Authors:
John Riefler, MD, MS
PSI Pharma Support America Inc, King of Prussia, Pennsylvania
Maxim Kosov, MD, PhD
PSI Pharma Support America Inc, King of Prussia, Pennsylvania
Maxim Belotserkovskiy, MD, PhD, DMedSci
PSI CRO Deutschland GmbH, Planegg/Munich, Germany
Citation:
Riefler J, Kosov M, Belotserkovskiy M. Incidental ill-defined nodular lung opacities discovered during CT colonoscopy [published online January 23, 2019]. Pulmonology Consultant.
Computed tomography (CT) colonoscopy revealed an incidental finding of ill-defined nodular opacities in the right upper lobe of a 70-year-old man.
History. The patient had a history of hay fever (allergic rhinitis) and exposure to moldy hay (thermophilic Actinomyces species) on a dairy farm. He had a history of exposure to Al Eskan disease, or Desert Storm pneumonitis, in a Bedouin village in Riyadh, Saudi Arabia in 1991. His history was also positive for scoliosis, pertussis (diagnosed at age 65), and left lower lobe community-acquired bacterial pneumonia. He had no history of smoking. He had reported no hemoptysis and no weight loss.
Diagnostic tests. Routine colonoscopy findings of the descending colon were unremarkable; however, the colonoscope could not be advanced in the transverse colon to the hepatic flexure. Consequently, CT colonography without contrast, more popularly referred to as virtual colonoscopy, was performed, the results of which showed eventration of the left hemidiaphragm with a large hiatal hernia containing transverse colon (Figure 1), as well as incidentally noted ill-defined nodular opacities within the right upper lobe with concomitant ground-glass attenuation (Figure 2).

Figure 1. Virtual colonoscopy transverse image of the abdomen showed eventration of the left hemidiaphragm with a large hiatal hernia containing transverse colon.
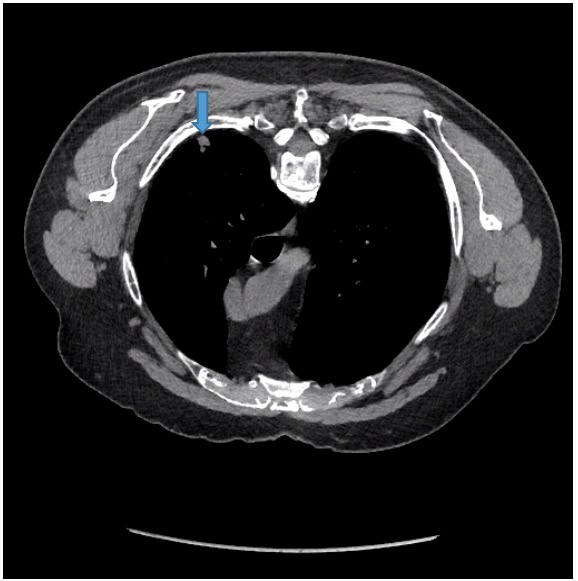
Figure 2. Also noted incidentally on virtual CT were ill-defined nodular opacities within the right upper lobe with concomitant ground-glass attenuation.
Given the finding of nodular lung opacities, a contrast-enhanced chest CT scan was ordered. A pre-CT scout radiograph (Figure 3) showed a hernia containing stomach, colon, and the tail of the pancreas. There was no axillary, mediastinal, or hilar lymphadenopathy. There was no pleural or pericardial effusion. The central airways were patent. There was left upper lobe, left lower lobe, and right lower lobe compressive atelectasis.
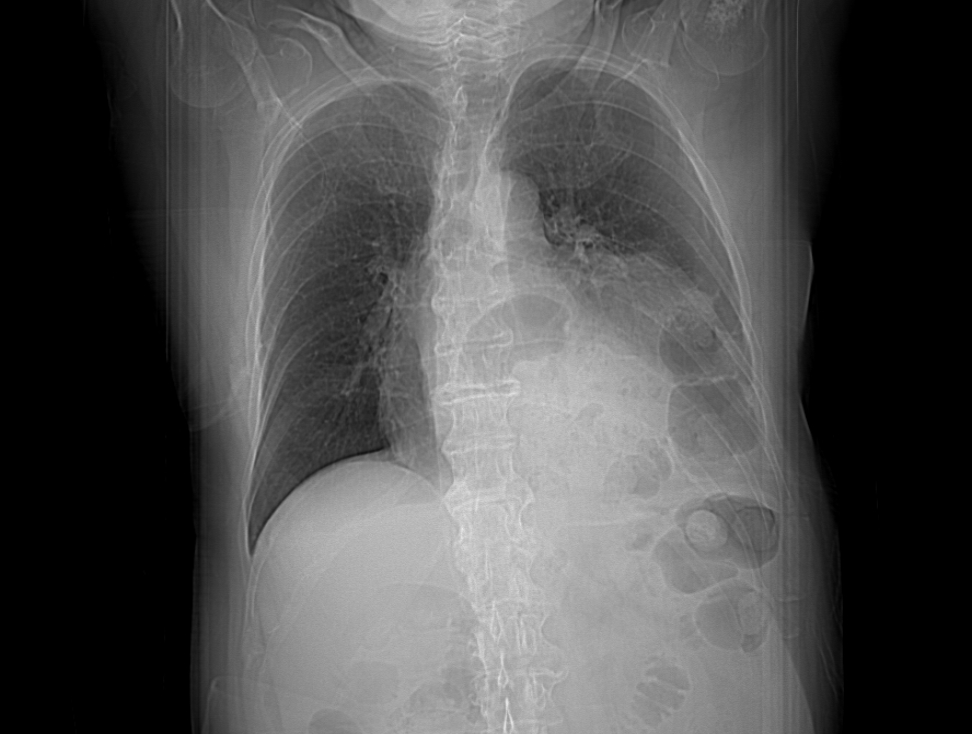
Figure 3. A pre-CT scout radiograph showed hernia containing stomach, colon and tail of pancreas with left upper lobe, left lower lobe, and right lower lobe compressive atelectasis.
