acute onset of painful, erythematous nodules.1,4 While these lesions classically present on the pretibial surfaces, it is common to see lesions develop on the arms, thighs, trunk, or even face in pediatric patients.5 Identification of the underlying etiology is key to definitive management. While approximately 30% to 50% of pediatric patients with EN are idiopathic, beta-hemolytic group B streptococcal infection remains the most common identifiable etiology.1,5
Physical manifestations of post-streptococcal EN usually occur two to three weeks after infection and may be diagnosed with streptococcal rapid antigen test or ASO titer, the latter of which begins to increase approximately one week after infection.4 Although rapid streptococcal antigen detection tests (RADTs) are frequently utilized to make the initial diagnosis of streptococcal infection, false negatives must be considered within the clinical context. Studies have demonstrated that RADTs are highly specific (around 95%); however, their sensitivity has been found to range from 70% to 99%.6 For this reason, several clinical practice guidelines recommend supplementing negative RADT results with a throat culture. While still considered the gold standard, throat cultures are not perfect either. Under ideal scenarios, throat cultures for group A beta-hemolytic Streptococci have been reported to have a sensitivity of 90% and specificity of 99%.7 However, in outpatient practice settings, these values are lower – with a sensitivity ranging from 29% to 90% and a specificity ranging from 76% to 99%.7 Such differences could be related to human error in procurement of the cultures themselves.
Conclusion. This patient’s case underscores the importance of maintaining a high index of suspicion and for working comprehensively through the differential diagnosis for causes of EN within the pediatric population. More specifically, it highlights the crucial role that ASO titers can play in helping to identify Streptococcus as the underlying culprit, even within the setting of a negative RADT and a negative throat culture. Enhanced awareness around the limitations of RADTs and throat cultures and the temporal utility of ASO titers can aid clinicians in elucidating the underlying cause of EN and help guide them to make prompt, effective management decisions.
AUTHORS:
Romina Garakani, BS1,2 • Zane Sejdiu, BS1,2 • Hannah Kahn, BS3 • Aspen Trautz, MD1 • Blake E. Brooks, MD, MS1 • Mary M. Braden, MD1 • Farhaan Hafeez, MD1 • Andrew C. Krakowski, MD1
AFFILIATIONS:
1Department of Dermatology, St. Luke's University Health Network, Easton, PA, USA
2Drexel University College of Medicine, Philadelphia, PA, USA
3Temple University School of Medicine, Philadelphia, PA, USA
CITATION:
Garakani R, Sejdiu Z, Kahn H, et al. Erythema nodosum in an adolescent following acute streptococcal pharyngitis. Consultant. Published online July 16, 2025. DOI: 10.25270/con.2025.07.000004
Received December 18, 2024. Accepted April 8, 2025
DISCLOSURES:
The authors have no conflicts of interest to disclose.
ACKNOWLEDGEMENTS:
The patient’s guardian has provided informed consent for the publication of the clinical photos included in this case report.
CORRESPONDENCE:
Romina Garakani, B.S., Department of Dermatology, 1600 St. Luke's Blvd, Easton, PA 18045 (rag338@drexel.edu)
References
- Hafsi W, Badri T. Erythema nodosum. In: StatPearls. StatPearls Publishing; 2022. Updated November 28, 2022. Accessed May 10, 2023. https://www.ncbi.nlm.nih.gov/books/NBK470369/
- Hafer J, Soumekh L, Thang C. Erythema nodosum after COVID-19 vaccination in a pediatric patient. JAAD Case Rep. 2022;23:12-14. doi:10.1016/j.jdcr.2022.03.009
- Pile H, Harp T, Altman D. Atypical erythema nodosum triggered by COVID-19 infection. JAAD Case Rep. 2021;14:91-93. doi:10.1016/j.jdcr.2021.06.016
- Pérez-Garza DM, Chavez-Alvarez S, Ocampo-Candiani J, Gomez-Flores M. Erythema nodosum: a practical approach and diagnostic algorithm. Am J Clin Dermatol. 2021;22(3):367-378. doi:10.1007/s40257-021-00592-w
- Paller AS, Mancini AJ. The hypersensitivity syndromes. In: Hurwitz Clinical Pediatric Dermatology. 5th ed. Elsevier; 2016:485-487.
- Dubois C, Smeesters PR, Refes Y, et al. Diagnostic accuracy of rapid nucleic acid tests for group A streptococcal pharyngitis: systematic review and meta-analysis. Clin Microbiol Infect. 2021;27(12):1736-1745. doi:10.1016/j.cmi.2021.04.021
- Hayes CS, Williamson H. Management of group A beta-hemolytic streptococcal pharyngitis. Am Fam Physician. 2001;63:1557-1564. Published correction appears in Am Fam Physician. 2002;65:1282.


