Peer Reviewed
Infantile Hemangioma
Authors:
Nujeen Zibari, MD
Baylor College of Medicine, Houston, Texas
Brandon Esianor, MD
Vanderbilt University, Nashville, Tennessee
Lynnette Mazur, MD, MPH
McGovern Medical School at UT Health, Houston, Texas
Citation:
Zibari N, Esianor B, Mazur L. Infantile hemangioma [published online April 4, 2019]. Consultant360.
A 2-week-old African American girl presented to clinic for a weight check and her second newborn screening examination. She had been born to a 34-year-old gravida 2 mother at 35 weeks of gestation via cesarean delivery secondary to maternal preeclampsia. Physical examination findings at that time were remarkable for a weight of 2.045 kg (0.6 percentile) and the presence of a 5 × 5-cm blue macule on the right neck. The mother and newborn had been discharged home after 5 days without complication. Because of the size and location of the lesion, she was referred to a dermatologist.
During the clinic visit, a localized, superficial 5 × 5-cm infantile hemangioma (IH) was present on the right neck (Figure 1). There was no bleeding or ulceration, and the child had no barky cough, stridor, or difficulty breathing.
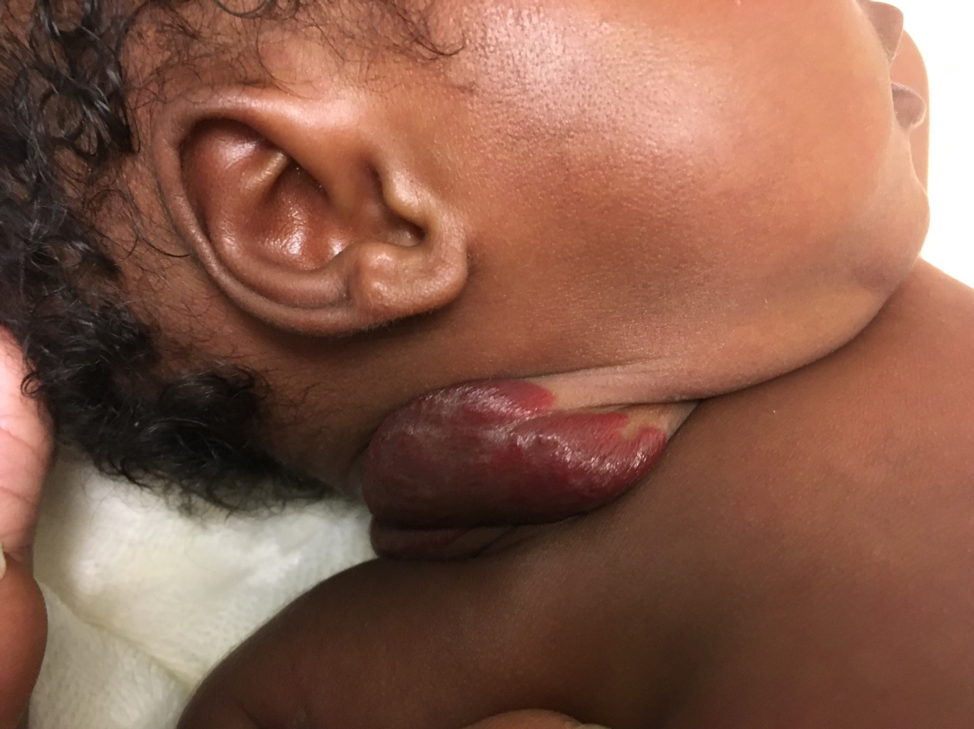
Figure 1. At presentation at 2 weeks of age, a localized, superficial 5 × 5-cm hemangioma was present on the newborn’s right neck.
In the few weeks’ interim between the initial visit and the visit to the dermatologist, the IH had increased in size and had ulcerated, and the girl had trouble turning her neck to breastfeed. The dermatologist started her on propranolol, 2 mg/kg/d divided 3 times a day, and daily wound care with topical mupirocin and petroleum jelly gauze. Because of the risk of hypoglycemia, the mother was advised to give propranolol with or after feeds and to hold the dose at times of diminished oral intake or vomiting.1 She was also educated on possible adverse effects of sleep disturbances, bronchial irritation, symptomatic bradycardia, and hypotension.1
When the patient returned to clinic for her 2-month immunizations, the ulceration had begun to heal. (Figure 2).
