Pegloticase Achieves High Urate-Lowering Efficacy and Quality-of-Life Gains in Kidney Transplant Recipients With Uncontrolled Gout
Key Highlights
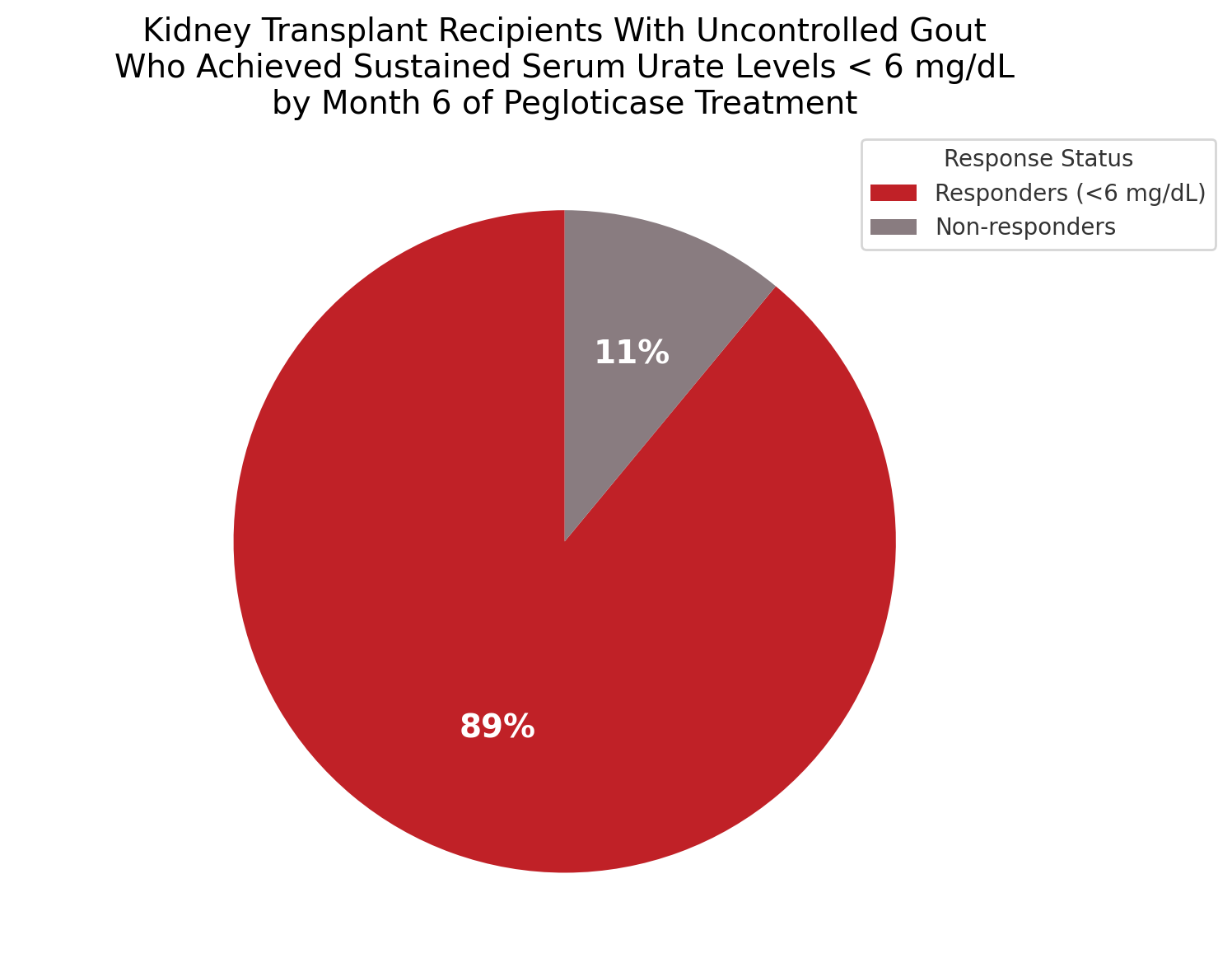
- 89% of kidney transplant (KT) recipients with uncontrolled gout achieved sustained serum urate (SU) levels < 6 mg/dL by month 6 of pegloticase treatment.
- Clinically meaningful improvements in patient-reported quality-of-life (QOL) measures were observed, including reductions in pain and disability.
- Blood pressure and albuminuria improved or remained stable during and after pegloticase therapy, suggesting potential renal and cardiovascular benefits.
In the PROTECT phase 4 clinical trial, pegloticase demonstrated high urate-lowering efficacy in KT recipients with uncontrolled gout. By month 6, 89% of participants achieved sustained serum urate levels < 6 mg/dL. The study also revealed substantial improvements in QOL metrics, including pain, physical function, and physician-assessed disease severity, alongside stabilization of renal function and decreases in blood pressure.
KT recipients have a 12 times higher risk for gout than the general population due to impaired uric acid clearance and the effects of calcineurin inhibitors. Management of gout in this group is complicated by limited efficacy and safety concerns associated with oral urate-lowering therapies. Pegloticase, a recombinant PEGylated uricase administered intravenously, has shown promise in patients with advanced kidney disease but had not been specifically studied in transplant recipients until now.
This open-label, multisite study enrolled 20 adult KT recipients ≥ 1-year post-transplant, all of whom had serum urate ≥ 7 mg/dL and failed or were intolerant to conventional oral therapy. Patients received biweekly pegloticase infusions for 24 weeks, alongside standard prophylaxis for gout flares and infusion reactions. QOL was assessed using the Health Assessment Questionnaire (HAQ) and Physician Global Assessment (PhGA), and clinical outcomes included tophus resolution and changes in renal parameters and blood pressure.
Of the 20 patients treated (85% male, mean age 53.9 years), 14 completed the full 24-week course. Sustained SU lowering was achieved in 16 of 18 evaluable patients (89%). Among patients with tophi at baseline, 43% experienced complete resolution of ≥ 1 tophus by Week 24. Clinically meaningful improvements in HAQ-Pain (-35.5), HAQ-DI (-0.3), HAQ-Health (-22.4), and PhGA (-2.4) scores were observed, with these benefits persisting during a 3-month follow-up. Renal function remained stable (mean eGFR change: +0.6 mL/min/1.73 m²), and blood pressure decreased significantly (mean arterial pressure: -6.8 mmHg). Albuminuria also showed signs of improvement in patients with severe baseline involvement.
“Given the high prevalence of uncontrolled gout in KT recipients, proper SU management is of particular importance,” the study authors concluded. “Additionally, intensive urate-lowering with pegloticase may have clinical and QOL benefits.”
Reference:
Abdellatif A, Zhao L, Obermeyer K, Vranic Z, Marder BA, Scandling JD. Quality of life improvements and clinical assessments in kidney transplant recipients undergoing pegloticase treatment for uncontrolled gout: findings of the phase 4 PROTECT clinical trial. Front Immunol. 2025;16:1516146. Published 2025 Mar 13. doi:10.3389/fimmu


