Optimizing Seizure Management in Treatment-Resistant Epilepsy: A Patient Case Walk-Through With David Burdette, MD
This hosted case study illustrates optimized dosing of EPIDIOLEX® (cannabidiol) including co-administration with clobazam.
Introduction:
A panel of epilepsy experts came together to form consensus recommendations for optimizing the use of EPIDIOLEX for treatment of seizures associated with Lennox-Gastaut syndrome (LGS), Dravet syndrome (DS), and tuberous sclerosis complex (TSC).1 In this article, we’ll see those insights in action via a case of an adult patient from one of those experts, David Burdette, MD.
EPIDIOLEX is indicated for the treatment of seizures associated with LGS, Dravet syndrome, or tuberous sclerosis complex in patients 1 year of age and older.2 Contraindications are hypersensitivity to cannabidiol or any of the ingredients in EPIDIOLEX.2
Please see the full Important Safety Information below.
The Consensus Recommendations publication was sponsored by Jazz Pharmaceuticals, Inc.
Click here to access the Consensus Recommendations publication
David Burdette, MD, is board certified in neurology, epilepsy, and clinical neurophysiology. He regularly treats patients with treatment-resistant epilepsy, such as LGS.
In this article, Dr Burdette discusses a patient case from his practice. He walks through the patient’s symptoms and treatment history before sharing insights on how this patient has responded to an optimized treatment regimen with EPIDIOLEX.
Introducing the patient
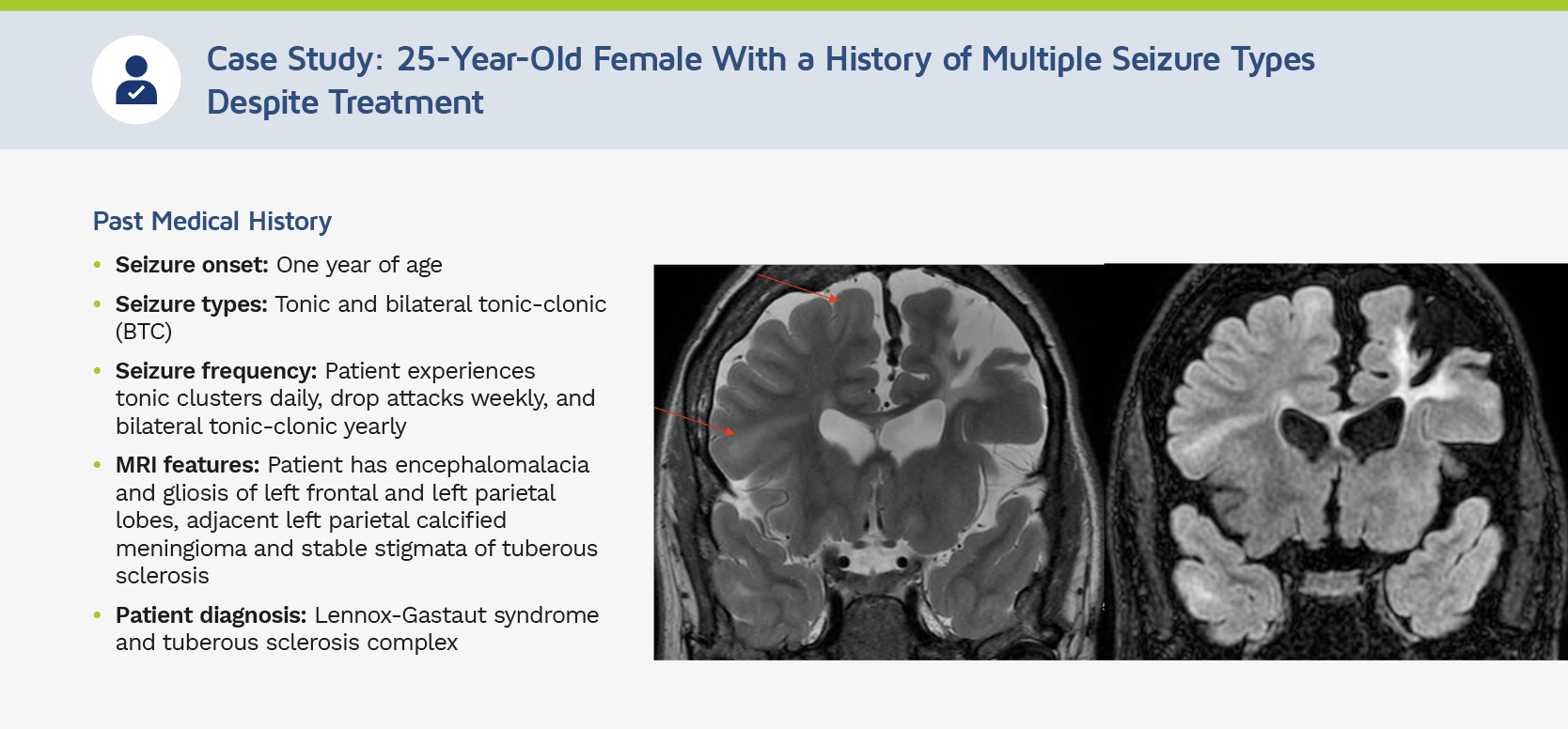
“This patient had a presentation of epileptic encephalopathy and experienced prolonged seizure clusters. With her having some degree of cognitive impairment, multiple seizure types, and prior history of slow spike wave discharges, she fell along the continuum of LGS.”
Patient's treatment history

“She has been tried on a number of anti-seizure medications (ASMs) dating back to 2 years of age. She was still experiencing ongoing seizures and had inadequate seizure control. With her diagnosis of LGS, she was an appropriate candidate to start treatment with EPIDIOLEX.”
The decision to add EPIDIOLEX
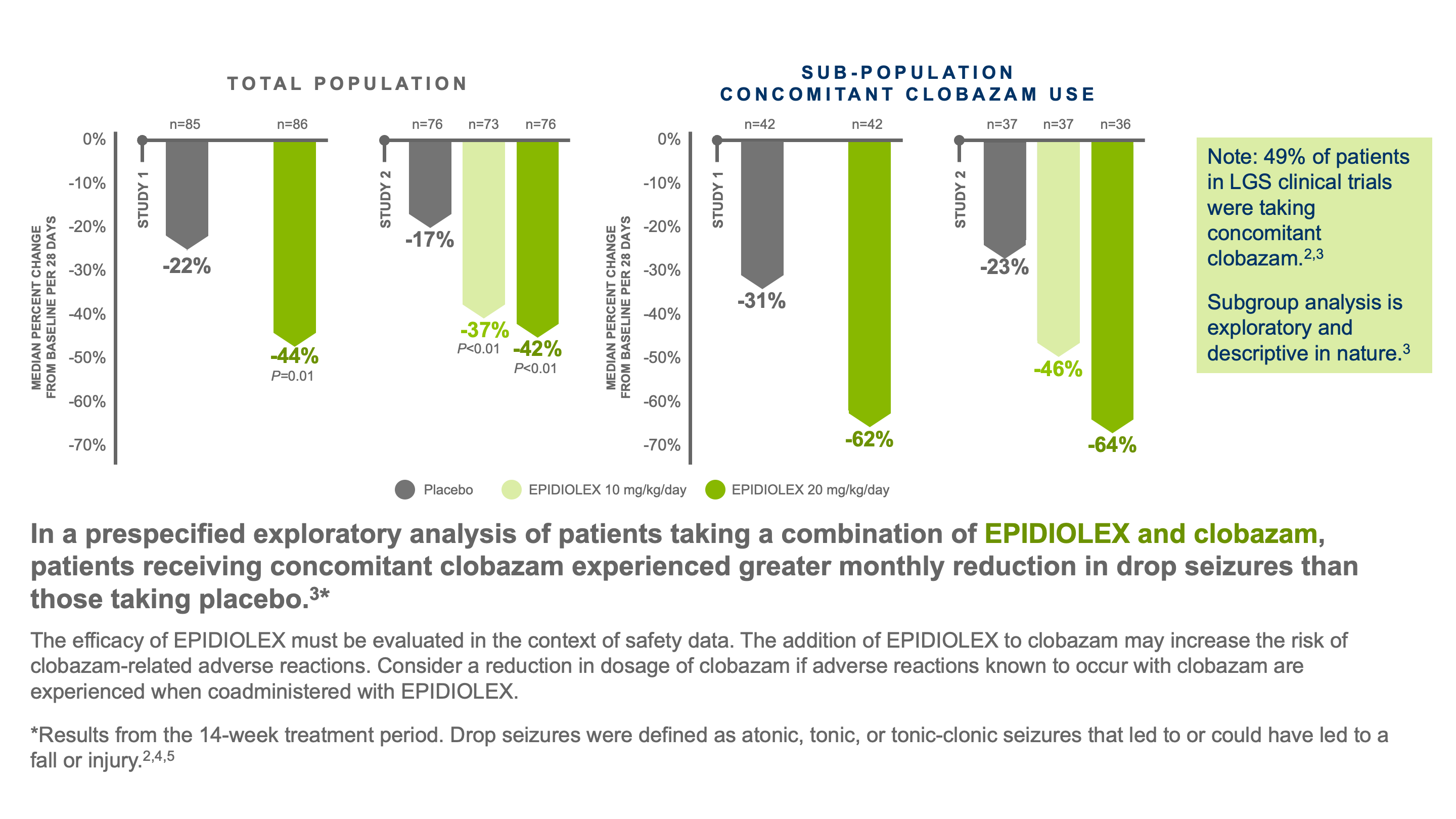
In LGS clinical trials, EPIDIOLEX significantly reduced the frequency of drop and total seizures. The median percent reduction in monthly frequency of drop seizures was measured across 2 trials.2
Two randomized, double-blind, placebo-controlled trials compared doses of EPIDIOLEX (Study 1: 20 mg/kg/day; Study 2: 10 mg/kg/day and 20 mg/kg/day) vs placebo in patients diagnosed with LGS and aged 2 to 55 years2:

Titrating EPIDIOLEX
"If we are going to make a dent in seizure frequency for patients with LGS, we are running a marathon, not a sprint. I start the patient on a lower EPIDIOLEX dose and titrate them to a reasonable target and see how the patient responds before titrating further.”

Consensus Panel Recommendations:
- To address tolerability, start with a low dose and titrate up slowly to the maximum recommended maintenance dosage (20 mg/kg/day) for patients living with LGS1
- An adequate trial of EPIDIOLEX should involve, if tolerated, dosing to the minimum recommended dose of 10 mg/kg/day1
- Patients should be encouraged not to discontinue EPIDIOLEX due to lack of efficacy if they have not been given a sufficient trial at the maximum tolerated dose1
Common adverse reactions with EPIDIOLEX include transaminase elevations, somnolence, decreased appetite, and diarrhea.2 Ongoing liver monitoring is recommended as EPIDIOLEX can cause dose-related elevations of liver transaminases.2 Continue reading for additional Important Safety Information.
EPIDIOLEX is commonly used concomitantly with other antiseizure medications.1 One medication that Dr Burdette’s patient was taking at this time was clobazam.