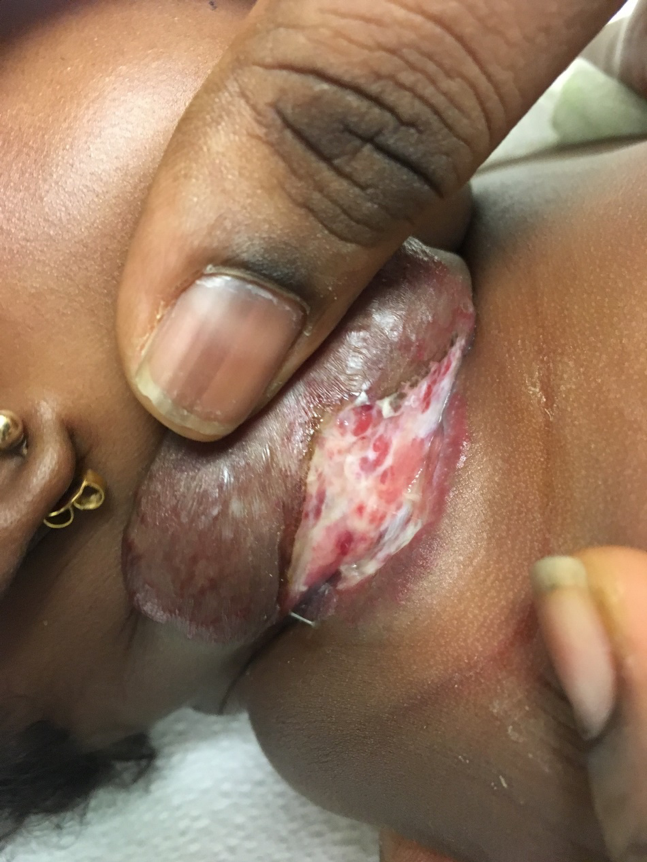
Figure 2. The patient’s infantile hemangioma at 6 weeks of age.
Discussion. Vascular lesions are characterized as malformations or neoplasms (Table 1). They are no longer clinically classified as a salmon patch or port-wine stain but rather are classified based on their cellular features, natural history, and clinical behavior.2
|
Table 1. Differential Diagnosis of Vascular Lesions |
|
Vascular Neoplasms |
|
Infantile hemangiomas |
|
Congenital hemangiomas (noninvoluting congenital hemangioma, rapidly involuting congenital hemangioma) |
|
Kaposiform hemangioendothelioma |
|
PHACE(S) association (posterior fossa abnormalities, segmental facial hemangiomas, arterial malformations, cardiac anomalies, eye abnormalities [sternal or supraumbilical defects]) |
|
Pyogenic granulomas |
|
Tufted angiomas |
|
Vascular Malformations |
|
Arterial |
|
Capillary or venulocapillary (formerly called port-wine stain or nevus flammeus) |
|
Lymphatic (formerly called cystic hygromas or lymphangiomas) |
|
Venous (formerly called cavernous hemangiomas or venous hemangiomas) |
Vascular malformations are structural anomalies that do not involute with time. IHs are benign vascular neoplasms characterized by abnormal proliferation of endothelial cells and aberrant blood vessel architecture that involute over time. A distinguishing feature of IHs is the expression of the glucose transporter protein isoform 1, a finding not present with other vascular lesions.
IHs affect up to 10% of infants in the first year of life. They are characterized by soft-tissue depth (superficial, deep, or combined) and anatomic appearance (localized, segmental, indeterminate, or multifocal). More than half occur in the head and neck, and two-thirds are localized rather than segmental or multifocal.2 The incidence is higher in infants who are white, female, breech, first-born, premature, and of low birth weight. Associations are also reported with older mothers, multiple-gestation pregnancies, placenta previa, preeclampsia, the use of fertility drugs, and a history of chorionic villus sampling or amniocentesis.2 Although not typically present at birth, a precursor blue-gray macule or cluster of telangiectasias may be noted.3
The natural history includes 2 phases: proliferation for approximately 12 months followed by spontaneous regression over the next several months.2 The most rapid growth occurs between 1 and 3 months of age, and 80% of size is reached by 3 months of age; almost all have completed growth by 5 months of age.2 Therefore, if treatment is indicated, the optimal time is before 1 month of age.
