Vaginal Fluid DNA Methylation Test Shows High Accuracy for Detecting Endometrial Cancer in Women with Abnormal Uterine Bleeding
A prospective study conducted by researchers at the Mayo Clinic and Exact Sciences has shown promising results for a non-invasive method to detect endometrial cancer (EC) in patients presenting with abnormal uterine bleeding (AUB).
The study, presented at the Society of Gynecologic Oncology 2025 Annual Meeting on Women’s Cancer in Seattle, WA, evaluated the performance of a panel of six methylated DNA markers (MDMs) detectable in vaginal fluid collected via tampon. The findings indicate that this test could serve as a high-sensitivity screening tool to identify patients at elevated risk for EC, potentially reducing the number of unnecessary endometrial biopsies currently recommended by clinical guidelines.
Abnormal uterine bleeding affects a significant number of women each year, including 1.4 million women aged 18 to 50 in the United States, as well as 4% to 11% of postmenopausal women. Although only 5% to 10% of AUB cases are caused by underlying endometrial cancer or precancerous conditions, current American College of Obstetricians and Gynecologists (ACOG) guidelines recommend endometrial sampling in all patients aged 45 years and older, or younger patients with additional risk factors. Given the invasiveness and discomfort associated with endometrial biopsy, the investigators sought to develop a less invasive method to identify patients who would benefit most from further diagnostic evaluation.
In the study, participants self-collected vaginal fluid using a tampon while under clinical supervision. Participants included two cohorts: women aged 45 and older with AUB or women aged 18 and older with Lynch syndrome, and women aged 18 and older with biopsy-confirmed endometrial cancer or atypical hyperplasia (AH). Exclusion criteria included current pregnancy, prior pelvic radiation, recent or current cancers, cervical dysplasia, or prior hysterectomy. The collected fluid was processed to extract DNA, which was then bisulfite-converted and analyzed using long-probe quantitative amplified signal (LQAS) assays. A total of 19 candidate MDMs and four reference genes were initially tested, and data were analyzed using random forest modeling to identify the most predictive markers.
