Peer Reviewed
Gastric Varices Originating From an Aberrant Spleno-Gastro-Renal Venous Circulation
Authors:
Vinh-Quang Do Nguyen, DO
Corpus Christi Medical Center – Bay Area, Corpus Christi, Texas
Raghujit Singh, MD
Abdominal Specialists of South Texas, Corpus Christi, Texas
Citation:
Singh R, Nguyen VQD. Gastric varices originating from an aberrant spleno-gastro-renal venous circulation. Consultant. 2017;57(10):614-615.
A 72-year-old woman with a history of hypertension, type 2 diabetes mellitus, degenerative joint disease, and gastroesophageal reflux disease presented with massive hematemesis. She reported having heartburn, abdominal pain, nausea, and melena. She denied chest pain, dyspnea, alcohol use, a history of liver disease, and a history of gastrointestinal (GI) tract bleeding.
She reported using aspirin powder intermittently for her joint disease, but she denied using other nonsteroidal anti-inflammatory drugs.
Physical examination. On physical examination, the patient’s vital signs were significant for hypotension (92/51 mm Hg) and tachycardia (100 beats/min). The patient was obese, and she was alert, awake, and oriented. She had dry mucosal membranes, a mildly tender abdomen, and pale skin. The rest of the physical examination findings were unremarkable.
Diagnostic tests. Laboratory test results were significant for a white blood cell count of 15,100/µL (reference range, 4300-10,800/µL), a hemoglobin level of 8.6 g/dL (reference range, 14-18 g/dL); a hematocrit of 26.3% (reference range, 42%-52%); a blood urea nitrogen level of 49 mg/dL (reference range, 6-20 mg/dL), and a creatinine level of 1.6 mg/dL (reference range, 0.6-1.0 mg/dL).
Computed tomography (CT) scans of the abdomen and pelvis revealed varices near the gastric fundus and along the medial portion of the greater curvature of the stomach; the varices appeared to originate from the splenic hilum with some draining into the left renal vein.
CT also showed a normal pancreas, no morphologic features of cirrhosis, no esophageal varices, no splenomegaly, and no splenic vein thrombosis (SVT).
Treatment. The patient underwent an emergent esophagogastroduodenoscopy (EGD), the results of which revealed active intermittent bleeding from moderate-sized gastric varices (GVs) in the fundus (Figure 1). The GVs could not be treated endoscopically. Emergent interventional radiology evaluation was advised with transjugular intrahepatic portosystemic shunt (TIPS) placement. The patient underwent hepatic venography and pressure measurements, which revealed no portal hypertension. Thus, TIPS was not indicated owing to the absence of portal hypertension, and proceeding with splenic angiography was advised.
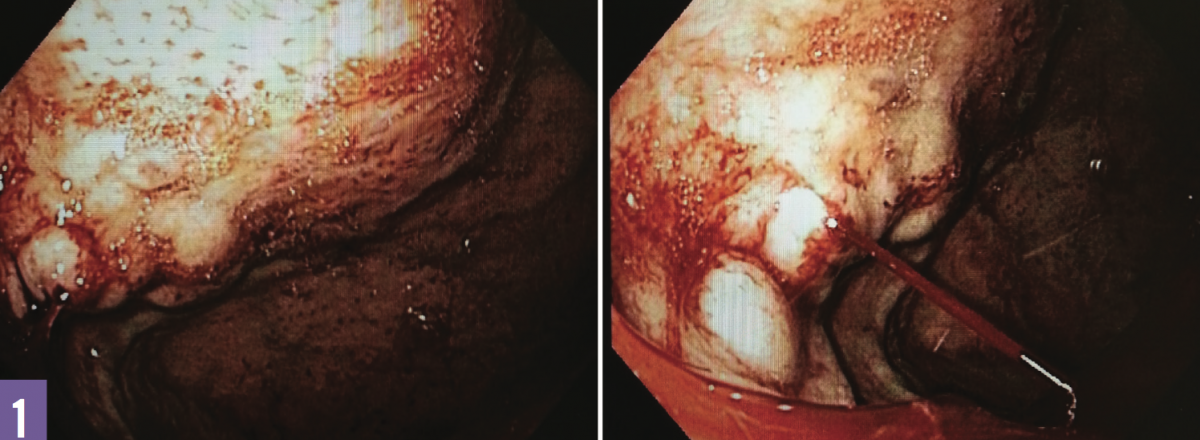
Figure 1: Gastric varices in the fundus (left); an actively bleeding varix (right).
