Biologic Switching in Psoriasis Improves Outcomes, Raises Infection Risk
Key Highlights
- Interclass biologic switching in psoriasis demonstrated faster short-term and more stable long-term effectiveness.
- Switching from anti–TNF-α or anti–IL-12/23p40 agents to anti–IL-23p19 agents demonstrated significantly improved outcomes.
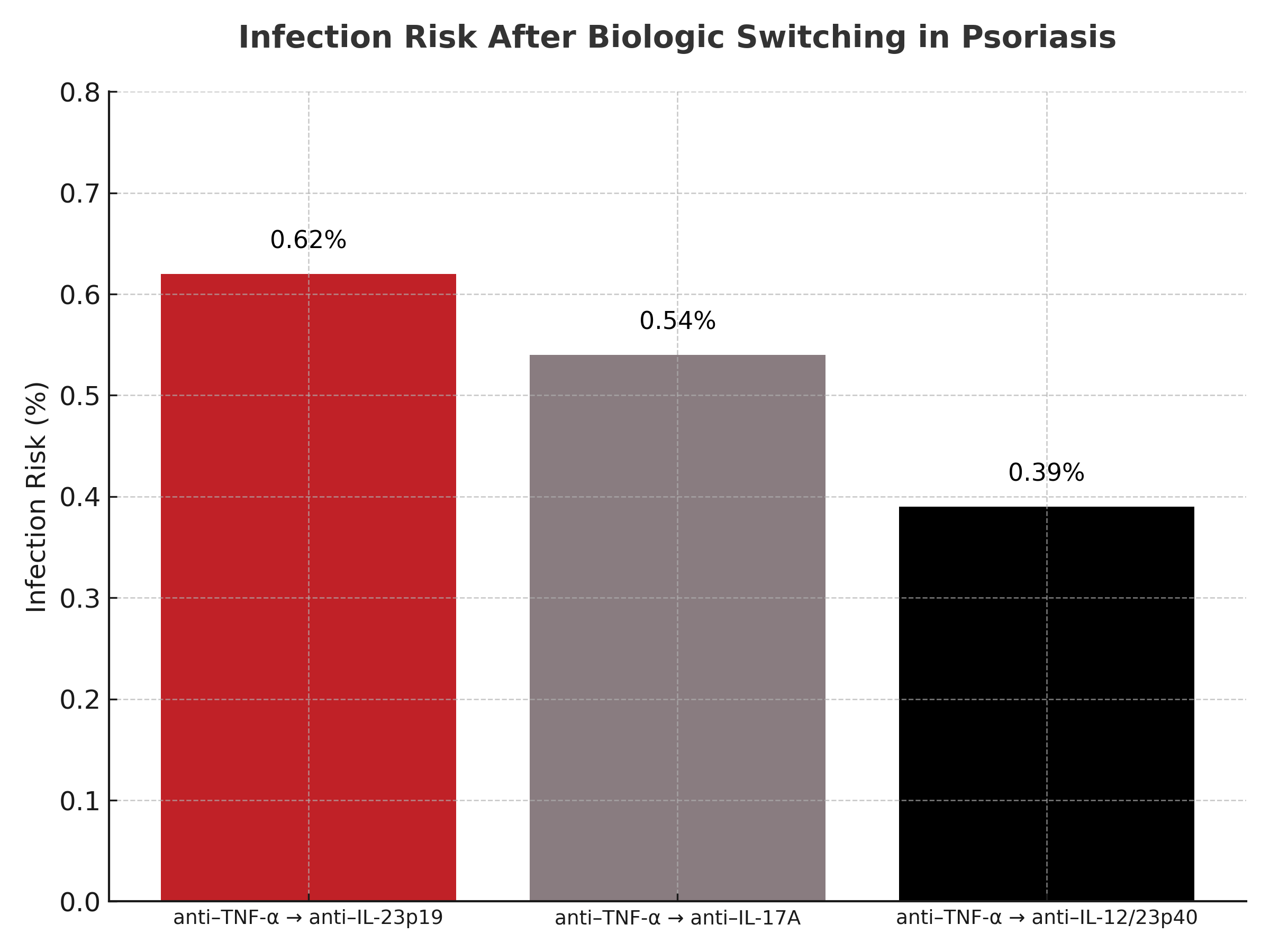
- Increased risk of infection was associated with switches, particularly from anti–TNF-α to anti–IL-23p19, anti–IL-17A, and anti–IL-12/23p40 agents.
- No significant differences were observed in serious or treatment-related adverse events overall.
This systematic review and meta-analysis found that interclass switching of biologic agents in psoriasis treatment significantly improved disease outcomes, with faster short-term response and more sustained long-term control. However, the analysis also identified a higher risk of infection when switching between certain classes, underscoring the need for careful clinical consideration.
Psoriasis management often requires therapy adjustment due to cost, primary or secondary treatment failure, or adverse events with the initial biologic. Despite the increasing use of newer biologics with distinct mechanisms of action, evidence regarding the safety and effectiveness of switching across biologic classes has been limited. This study aimed to address that evidence gap by systematically evaluating randomized clinical trials involving interclass switching.
Researchers searched PubMed, Embase, and the Cochrane Library from inception to January 25, 2025, identifying 24 randomized clinical trials involving 12,661 patients with plaque psoriasis. Eligible studies included patients aged 18 years or older undergoing crossover or switching from one biologic agent to another, either within the same class or across different classes. Data were extracted independently by two researchers and analyzed using random-effects models in accordance with PRISMA guidelines. The primary endpoint was achievement of PASI 90, with additional assessments including PASI 75, PASI 100, clinician- and patient-reported outcomes, and safety endpoints such as adverse events (AEs), serious AEs, and infections.
Results showed substantial improvements in PASI responses after switching. Odds ratios for PASI 90, PASI 75, and PASI 100 were 28.61, 11.11, and 18.76, respectively, indicating strong effectiveness after transition to a new agent. The most favorable outcomes were observed when patients switched from anti–TNF-α or anti–IL-12/23p40 agents to anti–IL-23p19 agents. Safety outcomes did not differ significantly in terms of serious or treatment-related AEs; however, infection risk was higher for switches from anti–TNF-α to anti–IL-23p19 (0.62%), anti–IL-17A (0.54%), and anti–IL-12/23p40 (0.39%).
“With the introduction of biologics with diverse mechanisms of action, these options may become viable and potentially superior,” Zhang et al concluded. “However, further confirmatory data from larger patient cohorts and longer follow-up periods are required to obtain more conclusive results.”
Reference:
Zhang M, Hong S, Wang Q, et al. Biopharmaceutical switching in psoriasis treatment: a systematic review and meta-analysis. JAMA Dermatol. 2025:e252714. doi:10.1001/jamadermatol.2025.2714