Contrast-enhanced chest CT scans showed a 1-cm nodular opacity (Figure 4) and a 1.6-cm subpleural nodular opacity (Figure 5) within the apical segment of the right upper lobe, and ill-defined ground-glass opacity within the posterior segment of the right upper lobe (Figure 6).
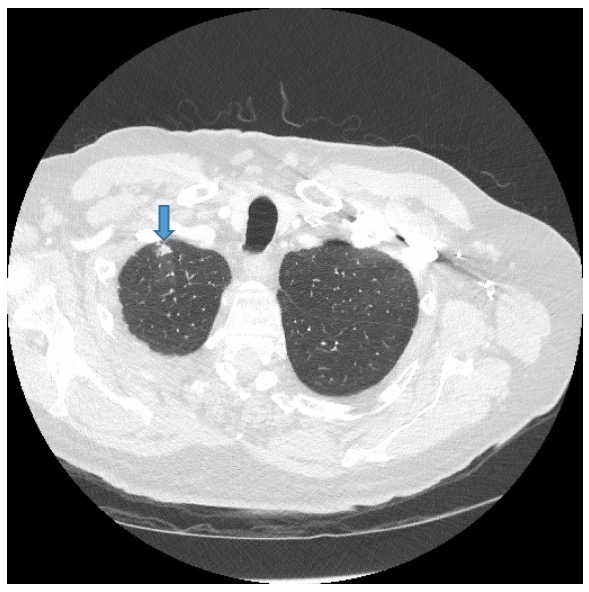
Figure 4. Contrast-enhanced chest CT showed a 1-cm nodular opacity in the apical segment of the right upper lobe.
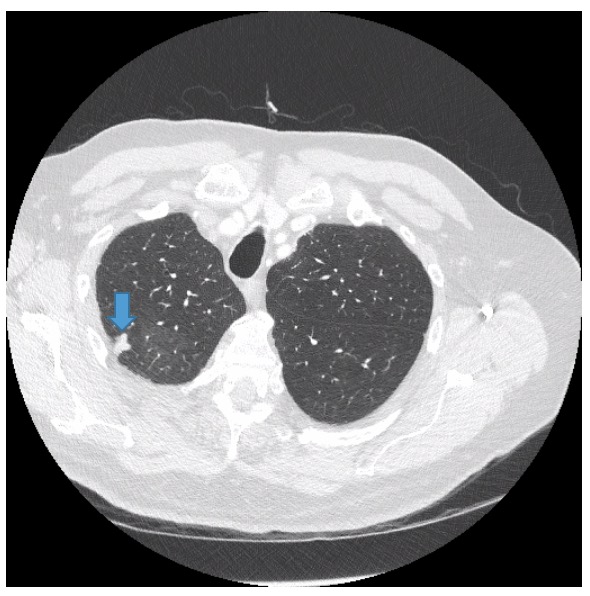
Figure 5. Contrast-enhanced chest CT showed a 1.6-cm subpleural nodular opacity within the apical segment of the right upper lobe.
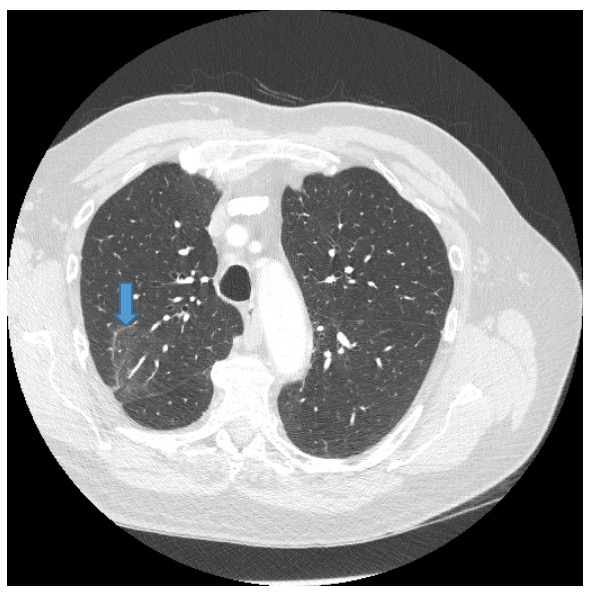
Figure 6. Contrast-enhanced chest CT showed ill-defined ground-glass opacity within the posterior segment of the right upper lobe.
