Two-Year Study Confirms Booster Doses Strengthen COVID-19 Immunity Among Those With CLL, Other Immunocompromised Patient Groups
The results of a 2-year follow-up of the COVAXID clinical trial indicate a clinical benefit for the continued administration of SARS-CoV-2 mRNA vaccine booster doses among immunocompromised patients. Conducted at Karolinska Institutet in Stockholm and involving 355 participants, the study tracked immunogenicity over an extended period following initial vaccination with the Pfizer-BioNTech BNT162b2 vaccine.
The COVAXID trial originally enrolled 539 adults with either primary or secondary immunodeficiency conditions, as well as healthy controls. Participants in the 2-year extension phase received up to six doses of SARS-CoV-2 mRNA vaccines in line with national recommendations, including monovalent and later bivalent formulations. Immune responses were evaluated through binding antibody titres, pseudo-neutralisation capacity against both ancestral and Omicron SARS-CoV-2 variants, and cellular immunity assays.
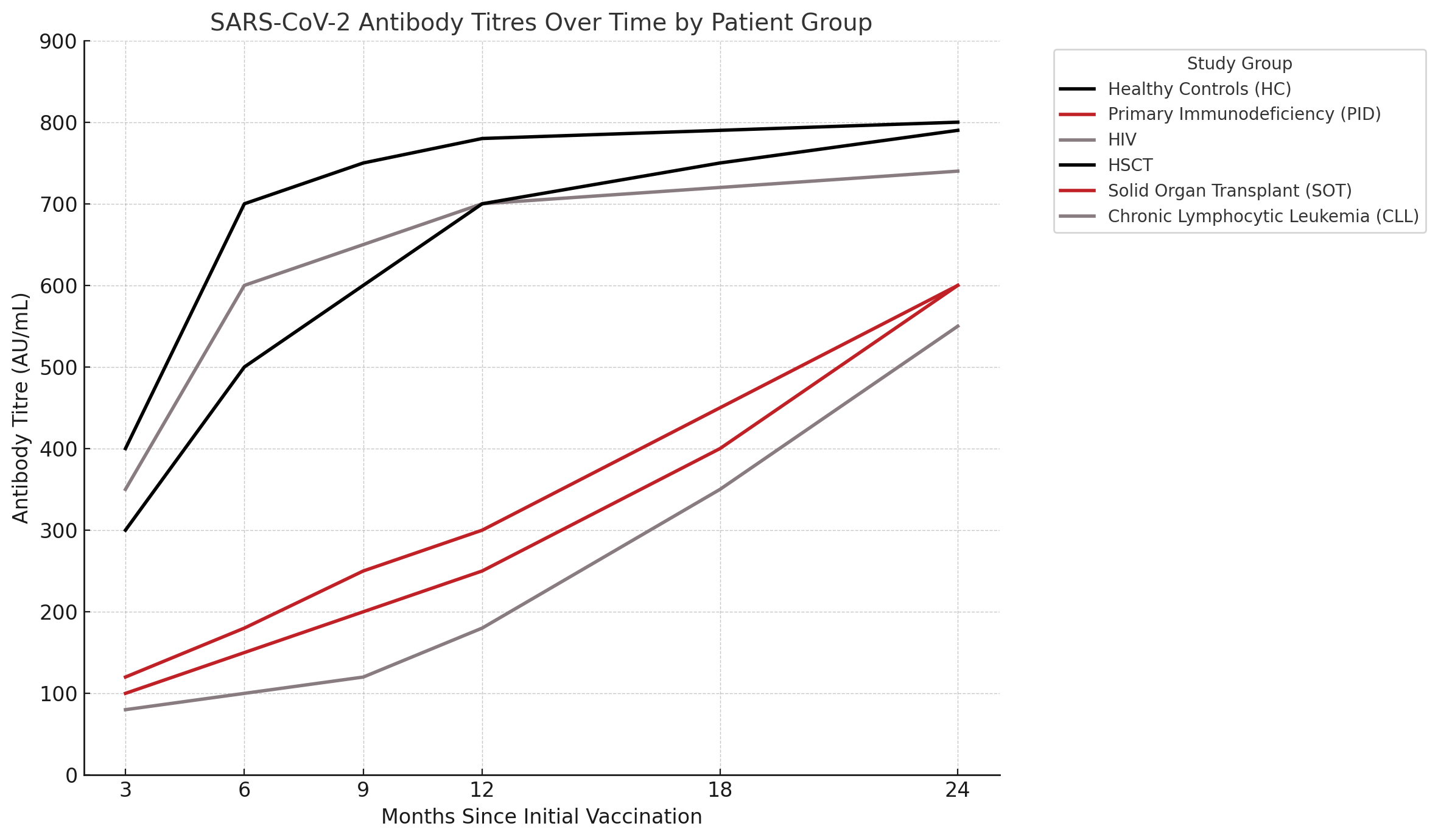
Two distinct patterns of immune response emerged. Healthy controls, individuals with HIV, and hematopoietic stem cell transplant recipients reached a plateau in antibody titres by the 12-month mark, with sustained levels thereafter. In contrast, participants with common variable immunodeficiency, those receiving immunosuppressive therapies such as ibrutinib or mycophenolate mofetil, and patients in the solid organ transplant and chronic lymphocytic leukemia groups exhibited gradual increases in antibody levels over time. By the 24-month point, these previously low-responding groups had reached levels comparable to their healthier counterparts.
While antibody binding remained strong across all groups, neutralizing capacity against Omicron variants remained lower than against the ancestral virus, even at 2 years. Despite this, repeated booster doses and natural SARS-CoV-2 infection were both positively correlated with stronger neutralization responses, supporting a cumulative immunity-building effect. Some patients with conditions that limit antibody production, such as X-linked agammaglobulinemia, still demonstrated increases in antibody titres over time—potentially aided by commercial immunoglobulin products—although these effects were not statistically significant across the cohort.
Cellular immune responses were preserved across all groups and were strikingly similar in magnitude when measured against both the wild-type virus and the Omicron XBB.1.5 variant. CD4+ and CD8+ T cell responses were observed even in patients who did not seroconvert, reinforcing the idea that T cell immunity may play a critical protective role when humoral responses are weak or absent.
Multivariate analyses revealed that the number of antigen exposures—through vaccination and/or infection—was the most robust positive predictor of immune response. In contrast, concurrent immunosuppressive treatments during vaccination, particularly with ibrutinib and mycophenolate mofetil, were strongly associated with diminished antibody production. Underlying immunosuppressive disease states also negatively impacted responses. Age and immunoglobulin replacement therapy were not significant modifiers in this analysis.
No patients in the study cohort required hospitalization for COVID-19 between months 12 and 24, despite the circulation of multiple Omicron subvariants in Sweden during this period. While the precise relationship between laboratory immune markers and protection against clinical disease remains an area for further research, these data suggest that even suboptimal immune responses may confer meaningful protection from severe outcomes.
“Taken together, the present data add additional information serving to improve the management of immunocompromised patients, many of which represent risk groups for severe COVID-19,” the study authors concluded. “It underscores the importance of addressing known factors that interfere with vaccine responses in the individual management of vaccine regimens in immunocompromised individuals.”
Reference:
Chen P, Bergman P, Blennow O, et al. Real-world assessment of immunogenicity in immunocompromised individuals following SARS-CoV-2 mRNA vaccination: a two-year follow-up of the prospective clinical trial COVAXID. EBioMedicine. 2024;109:105385. doi:10.1016/j.ebiom.2024.105385


