NeuroSAFE Technique Enhances Erectile Function and Early Continence Recovery After Robot-Assisted Prostatectomy, Randomized Trial Shows
Key Highlights
- NeuroSAFE-guided robot-assisted radical prostatectomy (RARP) significantly improved erectile function at 12 months compared with standard RARP.
- Early urinary continence recovery at 3 months was better in the NeuroSAFE group.
- NeuroSAFE enabled higher rates of bilateral nerve-sparing (82% vs 56%) without compromising short-term oncologic safety.
In a phase 3 randomized controlled trial, use of the NeuroSAFE technique during robot-assisted radical prostatectomy (RARP) led to significantly improved functional outcomes. Patients who underwent NeuroSAFE-guided RARP reported higher erectile function scores at 12 months postoperatively and experienced a more rapid recovery of urinary continence at 3 months, compared with those receiving standard RARP. These improvements were most pronounced in patients who were not initially recommended for bilateral nerve-sparing.
Erectile dysfunction and urinary incontinence are common and distressing sequelae of RARP, primarily resulting from damage to the neurovascular bundles. Despite the rise of nerve-sparing strategies and robotic approaches, accurate intraoperative assessment of tumor margins near these critical structures remains challenging. NeuroSAFE, a frozen section technique allowing real-time evaluation of prostate margins adjacent to the neurovascular bundles, offers a potential solution. Prior to this trial, however, evidence supporting its efficacy was limited to retrospective and non-randomized studies, creating a need for high-quality data.
The NeuroSAFE PROOF study was conducted across five NHS hospitals in the UK, enrolling 407 patients with non-metastatic prostate cancer and good baseline erectile function. Participants were randomized 1:1 to receive either standard RARP or NeuroSAFE-guided RARP, with masking maintained for patients throughout. In the NeuroSAFE arm, intraoperative margin assessment guided nerve-sparing decisions, with secondary resections performed when adverse margin features were detected. The primary endpoint was IIEF-5 erectile function score at 12 months; secondary outcomes included ICIQ continence scores at 3 and 6 months and IIEF-6 scores at 12 months.