The impression was right upper lobe nodular opacities for which 3-month follow-up would be recommended.
The gastroenterologist recommended a pulmonary consultation, at which pulmonary function tests showed normal diffusing capacity of the lungs for carbon monoxide, a restrictive pattern consistent with scoliosis, no evidence of interstitial lung disease, and no emphysema.
The 1-cm apical nodule in the right upper lobe was thought to be consistent with an old healed infection. To evaluate the 1.6 cm posterior subpleural right upper lobe nodule, a positron-emission tomography (PET) CT scan was ordered (Figure 7).
Figure 7. PET-CT scan.
The right upper lobe subpleural nodular opacity was not hypermetabolic, favoring a benign etiology. Follow-up with chest CT would be warranted in 3 months, per Fleischner Society guidelines for management of solid pulmonary nodules. The residual ground-glass opacities were likely inflammatory, with 1 of the nodules less-dense on the current study than on the prior CT. There was no evidence of hypermetabolic tumor or adenopathy to suggest neoplasm.
Although clinical and radiographic characteristics cannot reliably distinguish between benign and malignant nodules in most individuals, it is nevertheless important to estimate the clinical probability of malignancy before ordering imaging tests or biopsy procedures.1 Based on the extensively validated prediction model developed at the Mayo Clinic, our patient had a 21% chance of malignancy. The American College of Chest Physicians defines high-risk lung nodules as nodules with an estimated cancer risk of at least 65%.1
DISCUSSION
Large hiatal hernias (type 4) are associated with a large defect in the phrenoesophageal ligament and abnormal laxity of the gastrosplenic and gastrocolic ligaments, which allow parts of the stomach and other intra-abdominal organs to enter the hernia sac.2-4 It is unknown whether these predisposing abnormalities are acquired, congenital, or a combination of both.4 The average age at presentation is 72 years.
Approximately 1% to 2% of patients require a virtual CT colonoscopy to complete the examination, the results of which were normal in our patient’s case. Because there were no symptoms, no treatment was required. The gastroenterologist advised follow-up colonoscopy in 5 years, unless symptoms develop.
Extracolonic findings during virtual colonoscopy are common. Findings of high importance are those deemed likely to be of significance to the patient and the ones likely to require additional attention, either surgical or medical management, or mandating additional diagnostic tests. This category includes lung nodules and other masses.5,6
Countless pulmonary nodules are discovered each year during chest radiographs or CT scans. Most nodules are noncancerous and benign. A solitary pulmonary nodule is found on up to 0.2% of all chest radiographs. Lung nodules can be found on up to half of all lung CT scans. High-risk factors for malignant pulmonary nodules include older age, heavy smoking, larger nodule size, irregular or spiculated margins, and upper lobe location. Lung cancers occur more frequently in the upper lobes (70% of cancers), with a predilection for the right lung (odds ratio of approximately 2.0).7
Small solid nodules in a perifissural or subpleural location often represent intrapulmonary lymph nodes. Most functional imaging of lung nodules is done with fluorine-18 (18F)-labeled fluorodeoxyglucose (FDG) PET scan to differentiate between benign and malignant nodules. PET has sensitivity and specificity of approximately 90% for detecting malignant nodules with a diameter of 10 mm or larger.7 For solid nodules, low or no FDG uptake at PET/CT suggests benignity.
All PET-negative nodules should be observed with follow up CT scans to confirm stability for at least 2 years; if growth is observed, then biopsy is warranted.
REFERENCES:
- Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 suppl):e93S-120S.
- Krause W, Roberts J, Garcia-Montilla RJ. Bowel in chest: type IV hiatal hernia. Clin Med Res. 2016;14(2):93-96.
- Landreneau RJ, Del Pino M, Santos R. Management of paraesophageal hernias. Surg Clin North Am. 2005;85(3):411-432.
- Davis SS Jr. Current controversies in paraesophageal hernia repair. Surg Clin North Am. 2008;88(5):959-978.
- MacMahon H, Naidich DP, Goo JM, et al. Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology. 2017;284(1):228-243.
- Kimberly JR, Phillips KC, Santago P, et al. Extracolonic findings at virtual colonoscopy: an important consideration in asymptomatic colorectal screening. J Gen Intern Med. 2009;24(1):69-73.
- Truong MT, Ko JP, Rossi SE, et al. Update in the evaluation of the solitary pulmonary nodule. Radiographics. 2014;34(6):1658-1679.


