Peer Reviewed
Alopecia Areata Possibly Secondary to COVID-19 Vaccine
Introduction. A 56-year-old Black man presented at the dermatology clinic with patchy scalp hair loss that began 4 months after receiving his second COVID-19 vaccination.
History. The patient’s past medical history was remarkable for essential hypertension and type 2 diabetes mellitus. He presented with patchy hair loss on the scalp shortly after receiving a second COVID-19 vaccination.
The patient initially received the Ad26.COV2.S (Johnson & Johnson, New Brunswick, NJ, USA) COVID-19 vector vaccine and denied any adverse events aside from injection site tenderness. He then received the COVID-19 BNT162b2 messenger RNA vaccine Pfizer (New York, NY, USA) 4 months later in December 2021. (Figure 1)
Four months after his second vaccination, the patient noticed patches of hair loss. The patient denied any changes in medications, receiving other vaccinations, abnormal amount of stress, trauma, infection or illness, or hospitalization 6 months prior to onset of the symptoms. He denied any personal history or family history of hair loss. He also denied any personal or family history of thyroid or autoimmune conditions. The patient’s diabetes was well controlled with glycosylated Hemoglobin A1c of 6.5% 2 months prior to his initial visit.
Soon after noticing his hair loss, the patient presented to his local urgent care facility, where he was initially treated with oral griseofulvin, ketoconazole cream, and shampoo. Although he reported poor compliance with the initial regimen, his condition did not improve. In fact, it progressively worsened during the past year.

Figure 1. Vaccination card revealed the patient received Johnson & Johnson vaccine on August 05, 2021 and subsequent Pfizer/BioNTech (BNT162b2) booster on December 09, 2021.
Physical examination. Although the patient showed no acute stress, his skin examination showed several, large, polycyclic nonscarring alopecia patches on the scalp. (Figure 2) Other areas of the body, including the eyebrows and beard, were not affected.
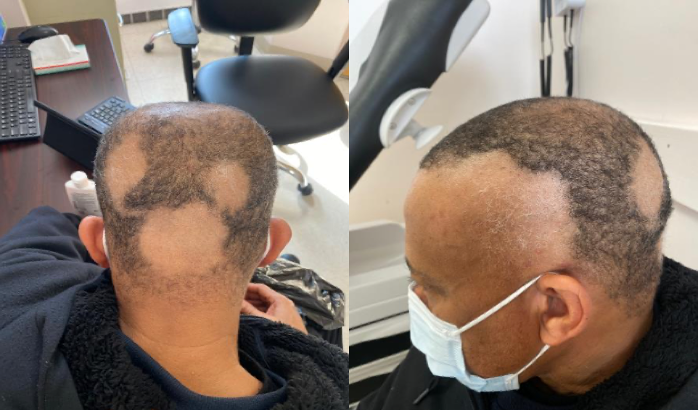
Figure 2. Several nonscarring alopecia patches at baseline.
Diagnostic testing. The location and shape of the alopecia patches suggested a clinical diagnosis of alopecia areata (AA). The patient's lack of response to oral and topical antifungal therapies also made a fungal infection unlikely.
Differential diagnosis. The differential diagnosis for patchy, non-scarring hair loss includes AA, tinea capitis, trichotillomania, and secondary syphilis. On clinical examination, the alopecia patches lacked a moth-eaten border, greasy scale, broken hair and pigmented dots. This constellation of findings, along with the onset following COVID-19 vaccination, makes AA the most likely diagnosis. The patient's age (56 years) is atypical for alopecia associated with secondary syphilis or trichotillomania.
Treatment and management. The patient was subsequently treated with monthly intralesional Kenalog 2.5 mg/mL injections, topical minoxidil foam twice daily, and fluocinonide solution daily for 6 months without clinically significant improvement. The patient was then started on oral baricitinib 4 mg once daily. Baseline laboratory tests were obtained, and the patient tested negative for hepatitis B, hepatitis C, and tuberculosis.
Outcome and follow-up. At his 9-month follow-up, the patient showed significant regrowth of black and white hair. He had a total of nine sessions of intralesional Kenalog injection. (Figure 3) The patient has been prescribed oral baricitinib 4 mg daily, as well as oral minoxidil 2.5 mg daily, intralesional Kenalog injection, and topical minoxidil and for almost 2 years.