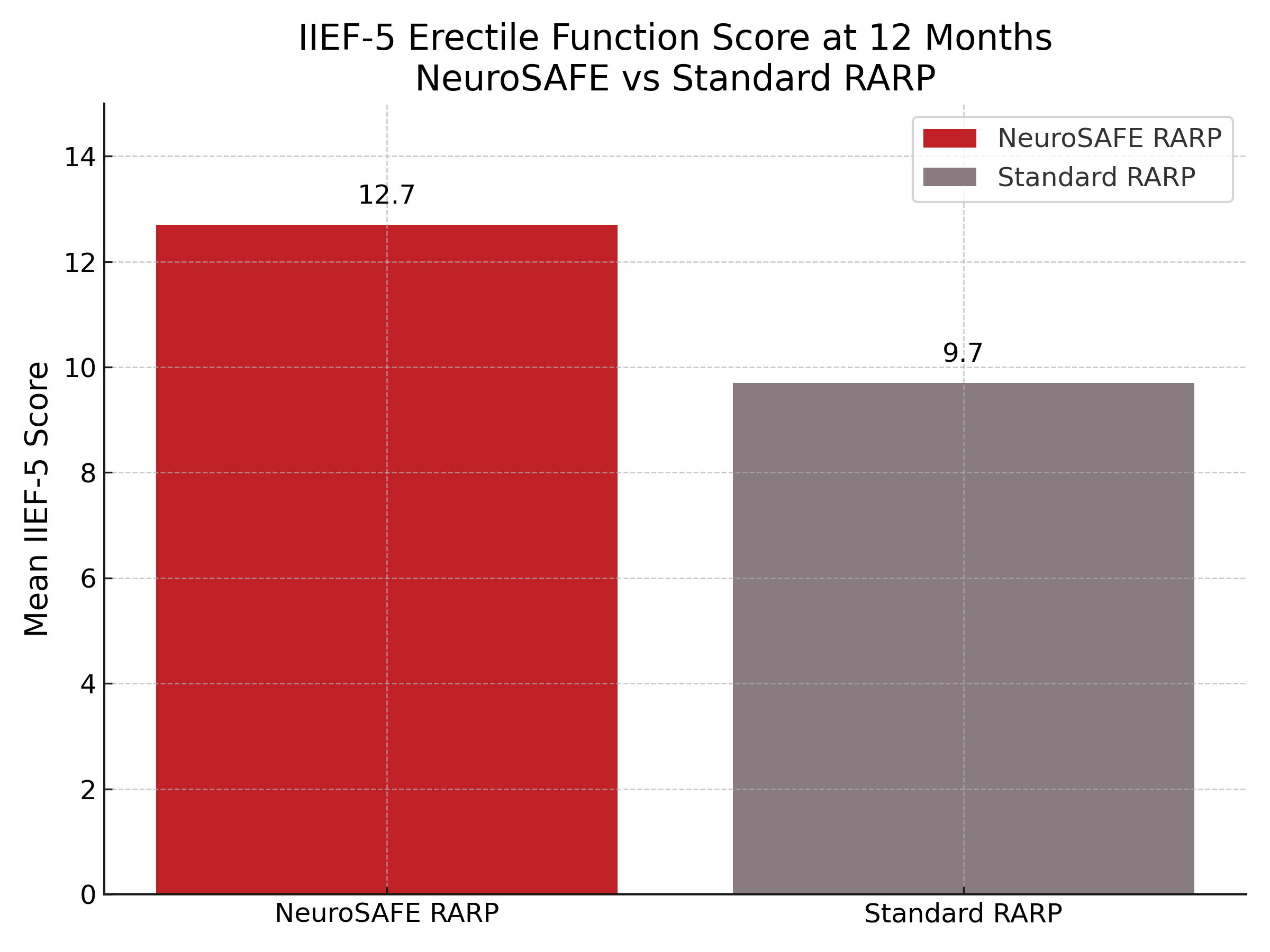
Of the 381 patients who underwent surgery, 344 had evaluable IIEF-5 scores at 12 months. The NeuroSAFE group had a significantly higher mean IIEF-5 score (12.7 vs 9.7; adjusted mean difference 3.18; 95% CI 1.62–4.75; P < .0001) and a higher proportion of patients with mild or no erectile dysfunction at 12 months (39% vs 23%). Similarly, IIEF-6 scores favored the NeuroSAFE group (15.3 vs 11.5; adjusted mean difference 3.92; P < .0001). Early continence outcomes also improved: at 3 months, ICIQ scores were significantly better in the NeuroSAFE group, though no difference was seen at 6 months. Bilateral nerve-sparing was achieved in 82% of NeuroSAFE patients compared to 56% in the standard group. Oncologic outcomes, including rates of biochemical recurrence and PSA persistence, were similar between groups at 12 months.
Limitations of the study include its limited duration of oncologic follow-up, concentration of procedures at a single high-volume center, and unblinded clinical teams for follow-up assessments. Additionally, the need for specialized pathology services and longer operative time may challenge widespread adoption in resource-constrained settings.
“The use of NeuroSAFE to guide nerve-sparing during RARP improves patient-reported IIEF-5 scores at 12 months and short-term urinary continence,” the study authors concluded. “The erectile function benefit is enhanced in patients who would not otherwise have undergone bilateral nerve-sparing by standard practice.”
Reference:
Dinneen E, Almeida-Magana R, Al-Hammouri T, et al. Effect of NeuroSAFE-guided RARP versus standard RARP on erectile function and urinary continence in patients with localised prostate cancer (NeuroSAFE PROOF): a multicentre, patient-blinded, randomised, controlled phase 3 trial. Lancet Oncol. 2025;26(4):447-458. doi:10.1016/S1470-2045(25)00091-9


