Oral Probiotics Fail to Prevent Recurrent Vulvovaginal Infections in Pregnant Women, Randomized Trial Finds
Key Highlights:
- Oral probiotics did not significantly reduce the recurrence of vulvovaginal infections in pregnant women after successful treatment.
- No differences were observed in the time to first infection, pregnancy outcomes, or vaginal colonization by probiotic strains.
- None of the specific probiotic strains used were detected in vaginal cultures despite 4 months of supplementation.
- A minor symptom benefit was noted: fewer women in the probiotic group reported pruritus at the end of the study.
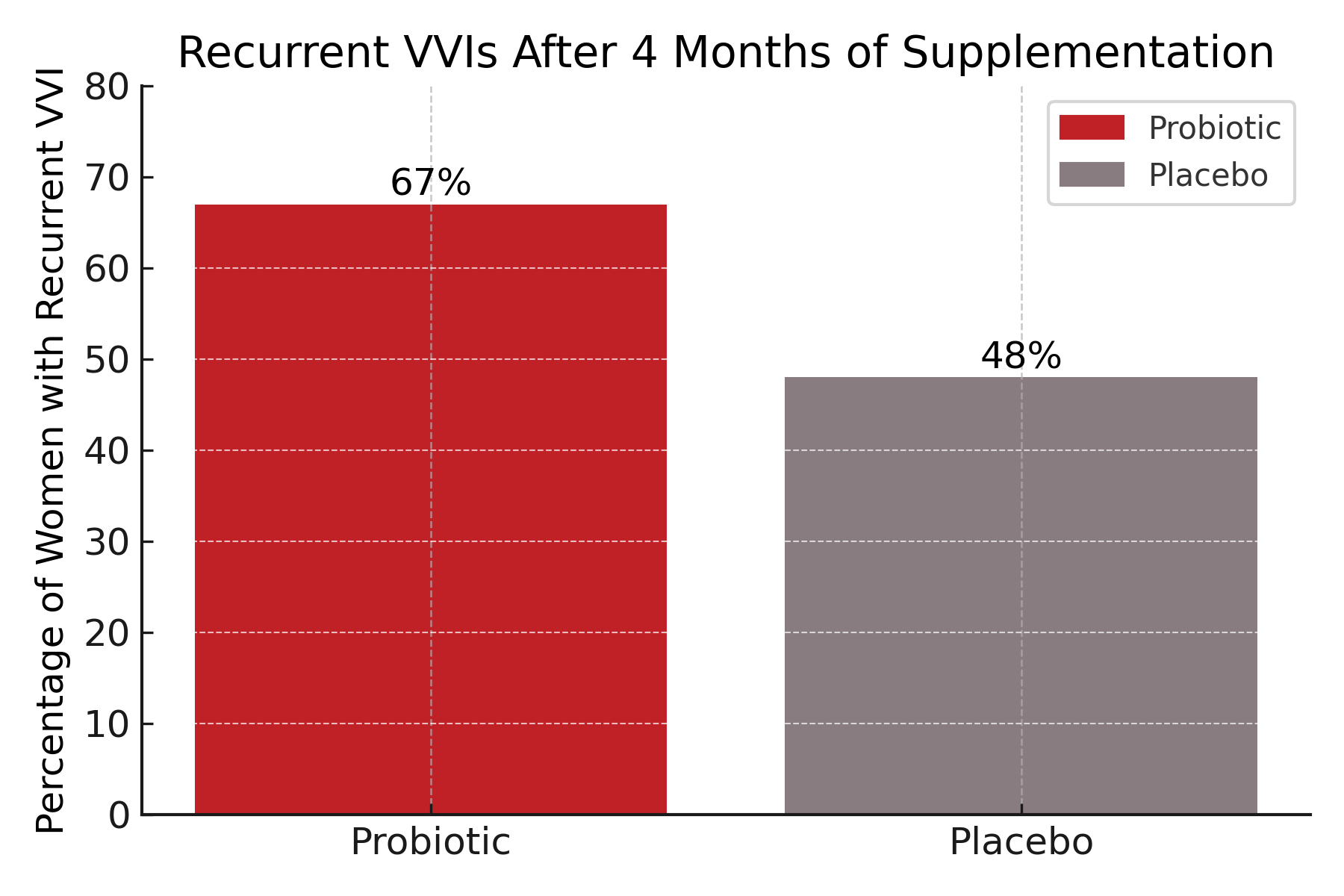
The results of a multicenter, double-blind, randomized, placebo-controlled trial showed that oral probiotics did not reduce the recurrence rate of vulvovaginal infections (VVIs) in pregnant women after initial eradication of infection. Over the course of approximately 4 months of supplementation, 67% of women in the probiotic group experienced a recurrent VVI, compared with 48% in the placebo group—a difference that was not statistically significant (P = .19). The probiotics also failed to impact vaginal colonization with the administered strains, and no significant changes were observed in maternal or neonatal outcomes.
Vulvovaginal infections, particularly bacterial vaginosis (BV), abnormal vaginal flora (AVF), and vulvovaginal candidiasis (VVC), are prevalent in pregnancy and associated with adverse outcomes such as preterm labor, chorioamnionitis, and neonatal thrush. While antibiotic and antifungal treatments are effective for initial eradication, they often fail to restore the normal lactobacilli-dominated microbiota, leading to frequent recurrence. Oral probiotic therapy has shown some promise in non-pregnant populations but had not been rigorously evaluated for secondary prevention during pregnancy.
In this study, 50 pregnant women with previously treated and eradicated VVIs were randomized to receive either oral probiotics (containing Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus, Lactobacillus paracasei, Lactobacillus rhamnosus, and Streptococcus thermophilus) or placebo, taken twice daily until delivery. Monthly vaginal smears were conducted to monitor recurrence, and adherence was confirmed by capsule count and participant interview. Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF-MS) was used to detect the presence of probiotic strains in vaginal cultures.
Results showed no statistically significant differences between groups in VVI recurrence, time to first infection, or vaginal colonization. In both groups, Lactobacillus gasseri remained the predominant vaginal species throughout the study. No colonization by the specific probiotic strains was detected in any participant. Pregnancy outcomes—including delivery mode, preterm birth, neonatal sepsis, and neonatal intensive care unit admissions—were similar between groups. Symptomatically, the only significant difference was a lower reported rate of pruritus in the probiotic group at the study’s conclusion (13% vs 39%, P = .03).
Limitations of this study include its small sample size (only 74% of the target enrollment was reached), limited power to assess pregnancy outcomes or to analyze AVF/BV and VVC separately, and the lack of stool analysis to confirm systemic probiotic colonization. Additionally, the findings are specific to the probiotic formulation used and cannot be generalized to other strains or formulations.
“The oral probiotic product tested in this study did not reduce the recurrence rate of VVIs in pregnant women following eradication,” the study authors concluded.
Reference:
Nachum Z, Suleiman A, Colodner R, et al. Oral probiotics to prevent recurrent vulvovaginal infections during pregnancy-multicenter double-blind, randomized, placebo-controlled trial. Nutrients. 2025;17(3):460. Published 2025 Jan 27. doi:10.3390/nu17030460