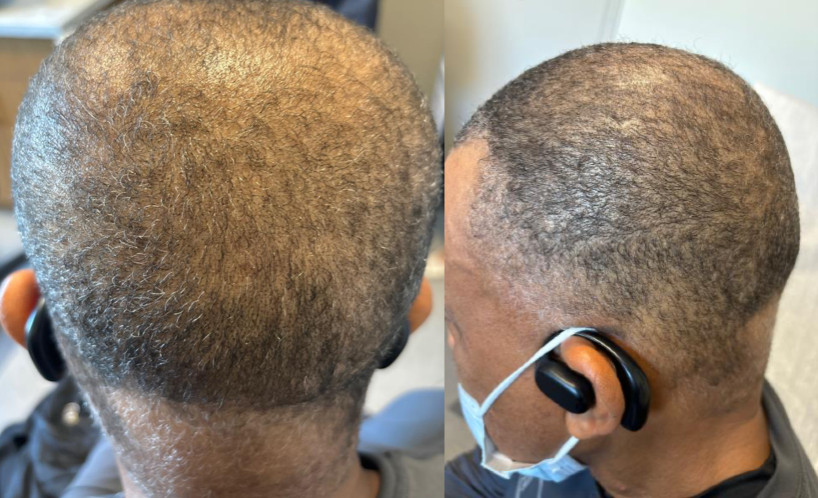
Figure 3. The patient showed significant improvement in alopecia after 9 months of 4 mg oral baricitinib.
Discussion. AA is an autoimmune condition that manifests as non-scarring hair loss with intact hair follicles affecting the scalp and other hair-bearing areas. It has a lifetime incidence of 2.1%. Microscopically, lymphocytic infiltrate around the bulb in anagen phase, termed “swarm of bees” can be appreciated sometimes. Several forms of AA have been identified in the literature, including alopecia totalis (AT; loss of all scalp hair), alopecia universalis (AU; loss of all body hair), ophiasis (symmetric, band-like hair loss pattern of the occipital, temporal, and parietal regions of the scalp), sisaipho (the reverse of ophasis pattern, involves the crown area but sparing the occipital, temporal, and parietal region), diffuse AA or AA incognito (diffuse shredding), patchy AA, and linear AA. Under trichoscopy, wide distal shaft size, and thin proximal shaft, described as an “exclamation mark.” Associated conditions of AA include atopic dermatitis, thyroid disease, regular nail pitting, and longitudinal ridging.
Black individuals have a higher risk of developing alopecia areata; however, no definitive genetic associations have been identified in the literature. A recent study found that specific genetic variations (single nucleotide polymorphisms [SNP]) associated with increased or decreased risk of AA in European/White populations did not show the same effects in other ethnic groups, such as Black and Hispanic individuals. These differences in SNP associations may help explain the higher prevalence of AA observed in Black populations, but more research is needed.13,14
Indeed, the potential link between COVID-19 vaccines and autoimmune conditions remains a topic of debate. This case study details refractory AA following heterologous vaccine administration of COVID-19 vaccines from two pharmaceutical companies. The patient's condition showed significant improvement following oral Janus Kinase (JAK) inhibitor therapy, after intralesional Kenalog injections and topical minoxidil did not improve the patient’s condition.
Neither a prior phase II/III trial of BNT162b2 mRNA COVID-19 Vaccine nor a phase III trial of Ad26.COV2.S vaccine reported any adverse events on hair loss or alopecia.4,5 However, in a systematic review of 25 articles involving 51 patients, AA was reported in 52.9% of cases, with the Pfizer vaccine being the most implicated. AA typically developed within 1 month of the first dose, with incidence gradually declining thereafter.6 A health care registry agency in the United Kingdom reported a total of 154 cases of AA, AT, or AU after COVID-19 vaccine; 50% were from Pfizer/BioNTech, and 10% with the mRNA-1273 Moderna vaccines. The US Centers for Disease Control and Prevention Vaccine Adverse Event Reporting System database showed a total of 126 incidence (114 of AA, 1 of AT, and 11 of AU). The vast majority were Pfizer/BioNTech (66%) and Moderna (29%). The actual numbers may be higher due to the under-reporting of mild alopecia cases.7
Several studies have researched the side effects of heterologous COVID-19 vaccinations; however, alopecia was never addressed as one of the common side effects.8 9 To the best of the author’s knowledge, this is the first report of AA development after mixing Pfizer/ BioNTech and Moderna mRNA vaccines. Various reports have discussed the role of COVID-19 vaccine in association with a myriad of rare autoimmune cutaneous conditions, including but not limited to, antineutrophil cytoplasmic autoantibodies-associated vasculitis, giant cell arteritis, adult onset still disease, systemic lupus erythematosus, vitiligo, and etc.10 11 An epidemiological study in Korea with more than 3 million participants showed the risk of AA and alopecia totalis are not significantly higher in those who received the mRNA COVID-19 vaccine compared with controls.12
This case highlights the need for clinicians to consider heterologous COVID-19 vaccination as a potential, albeit rare, trigger for autoimmune hair loss and underscores the efficacy of JAK inhibitors like baricitinib in treating refractory cases. Although the exact mechanism remains unclear, one possibility is that vaccination activates both innate and adaptive immune responses, leading to the release of proinflammatory cytokines such as interferon-gamma (IFN-γ), interleukin-6 (IL-6), among others. IFN-γ is believed to contribute to the collapse of immune privilege in hair follicles, potentially triggering autoimmune-mediated hair loss.6
Conclusion. Clinicians should maintain awareness of AA as a potential adverse event following COVID-19 vaccination, particularly in cases of heterologous administration. Oral baricitinib may offer effective treatment for refractory cases following vaccine-associated AA.
