Successful Valve-in-Valve Transcatheter Aortic Valve Replacement in a Nonagenarian With a Degenerated Bioprosthetic Prosthesis
Authors: Jeremy Freiwald, MD, Paul Zack, MD, Mayra Guerrero, MD
Abstract: Calcific aortic stenosis is a progressive cardiac valvular abnormality that is particularly prevalent in elderly adults. Historically, an open-heart procedure that replaced the damaged aortic valve with an artificial one was the only definitive treatment option; however, due to its invasiveness and high surgical risks, the oldest old (≥85 years) often declined these procedures or were deemed unsuitable candidates. In 2011, transcatheter aortic valve replacement (TAVR) was introduced as a minimally invasive option for treating calcific aortic stenosis in patients who would otherwise be ineligible for cardiac surgery. This procedure can also be used as an alternative to bioprosthetic valve replacement in select patients whose implanted valve has degenerated; this is known as a valve-in-valve procedure. In this article, the authors report the case of a community-dwelling nonagenarian with symptomatic critically severe aortic stenosis that was successfully treated with valve-in-valve TAVR. After the procedure, the patient experienced excellent improvement in her symptoms and was able to return to independent living. This case shows that TAVR can serve as an effective and safer alternative to conventional cardiac surgery for carefully selected elderly patients with severe aortic stenosis, even if they have previously received a bioprosthetic valve.
Keywords: Calcific aortic stenosis, cardiac valve replacement surgery, degenerated bioprosthetic aortic valve, severe aortic stenosis, transcatheter aortic valve replacement, TAVR, valve-in-valve TAVR.
The steadily increasing age of the US population has led to new challenges for physicians treating elderly patients, particularly the oldest old (≥85 years), due to their high prevalence of comorbidities, polypharmacy, and frailty. One such challenge is the management of calcific aortic stenosis (AS), a progressive cardiac valvular abnormality that becomes more prevalent with increasing age.1 It has been reported that the condition affects 5% of the US general population by age 70 and 20% of this population by age 90.2 The only treatment is surgical intervention, as no medications are available to manage the condition. Without treatment, severe AS has been found to have a mortality of greater than 50% during the 2-year period following the onset of symptoms.3 Although conventional aortic valvular replacement surgery can be considered, the oldest old often refuse surgery or are deemed to have an unacceptably high surgical risk. Advances in medical care have led to the development of transcatheter aortic valve replacement (TAVR), a minimally invasive approach to aortic valve replacement that was initially introduced for patients deemed ineligible for conventional cardiac surgery.4 In this article, we report the case of a 93-year-old woman with symptomatic critically severe AS who successfully underwent TAVR after having received a bioprosthetic valve 17 years earlier. Following the valve-in-valve procedure, she experienced excellent symptomatic improvement. Our case demonstrates that this procedure can be successful even in the oldest old with previously implanted bioprosthetic valves; thus, patients should be carefully screened to see if they are candidates, rather than having their eligibility discounted based on age alone.
_____________________________________________________________________________________________________________________________________________________________________
RELATED CONTENT
Transcatheter Aortic Valve Replacement for Aortic Stenosis
TAVI Guideline Update from the AHA/ACC
_____________________________________________________________________________________________________________________________________________________________________
Case Report
A 93-year-old woman with New York Heart Association class III heart failure was seen in our cardiology office following the recent onset of progressive dyspnea upon engaging in mild activities. At age 76, severe AS was diagnosed and she underwent conventional surgical aortic valve replacement with a 21-mm Carpentier-Edwards perimount aortic pericardial bioprosthesis. Four years later, she underwent pacemaker implantation to treat bradycardia that had caused near-syncope and intracoronary stent placement to treat symptomatic single-vessel coronary artery disease (CAD).
Six months prior to her current office visit, the patient had been able to perform moderate activities, including light housework and shopping for groceries with only mild dyspnea. She now reported progressive shortness of breath with the minimal exertion of walking within her home, as well as occasional episodes of orthopnea and shortness of breath at rest. She reported no chest pain upon exertion or at rest and no episodes of syncope or near-syncope, but noted experiencing occasional palpitations and lightheadedness. The patient’s cardiac risk factors included a history of hyperlipidemia, hypertension, and a family history of premature CAD. Her medications included an angiotensin-converting enzyme inhibitor, a diuretic, a statin, a potassium supplement, and aspirin.