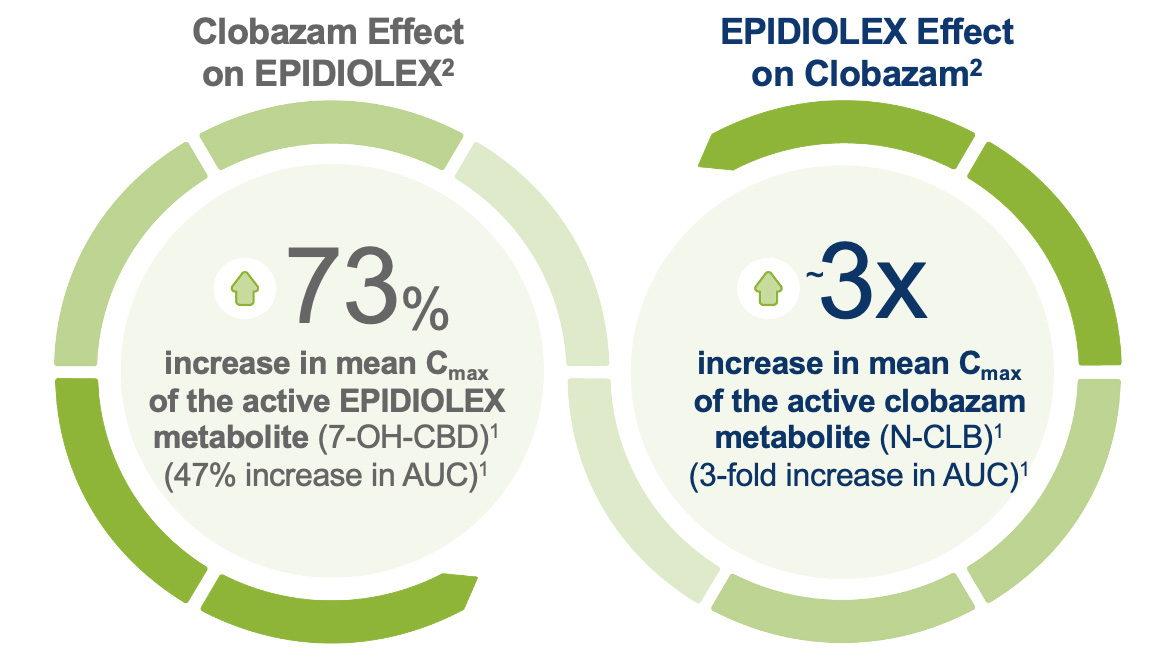
There are some important considerations for patients taking EPIDIOLEX and clobazam. When taken concomitantly with clobazam, EPIDIOLEX may cause somnolence and sedation, typically early in treatment, and this may lessen over time. Pneumonia was also reported in clinical trials in some patients taking EPIDIOLEX and clobazam. Elevated ammonia levels have been reported in some patients with transaminase elevations. Patients should be monitored until they have gained sufficient experience with EPIDIOLEX.2
“I did not down titrate or reduce the patient’s dose of clobazam after starting EPIDIOLEX. Along with the patient's caregiver, we monitored for somnolence and sedation. Following this I made stepwise reductions in the clobazam, reducing the dosage to 5 mg daily.
"This approach to managing EPIDIOLEX and clobazam is aligned to the recommendations discussed in the Consensus Panel Recommendations, published in the journal, Epilepsia Open.”1
How is the patient doing now?
As of November 2025, the patient is currently receiving EPIDIOLEX at a dosage of 20 mg/kg/day, alongside clobazam dosed at 5 mg/day. Her drop seizure severity has been reduced, and she has experienced significant reduction in overall seizure frequency. She continues to be monitored for gait ataxia and somnolence. Her family has shared that she displays less irritable mood since her seizures have reduced.
“While the patient continues to have some ongoing seizures, they are no longer prolonged clusters. We have made significant improvements to a more manageable seizure frequency, with subsequent improvement in her mood.”
WARNINGS & PRECAUTIONS
Hepatic Injury:
EPIDIOLEX can cause dose-related transaminase elevations. Concomitant use of valproate and elevated transaminase levels at baseline increase this risk. Obtain transaminase and bilirubin levels prior to starting treatment, at 1, 3, and 6 months after initiation of treatment, and periodically thereafter, or as clinically indicated. Resolution of transaminase elevations occurred with discontinuation of EPIDIOLEX, reduction of EPIDIOLEX and/or concomitant valproate, or without dose reduction. For patients with elevated transaminase levels, consider dose reduction or discontinuation of EPIDIOLEX or concomitant medications known to affect the liver (e.g., valproate or clobazam). Dose adjustment and slower dose titration is recommended in patients with moderate or severe hepatic impairment. Consider not initiating EPIDIOLEX in patients with evidence of significant liver injury. There have been postmarketing reports of cholestatic or mixed patterns of liver injury. Elevated ammonia levels were reported in some patients with transaminase elevations; most taking concomitant valproate, clobazam, or both. Consider discontinuation or dose adjustment of valproate or clobazam if ammonia is elevated.