AUTHORS:
Haowei Han, DO 1 • Brian How, DO1 • Suzanne Sirota-Rozenberg, DO1
AFFILIATIONS:
1Department of Dermatology, St. John’s Episcopal Hospital
CITATION:
Han H, How B, Sirota-Rozenberg S. Alopecia areata possibly secondary to COVID vaccine. Consultant. Published online October 22, 2025. doi: 10.25270/con.2025.12.000002
Received April 25, 2025. Accepted July 3, 2025.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Haowei Han, DO, 3-27 Beach 19th St, Far Rockaway, NY 11691 (email: haoweihansb@gmail.com)
References
- Alhanshali L, Buontempo MG, Lo Sicco KI, Shapiro J. Alopecia areata: burden of disease, approach to treatment, and current unmet needs. Clin Cosmet Investig Dermatol. 2023;16:803-820. doi:10.2147/CCID.S376096
- Asad U, Wallis D, Tarbox M. Ophiasis alopecia areata treated with microneedling. Proc (Bayl Univ Med Cent). 2020;33(3):413-414. doi:10.1080/08998280.2020.1753456
- Sibbald C. Alopecia areata: an updated review for 2023. J Cutan Med Surg. 2023;27(3):241-259. doi:10.1177/12034754231168839
- Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383(27):2603-2615. doi:10.1056/NEJMoa2034577
- Sadoff J, Gray G, Vandebosch A, et al. Safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19. N Engl J Med. 2021;384(23):2187-2201. doi:10.1056/NEJMoa2101544
- Zhu Y, Ouyang X, Zhang D, et al. Alopecia areata following COVID-19 vaccine: a systematic review. Eur J Med Res. 2024;29(1):356. doi:10.1186/s40001-024-01956-8
- Birkett L, Singh P, Mosahebi A, Dhar S. Possible associations between alopecia areata and COVID-19 vaccination and infection. Aesthet Surg J. 2022;42(11):NP699-NP702. doi:10.1093/asj/sjac165
- Van Hung P, Nguyen TD, Ha LT, Toi PL, Tram TH. Common adverse events from mixing COVID-19 vaccine booster in Hanoi, Vietnam. Vaccines (Basel). 2023;11(6):1097. doi:10.3390/vaccines11061097
- Rashedi R, Samieefar N, Masoumi N, Mohseni S, Rezaei N. COVID-19 vaccines mix-and-match: the concept, the efficacy and the doubts. J Med Virol. 2022;94(4):1294-1299. doi:10.1002/jmv.27463
- Guo M, Liu X, Chen X, Li Q. Insights into new-onset autoimmune diseases after COVID-19 vaccination. Autoimmun Rev. 2023;22(7):103340. doi:10.1016/j.autrev.2023.103340
- Ciccarese G, Drago F, Boldrin S, Pattaro M, Parodi A. Sudden onset of vitiligo after COVID-19 vaccine. Dermatol Ther. 2022;35(1):e15196. doi:10.1111/dth.15196
- Ju HJ, Lee JY, Han JH, Lee JH, Bae JM, Lee S. Risk of autoimmune skin and connective tissue disorders after mRNA-based COVID-19 vaccination. J Am Acad Dermatol. 2023;89(4):685-693. doi:10.1016/j.jaad.2023.05.017
- Lee H, Jung SJ, Patel AB, Thompson JM, Qureshi A, Cho E. Racial characteristics of alopecia areata in the United States. J Am Acad Dermatol. 2020;83(4):1064‑1070. doi:10.1016/j.jaad.2019.06.1300
- Gupta AK, Economopoulos V. Management and genetics of alopecia areata within the usa: a cross‑sectional study of all of us. Skin Appendage Disord. 2025;11(4):344‑354. doi:10.1159/000545194
©2025 HMP Global. All Rights Reserved. Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