On physical examination, the patient had a blood pressure of 140/82 mm Hg, a heart rate of 78 beats per minute, and a respiration rate of 18 breaths per minute. Inspection of her neck showed jugular venous distention. Chest auscultation revealed bilateral rhonchi at the lung bases. On cardiac examination, there was a regular rhythm with a grade III/VI systolic murmur heard loudest at the left sternal border with transmission across the precordium and to the neck. On extremity examination, mild pedal edema was present bilaterally. An electrocardiogram showed a paced rhythm and a chest radiograph revealed congestive changes in the lung fields bilaterally. Laboratory examination demonstrated a normal complete blood count, electrolytes, and renal function, but an elevated brain-type natriuretic peptide of 1128 pg/mL (normal, <107 pg/mL), which is consistent with congestive heart failure. A two-dimensional transthoracic echocardiogram with color Doppler flow mapping demonstrated left ventricular hypertrophy with normal left ventricular systolic function and severe stenosis of the bioprosthetic aortic valve (Figures 1 and 2). There was a peak systolic gradient across the valve of 81 mm Hg, a mean aortic valve gradient of 43 mm Hg, and a calculated aortic valve area of less than 0.5 cm2.
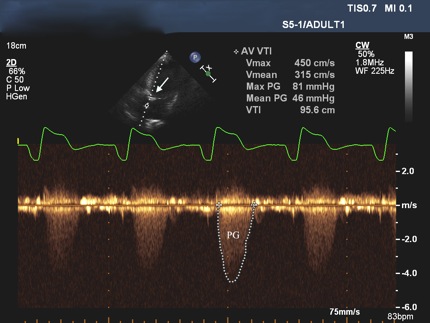
Figure 1. Echocardiogram showing a peak systolic gradient across the bioprosthetic aortic valve (arrow) of 81 mm Hg, a mean aortic gradient of 46 mm Hg, and an aortic valve area of less than 0.5 cm2.
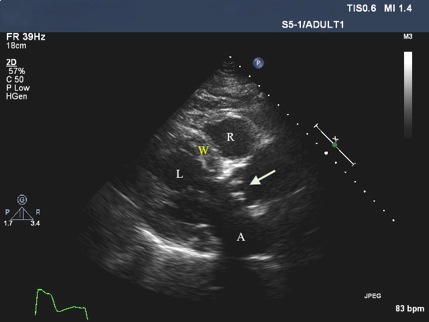
Figure 2. Echocardiogram showing the right ventricle (R) and left ventricle (L). The thickened left ventricular wall is consistent with left ventricular hypertrophy (W). The left atrium (A) demonstrates normal size limits. There is evidence of calcification of the bioprosthetic aortic valve (arrow).
(Case report continues on next page)
Based on these findings, we recommended immediate hospitalization for treatment of congestive heart failure and possible cardiac catheterization studies, which the patient declined. We increased her dose of oral diuretic and initiated home oxygen supplementation, but she developed worsening dyspnea, including severe orthopnea and episodes of paroxysmal nocturnal dyspnea. Subsequently, she agreed to hospitalization for more intensive treatment of her congestive heart failure. Despite administration of intravenous diuretics, her condition did not improve, and additional diuretic treatment was limited by the development of borderline hypotension. Because the patient and her family did not want to consider another open-heart surgery, the patient’s findings were reviewed with the multidisciplinary structural heart disease team at a nearby tertiary care center. The team determined the patient to be a suitable candidate for TAVR under the Global Valve-in-Valve Registry of the PARTNER (Placement of Aortic Transcatheter Valves) II trial5 using the Edwards Sapien XT valve. This option was discussed with the patient and her family, and they agreed to proceed with hospital transfer for the procedure.