The splenic angiogram showed filling of the splenic vein, which was patent, but branches from the splenic hilum were shown to be feeding the gastric varices, with the outflow leading toward the left renal vein, with some narrowing toward the renal vein entry (Figures 2 and 3). The patient underwent Blakemore tube placement and intubation and was started on pantoprazole drip, octreotide drip, and intravenous normal saline. She eventually underwent splenic artery embolization, which stopped the gastric variceal hemorrhage.

Figures 2 and 3: Angiography revealed an aberrant spleno-gastro-renal venous circulation (Figure 2). A schematic diagram of the aberrant sple- no-gastro-renal venous circulation (Figure 3). Abbreviations: PV, portal vein; SV, splenic vein; SMV, superior mesenteric vein; IVC, inferior vena cava; LRV, left renal vein; LGV, left gastric vein; RGV, right gastric vein.
Outcome, discussion
Discussion. GVs are dilated submucosal veins, most commonly detected in the proximal stomach. They are associated with portal hypertension and SVT. Gastroesophageal varices are present in approximately 50% of patients with cirrhosis, and GVs are less prevalent, occurring in 5% to 33% of these patients.1 GVs are the most common cause of upper GI tract bleeding in patients with portal hypertension after esophageal varices. They are more severe and are associated with a worse outcome than esophageal varices.2 In the United States, the majority of GV patients have underlying portal hypertension or SVT. GVs arising due to SVT are much less common than GVs due to portal hypertension.2 Fundal GVs usually arise from spleno-renal or gastro-renal shunts due to left-sided portal hypertension.3 The primary pathology of left-sided portal hypertension arises in the pancreas, and common etiologies include pancreatitis and pancreatic neoplasms.4,5
In our patient’s case, there was an aberrant spleno-gastro-renal venous circulation. There was no evidence of cirrhosis, esophageal varices, portal hypertension, pancreatitis, pancreatic neoplasm, SVT, splenic vein obstruction, or splenomegaly. The varices originated from the splenic hilum and drained into the left renal vein. The entry to the left renal vein was narrow, which led to increased blood flow through the spleno-gastro-renal venous circulation. The increased blood flow dilated the submucosal veins of the stomach, resulting in GVs.
According to the Sarin classification system, the GVs in our patient are isolated gastric varices (IGVs).2 IGVs are treated with cyanoacrylate glue after volume resuscitation and vasoactive drugs in the setting of an acute variceal hemorrhage. If hemorrhage is not controlled, the next steps in the management of IGVs include balloon tamponade, salvage TIPS, balloon-occluded retrograde transvenous obliteration, splenic embolization, and splenectomy.6
Outcome of the case. After splenic artery embolization had stopped the acute variceal hemorrhage, the patient was successfully extubated and had no further symptoms of hematemesis or melena. She received blood transfusions as needed, and her hemoglobin level stabilized.
She was placed on a regimen of appropriate prophylactic antibiotics and received the appropriate vaccinations. She ambulated well, tolerated her diet, and was discharged in a stable condition. She was referred for endoscopic cyanoacrylate glue injection.
REFERENCES:
- Garcia-Tsao G, Sanyal AJ, Grace N, Carey WD; Practice Guidelines Committee of the American Association for the Study of Liver Diseases; Practice Parameters Committee of the American College of Gastroenterology. Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology. 2007;46(3):922-938.
- Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence, classification and natural history of gastric varices: a long-term follow-up study in 568 portal hypertension patients. Hepatology. 1992;16(6):1343-1349.
- Al-Osaimi AMS, Caldwell SH. Medical and endoscopic management of gastric varices. Semin Intervent Radiol. 2011;28(3):273-282.
- Thompson RJ, Taylor MA, McKie LD, Diamond T. Sinistral portal hypertension. Ulster Med J. 2006;75(3):175-177.
- Sato T, Kitagawa S, Kimura M, Ohmura T, Karino Y, Toyota J. Gastric varices secondary to splenic vein occlusion due to pancreatic diseases. Pancreat Disord Ther. 2013;S3:001. doi:10.4172/2165-7092.S3-001
- Hwang JH, Shergill AK, Acosta RD, et al; ASGE Standards of Practice Committee. The role of endoscopy in the management of variceal hemorrhage. Gastrointest Endosc. 2014;80(2):221-227