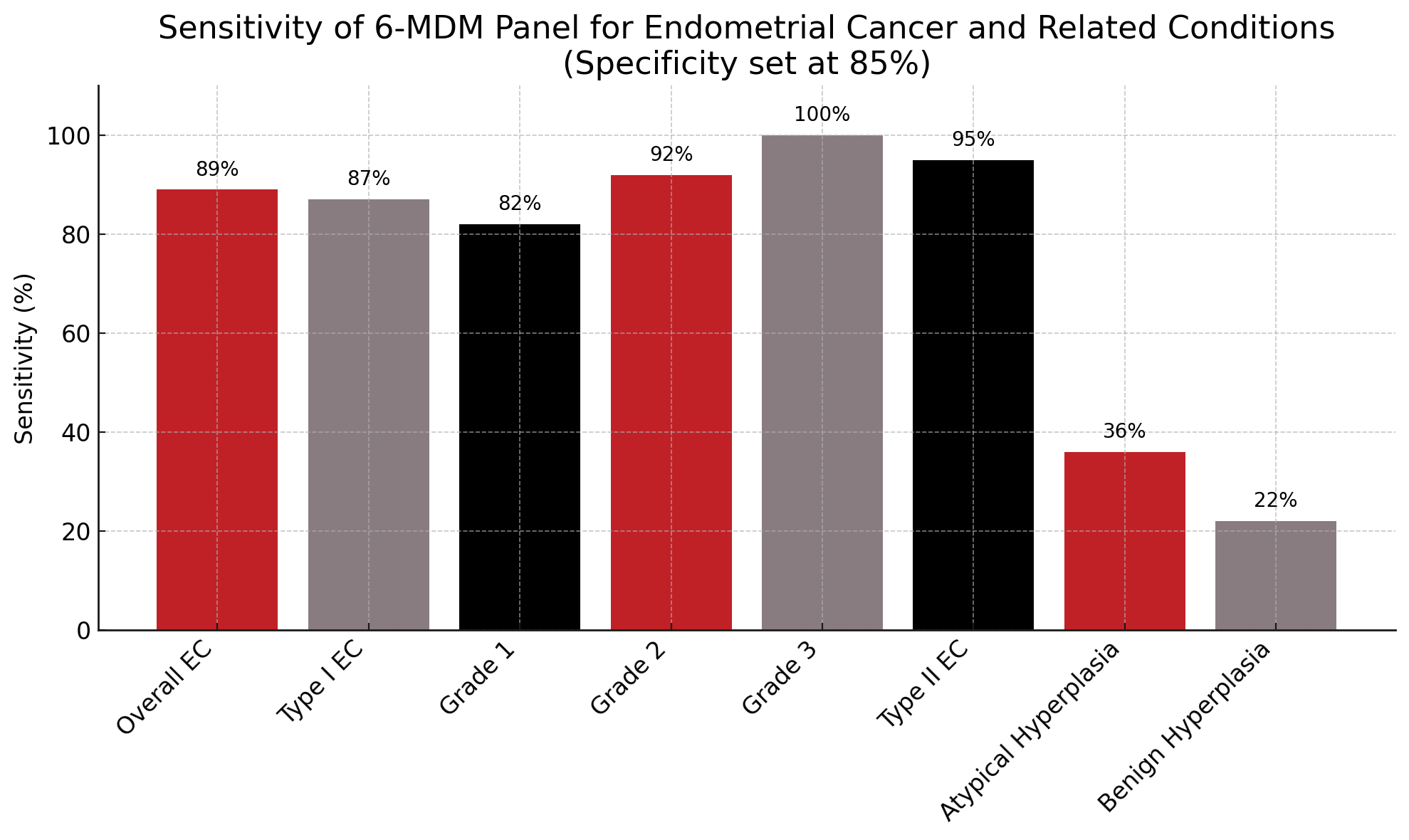
A total of 694 participants met the inclusion criteria, including 256 patients with EC, 374 benign AUB controls, 37 with AH, and 27 with benign hyperplasia. The average age of participants with EC was 63 years, compared with 59 years among the benign group. Postmenopausal status was reported in 86% of EC patients and 65% of controls. Among the EC cases, 46% had grade 1 endometrioid histology, 19% had grade 2, and 11% had grade 3. Type II histologies were also represented, including serous (8%), carcinosarcoma (5%), clear cell (4%), and mixed histologies (7%).
The final 6-MDM panel yielded an area under the curve of 0.93 (95% CI, 0.91-0.96) for overall EC. At a fixed specificity of 85% (95% CI, 81%-89%), the panel demonstrated a sensitivity of 89% (84%-93%) for detecting any EC. Sensitivity for Type II EC was 95% (85%-99%), while for Type I EC it was 87% (81%-91%), with grade-specific sensitivities of 82% for grade 1, 92% for grade 2, and 100% for grade 3. For atypical hyperplasia, sensitivity was 36% (21%-54%), and for benign hyperplasia without atypia, sensitivity was 22% (9%-43%).
The study also found that the test detected EC in patients who had not undergone biopsy at the time of vaginal fluid collection, as well as in some patients with atypical hyperplasia who were later found to have EC at hysterectomy. Twenty-four percent of EC cases were biopsy-naïve at the time of sampling, and 41% of patients with AH on pre-hysterectomy biopsy were ultimately diagnosed with EC on surgical pathology. These findings suggest that the test may detect underlying EC even before histologic confirmation.
While the study was limited by its relatively homogeneous population and small numbers of certain rare histologies, it demonstrated strong methodological rigor, including pathology-confirmed diagnoses and blinded assay processing.
“Subsequent studies are underway to further develop and validate a vaginal fluid-based test for EC that is generalizable to all elevated risk people with AUB,” the study authors concluded.
Reference:
Bakkum-Gamez J, Kisiel J, Slettedahl S, et al. Development of an endometrial cancer test for evaluating abnormal uterine bleeding. Presented at: Society of Gynecologic Oncology Annual Meeting; March 14-17, 2025; Seattle, WA.