_____________________________________________________________________________________________________________________________________________________________________
RELATED CONTENT
Aortic Atheroma
Ruptured Aortic Aneurysm
_____________________________________________________________________________________________________________________________________________________________________
Shortly after transfer, the patient underwent valve-in-valve TAVR within the previous bioprosthetic aortic valve while under general anesthesia (Figures 3 and 4). The procedure was performed via a transapical approach, as she was not a candidate for a transfemoral approach due to the small size of her femoral arteries. To enable left ventricular transapical access, an approximately 2- to 3-inch left thoracotomy was performed. An 18-gauge needle was introduced in the distal anterolateral wall of the left ventricle near the apex, and a 0.035-inch J-tipped guidewire was introduced in the left ventricle. The needle was removed and a 7 French (Fr) sheath was introduced in the left ventricle over the guidewire. The guidewire was then advanced across the aortic valve into the ascending aorta. A 6-Fr Judkins right number 4 (JR4) catheter was introduced over the guidewire, the guidewire and JR4 catheter were advanced into the descending aorta, and the guidewire was exchanged for a 0.035-inch Amplatz extra-stiff guidewire. The JR4 catheter and 7 Fr sheath were removed and the Edwards 24 Fr Ascendra sheath was introduced in the left ventricle. A 23-mm Edwards Sapien XT valve was introduced over the guidewire across the bioprosthetic aortic valve and was successfully deployed under rapid ventricular pacing at 180 beats per minute. The delivery catheter was then removed. Simultaneous left ventricular and aortic pressures revealed a mean aortic valve gradient of only 5 mm Hg. The valve function was evaluated by transesophageal echocardiography and found to be normal without paravalvular regurgitation. The Ascendra sheath was removed, and the left ventriculotomy and left thoracotomy were successfully closed. The patient was transferred to the cardiovascular intensive care unit and was extubated soon after the procedure. She had an uneventful recovery. Within 2 days following the procedure, the patient was able to ambulate independently. Her antihypertensive and diuretic medication doses were reduced from her prehospitalization doses and she was subsequently discharged to home.
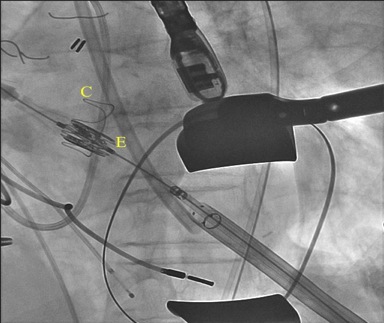
Figure 3. Fluoroscopy demonstrating a 23-mm Edwards Sapien XT valve (E) positioned across a 21-mm Carpentier-Edwards perimount aortic pericardial bioprosthesis (C) via a transapical approach.
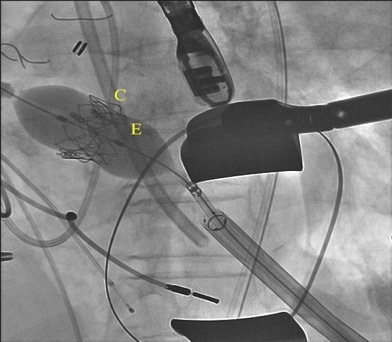
Figure 4. Valve-in-valve deployment of a 23-mm Edwards Sapien XT transcatheter valve (E) to replace the degenerated 21-mm Carpentier-Edwards perimount aortic pericardial bioprosthesis (C) via a transapical approach.
On a follow-up visit in our cardiology office 6 weeks after the procedure, the patient had resumed her previous daily activities, including light housework. She reported no chest pain or shortness of breath. On physical examination, her vital signs were satisfactory, her lungs were clear to auscultation, and cardiac auscultation revelaed only a soft systolic murmur. She had no pedal edema.
(Discussion on next page)
Discussion
Calcific AS, which is the most common etiology of aortic stenosis in older individuals, is caused by age-related deposition of calcium on the aortic valvular leaflets.1 Although only 10% of patients with calcific AS will progress to severe AS,6 older age is associated with progression.7 As the population continues to age, AS will become more prevalent, and the burden of AS is anticipated to double within the next 50 years.8 Therefore, it is important for healthcare providers to be aware of all of the available treatment options and counsel their patients accordingly.
