Tolebrutinib Reduces Disability Progression in Nonrelapsing Secondary Progressive Multiple Sclerosis
Key Highlights
- Treatment with tolebrutinib significantly reduced the risk of sustained disability progression in patients with nonrelapsing secondary progressive multiple sclerosis (SPMS).
- The hazard ratio for confirmed disability progression sustained for at least 6 months was 0.69 in favor of tolebrutinib (P = .003).
- Serious adverse events were higher in the tolebrutinib group (15.0%) compared with placebo (10.4%), including increased liver enzyme levels.
In a phase 3, placebo-controlled trial, tolebrutinib, an oral Bruton’s tyrosine kinase (BTK) inhibitor, demonstrated a significant reduction in the risk of confirmed disability progression in patients with nonrelapsing secondary progressive multiple sclerosis (SPMS). The study found that 22.6% of participants receiving tolebrutinib experienced disability progression sustained for at least 6 months, compared with 30.7% in the placebo group (HR, 0.69; P = .003), marking a clinically meaningful benefit in a population with limited treatment options.
Nonrelapsing SPMS is characterized by gradual neurologic decline that is not driven by acute relapses. Current disease-modifying therapies have limited efficacy in halting this form of progression, which is largely driven by chronic neuroinflammation within the central nervous system (CNS). Given the unmet need for effective treatments targeting this disease mechanism, the development of therapies such as BTK inhibitors, which modulate both myeloid cells and B cells in the CNS, holds substantial promise.
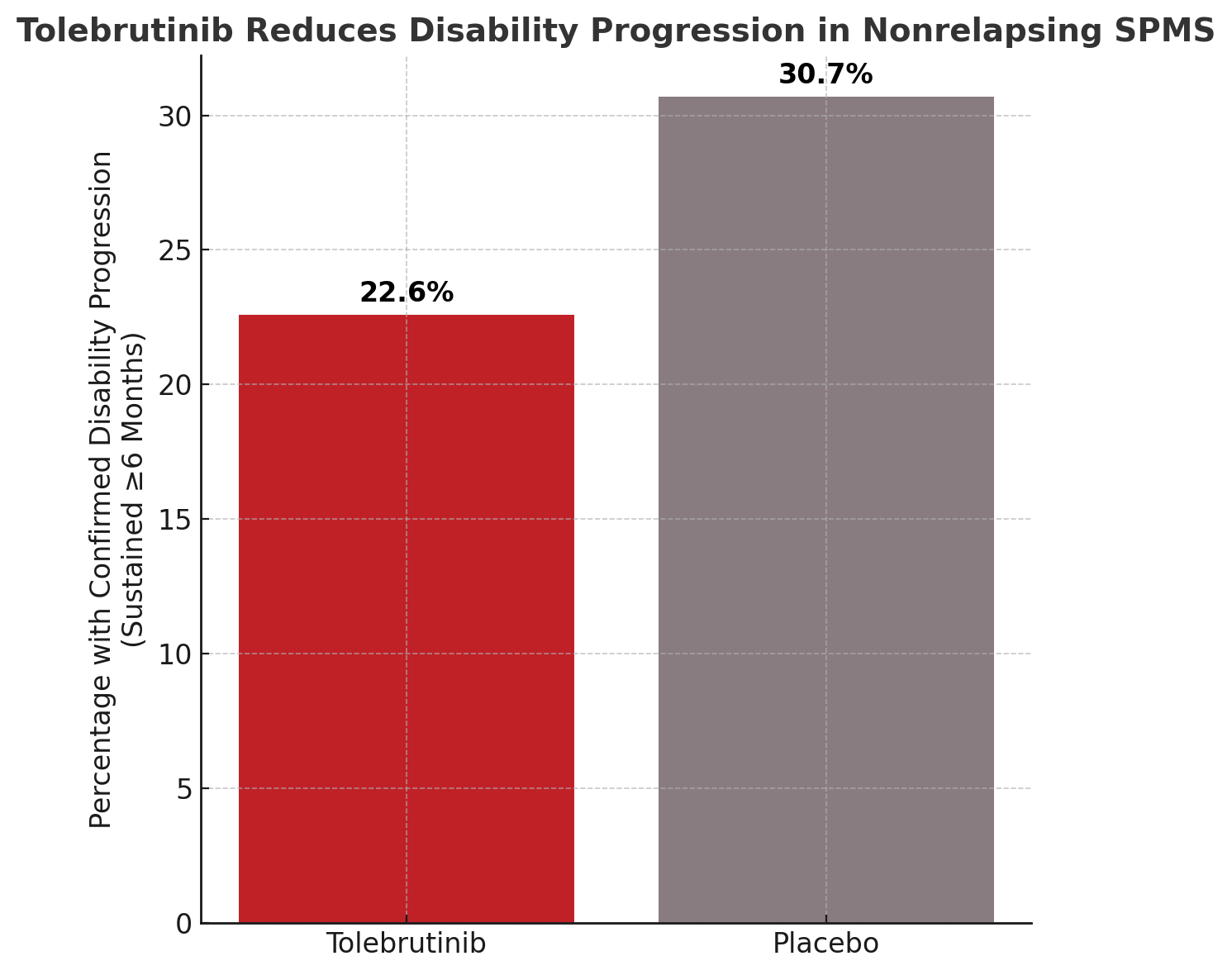
The trial enrolled 1131 participants with nonrelapsing SPMS, who were randomly assigned in a 2:1 ratio to receive either tolebrutinib (60 mg daily) or placebo. The study employed a double-blind, event-driven design, with the primary endpoint being time to confirmed disability progression sustained for at least 6 months. Participants were followed for a median of 133 weeks.
The study results showed a lower incidence of sustained disability progression in the tolebrutinib group compared with placebo (22.6% vs. 30.7%), corresponding to a 31% relative risk reduction. Serious adverse events occurred more frequently with tolebrutinib (15.0%) than placebo (10.4%). Elevated alanine aminotransferase levels (>3 times the upper limit of normal) were observed in 4.0% of patients treated with tolebrutinib versus 1.6% of those on placebo, indicating a need for liver function monitoring during treatment.
“In participants with nonrelapsing secondary progressive multiple sclerosis, the risk of disability progression was lower among those who received treatment with tolebrutinib than among those who received placebo,” the study authors concluded.
Reference:
Fox RJ, Bar-Or A, Traboulsee A, et al. Tolebrutinib in nonrelapsing secondary progressive multiple sclerosis. N Engl J Med. 2025;392(19):1883-1892. doi:10.1056/NEJMoa2415988