Somnolence and Sedation:
EPIDIOLEX can cause somnolence and sedation that generally occurs early in treatment and may diminish over time; these effects occur more commonly in patients using clobazam and may be potentiated by other CNS depressants.
Suicidal Behavior and Ideation:
Antiepileptic drugs (AEDs), including EPIDIOLEX, increase the risk of suicidal thoughts or behavior. Inform patients, caregivers, and families of the risk and advise them to monitor and report any signs of depression, suicidal thoughts or behavior, or unusual changes in mood or behavior. If these symptoms occur, consider if they are related to the AED or the underlying illness.
Withdrawal of Antiepileptic Drugs:
As with most AEDs, EPIDIOLEX should generally be withdrawn gradually because of the risk of increased seizure frequency and status epilepticus.
ADVERSE REACTIONS:
The most common adverse reactions in patients receiving EPIDIOLEX (≥10% and greater than placebo) include transaminase elevations; somnolence; decreased appetite; diarrhea; pyrexia; vomiting; fatigue, malaise, and asthenia; rash; insomnia, sleep disorder and poor-quality sleep; and infections. Hematologic abnormalities were also observed.
PREGNANCY:
EPIDIOLEX should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Encourage women who are taking EPIDIOLEX during pregnancy to enroll in the EPIDIOLEX Pregnancy Surveillance Program and the North American Antiepileptic Drug (NAAED) Pregnancy Registry.
DRUG INTERACTIONS:
Strong inducers of CYP3A4 and CYP2C19 may affect EPIDIOLEX exposure. EPIDIOLEX may affect exposure to CYP2C19 substrates (e.g., clobazam, diazepam, stiripentol), orally administered P-gp substrates, or other substrates (see full Prescribing Information). Consider dose reduction of orally administered everolimus, with appropriate therapeutic drug monitoring, when everolimus is combined with EPIDIOLEX. A lower starting dose of everolimus is recommended when added to EPIDIOLEX therapy. Concomitant use of EPIDIOLEX and valproate increases the incidence of liver enzyme elevations. Pneumonia was observed more frequently with concomitant use of EPIDIOLEX and clobazam. Dosage adjustment of EPIDIOLEX or other concomitant medications may be necessary.
INDICATIONS:
EPIDIOLEX (cannabidiol) oral solution is indicated for the treatment of seizures associated with Lennox-Gastaut syndrome (LGS), Dravet syndrome (DS), or tuberous sclerosis complex (TSC) in patients 1 year of age and older.
Please read the EPIDIOLEX full Prescribing Information for additional important information.
©2025 Jazz Pharmaceuticals plc or its subsidiaries.
All rights reserved. US-EPX-2500256
References
- Wechsler RT, Burdette DE, Gidal BE, et al. Consensus panel recommendations for the optimization of EPIDIOLEX® treatment for seizures associated with Lennox-Gastaut syndrome, Dravet syndrome, and tuberous sclerosis complex. Epilepsia Open. 2024;9(5):1632-1642.
- EPIDIOLEX [package insert]. Palo Alto, CA: Jazz Pharmaceuticals, Inc.
- Gunning B, Mazurkiewicz-Bełdzińska M, Chin RFM, et al. Cannabidiol in conjunction with clobazam: analysis of four randomized controlled trials. Acta Neurol Scand. 2021;143(2):154-163.
- Thiele EA, Marsh ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
- Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut Syndrome. N Engl J Med. 2018;378(20):1888-1897.