Until recently, an open-heart procedure replacing the defective aortic valve was the only definitive option for treating AS. In younger patients, the surgical risk of conventional aortic valve replacement is low, with a surgical mortality reported to be less than 1% at high-volume tertiary referral centers.9 In contrast, the surgical mortality for nonagenarians undergoing conventional aortic valve replacement surgery has been reported to be greater than 11%,10 with a hospital morbidity of greater than 70%, even in patients selected to reduce comorbidities other than age.11
In November 2011, a new option for treating AS became available when the US Food and Drug Administration approved TAVR as a minimally invasive approach for use in patients who are not candidates for open-heart procedures, and it expanded its approval in October 2012 to include high-risk patients (ie, those eligible for open-heart procedures but at high risk for serious surgical complications or death).4 Despite this high-risk cohort, the success of TAVR has been reported to be greater than 90%, with a procedural mortality less than 2% and a hospital morbidity less than 30%.12 Complications have been infrequently reported, but include possible myocardial injury, cardiac conduction disturbances, paravalvular aortic regurgitation, paravalvular leak, vascular injury, and neurological events, including stroke.13 The frequency of these complications is expected to further decrease as both TAVR operator experience and device technology improve.
TAVR has also been used for several off-label indications, including for patients with a degenerated implanted bioprosthetic valve and for those experiencing a paravalvular leak following a previous TAVR.14 These indications involvevalve-in-valve procedures, which are examined in greater detail in the section that follows.
_____________________________________________________________________________________________________________________________________________________________________
RELATED CONTENT
Dissecting Thoracic Aortic Aneurysm With Left-Sided Hemothorax
Aortic Stenosis: A Focused Review on the Elderly
_____________________________________________________________________________________________________________________________________________________________________
Valve-in-Valve Procedures
Our patient had initially undergone conventional aortic valve replacement surgery at age 76. As occasionally occurs, her bioprosthetic aortic valve became critically calcified and stenotic during the ensuing 17-year period. In our patient, conventional aortic valve replacement surgery would have required replacement of an existing bioprosthetic valve, a factor that would be expected to further increase the complexity of the surgery and the potential risk. Subsequently, she and her family declined this procedure; however, they agreed to the valve-in-valve procedure when they were made aware of the patient’s candidacy. Instead of replacing the defective valve, this procedure enabled a new catheter-based valve to be implanted directly inside of the patient’s already implanted bioprosthetic valve.
The valve-in-valve procedure to address a previously implanted bioprosthetic valve that has degenerated was assessed in the Global Valve-in-Valve Registry, which included 202 patients (aged 77.7±10.4 years; 52.5% men) treated at 38 centers worldwide.15 Patients required the procedure due to stenosis of their bioprosthesis (42%), regurgitation (34%), or a combination of these (24%). Procedural success was achieved in 93.1% of cases. The primary safety and efficacy concerns observed included device malposition (15.3% of cases) and ostial coronary obstruction (3.5% of cases). At 30-day follow-up, 84.1% of patients were at NYHA functional class I/II heart failure and all-cause mortality was 8.4%. At 1-year follow-up, which was obtained in 87 patients, the survival rate was 85.8%.15
Valve-in-valve procedures have also been used to treat complications following a previous TAVR, particularly paravalvular leak, which typically results from malpositioning of the prosthesis, as the prosthesis cannot be repositioned once deployed.14 A recent substudy of PARTNER that assessed the outcomes of valve-in-valve procedures in patients who received TAVR (63 of 2554 patients; 2.47%) found that these procedures were an independent predictor of 1-year cardiovascular mortality (hazard ratio, 1.86; 95% confidence interval, 1.03 to 3.38; P=.041)16; however, most of these valve-in-valve procedures were done in an acute, emergent setting during or immediately after the first TAVR, which likely contributed to the worse outcomes. The study authors also concluded that multiple valve implants could be avoided with judicious procedural planning.16
Conclusion
Physicians caring for the oldest old face the challenge of balancing the fragility of advanced age with the occasional need for invasive diagnostic procedures or treatment. In carefully selected patients with severe AS, the recent availability of TAVR provides an effective and safer alternative to conventional cardiac surgery, including for individuals who have previously received a bioprosthetic aortic valve that has degenerated. Our case report shows that age alone should not preclude a patient from receiving this potentially beneficial treatment, as our nonagenarian patient became symptom-free and was able to return to independent living following the procedure.
References
1. Selzer A. Changing aspects of the natural history of valvular aortic stenosis. N Engl J Med. 1987;317(2):91-98.
2. Transcatheter Aortic Valve Replacement for Aortic Stenosis: What Geriatricians Should Know and How They Can Help. www.consultant360.com/articles/transcatheter-aortic-valve-replacement-aortic-stenosis-what-geriatricians-should-know-and. Published June 21, 2013. Accessed June 2, 2014.
3. Schwarz F, Baumann P, Manthey J, et al. The effect of aortic valve replacement on survival. Circulation. 1982;66(5):1105-1110.
4. Leon MB, Smith CR, Mack M, et al; PARTNER Trial Investigators. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363(17):1597-1607.
5. Svensson LG, Tuzcu M, Kapadia S, et al. A comprehensive review of the PARTNER trial. J Thorac Cardiovasc Surg. 2013;145(3 Suppl):S11-16.
6. Freeman RV, Otto CM. Spectrum of calcific aortic valve disease: pathogenesis, disease progression, and treatment strategies. Circulation. 2005;111(24):3316-3326.
7. Novaro GM, Katz R, Aviles RJ, et al. Clinical factors, but not C-reactive protein, predict progression of calcific aortic-valve disease: the Cardiovascular Health Study. J Am Coll Cardiol. 2007;50(20):1992-1998.
8. Iung B, Vahanian A. Degenerative calcific AS: a natural history. Heart. 2012;98(suppl 4):iv7-13.
9. Bonow RO, Carabello BA, Chatterjee K, et al; American College of Cardiology/American Heart Association Task Force. 2008 Focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2008;118(15):e523-661.
10. Bridges CR, Edwards FH, Peterson ED, Coombs LP, Ferguson TB. Cardiac surgery in nonagenarians and centenarians. J Am Coll Surg. 2003;197(3):347-357.
11. Samuels LE, Sharma S, Morris RJ, et al. Cardiac surgery in nonagenarians. J Card Surg. 1996;11(2):121-127.
12. Piazza N, Grube E, Gerckens U, et al. Procedural and 30-day outcomes following transcatheter aortic valve implantation using the third generation (18 Fr) corevalve revalving system: results from the multicentre, expanded evaluation registry 1-year following CE mark approval. EuroIntervention. 2008;4(2):242-249.
13. Généreux P, Head SJ, Van Mieghem NM, et al. Clinical outcomes after transcatheter aortic valve replacement using valve academic research consortium definitions: a weighted meta-analysis of 3,519 patients from 16 studies. J Am Coll Cardiol. 2012;59(25):2317-2326.
14. Shah N, Singla S, Deshmukh A, Sachdeva R. Can we use valve-in-valve transcatheter aortic valve replacement in an unstable patient? J Invasive Cardiol. 2013;25(11). www.invasivecardiology.com/articles/can-we-use-valve-valve-transcatheter-aortic-valve-replacement-unstable-patient-0. Accessed June 3, 2014.
15. Dvir D, Webb J, Brecker S, et al. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in-valve registry.Circulation. 2012;126(19):2335-2344.
16. Makkar RR, Jilaihawi H, Chakravarty T, et al. Determinants and outcomes of acute transcatheter valve-in-valve therapy or embolization: a study of multiple valve implants in the U.S. PARTNER trial (Placement of AoRTic TraNscathetER Valve Trial Edwards SAPIEN Transcatheter Heart Valve). J Am Coll Cardiol. 2013;62(5):418-430.
Disclosures:
Dr. Guerrero is a consultant for Edwards Lifesciences, Irvine, CA.
Jeremy Freiwald, MD1; Paul Zack, MD1,2; Mayra Guerrero, MD3,4
Affiliations:
1McLaren Cardiovascular Group, McLaren–Greater Lansing Hospital, Lansing, MI
2Michigan State University College of Human Medicine, East Lansing, MI
3Henry Ford Heart and Vascular Institute, Detroit, MI
4Wayne State University Medical School, Detroit, MI