Ulceration is the most common complication and occurs in 5% to 20% of patients.2,4,5 Proposed mechanisms include increased tissue hypoxia, outgrowing the blood supply, or rapid expansion exceeding the elastic capabilities of the skin. IHs are more likely to ulcerate if they are large, are segmental, and are located in the head and neck, perioral, or perineal/perianal regions and intertriginous areas. Treatment may include topical antibiotics, barrier creams, wound dressings and oral analgesics or topical lidocaine for pain control. In refractory cases, pulsed-dye laser therapy may be effective.2,6
Patients with hemangiomas located in the head and neck area, specifically in the “beard” distribution (preauricular regions, chin, anterior neck, or lower lip), are at risk of airway compromise.7 Symptoms of obstruction include stridor, cough, and respiratory distress. Pain, disfigurement, functional impairment, and residual skin changes such as telangiectasia, fibrofatty tissue, redundant skin, anetoderma (atrophy and looseness of the skin), dyspigmentation, and scarring occur in 50% to 70% of patients.2,7
The traditional approach to treatment has been watchful waiting and parental reassurance. However, approximately one-third of patients eventually require some intervention.2 Treatment decisions are based on location, size, and stage of growth. Although topical β-blockers and high-potency corticosteroids are used for small, superficial IHs, systemic propranolol is preferred for larger lesions.2,8
Treatment of IHs with propranolol was incidentally discovered in 2008 when the drug was administered to a 4-month-old infant with cardiomyopathy, and the infant’s IH rapidly regressed.9 In 2014, propranolol was approved by the Food and Drug Administration in the United States for the treatment of IH. The mechanism of action likely involves vasoconstriction, angiogenesis inhibition, and stimulation of apoptosis of capillary endothelial cells.10 Relative contraindications include cardiogenic shock, sinus bradycardia, hypotension, heart block greater than first-degree, heart failure, bronchial asthma, and known hypersensitivity to the drug. Pretreatment cardiac screening is of limited value in patients with an unremarkable cardiac history and examination findings but may be recommended for infants with a baseline heart rate below normal for age, a family history of congenital heart conditions or arrhythmias, a maternal history of connective tissue disorder, or a family history of arrhythmia or arrhythmia on examination.11,12
Regression of the IH is typically seen within 2 weeks.2,13 Treatment is usually continued for a year and gradually tapered in order to prevent the rebound that may occur in up to 25% of patients.14 Alternative treatments can be considered for lesions that are refractory to β-blockers (Table 2).15
|
Table 2. Treatment Modalities |
|||
|
Treatment |
Indication |
Advantages |
Disadvantages |
|
β-blocker (oral) |
Large/complicated IHs; IHs affecting internal organs |
Safer, more effective than corticosteroids |
Acrocyanosis, bradycardia, bronchospasm, hypoglycemia, hypotension, seizures, sleep disturbances |
|
β-blocker (topical) |
Small/superficial IHs |
Avoids systemic side effects |
Bradycardia, bronchospasm, local irritation, sleep disturbances |
|
Corticosteroids (oral) |
Large/complicated IHs; IHs affecting internal organs |
When β-blockers are contraindicated |
Behavioral changes, cataracts, cushingoid changes, gastrointestinal upset, growth deceleration, hypertension, immunosuppression (increased risk of infection, poor response to vaccines), osteoporosis, suppression of hypothalamic-pituitary-adrenal axis, weight gain |
|
Corticosteroids (intralesional) |
Small, bulky, well-localized IHs |
Avoids systemic absorption |
Fat and/or dermal atrophy, growth deceleration, hypopigmentation |
|
Corticosteroids (topical) |
Small, localized, superficial IHs |
Avoids systemic absorption |
Acne, hypopigmentation, striae |
|
Interferon-α |
Refractory/corticosteroid-resistant IHs; Kasabach-Merritt phenomenon |
“Last resort” when all else fails |
Neurologic toxicities, cortical and motor function impairment, spastic diplegia (may not be reversible); requires subcutaneous administration |
|
Imiquimod |
Refractory/corticosteroid-resistant IHs |
Ease of use, lack of systemic side effects |
Hyperpigmentation, irritation and crusting at application site |
|
Pingyangmycin (bleomycin A5) |
Complicated IHs unresponsive to other therapies |
No systemic toxicity |
Pulmonary fibrosis |
|
Pulsed-dye Laser |
Refractory/corticosteroid-resistant IHs, superficial IHs |
Accelerates regression and rapidly reduces size of IH |
Atrophy, ulceration/scarring; large lesions may require general anesthesia |
|
Surgical excision |
Small, contraindication to or failure of pharmacotherapy, focal involvement in an anatomically favorable area, high likelihood that resection will be necessary in the future, revision of residual or redundant IH, areas in nose and lip that do not respond well to other therapy |
IH becomes relatively smaller with age and scar may be smaller |
Anesthetic risk in infants, blood loss, skin loss, nerve paralysis, wound infection |
|
Vincristine |
Refractory/corticosteroid-resistant/life-threatening IHs |
Limited side effects at therapeutic doses |
Alopecia, constipation, neurotoxicity (loss of deep tendon reflexes, cranial nerve palsies); requires central catheter |
Outcome of the case. Because our patient met several high-risk criteria, oral propranolol was started at 6 weeks of age. After a year of treatment, the IH had resolved but with residual skin changes (Figure 3). Future surgical resection and revision are planned.
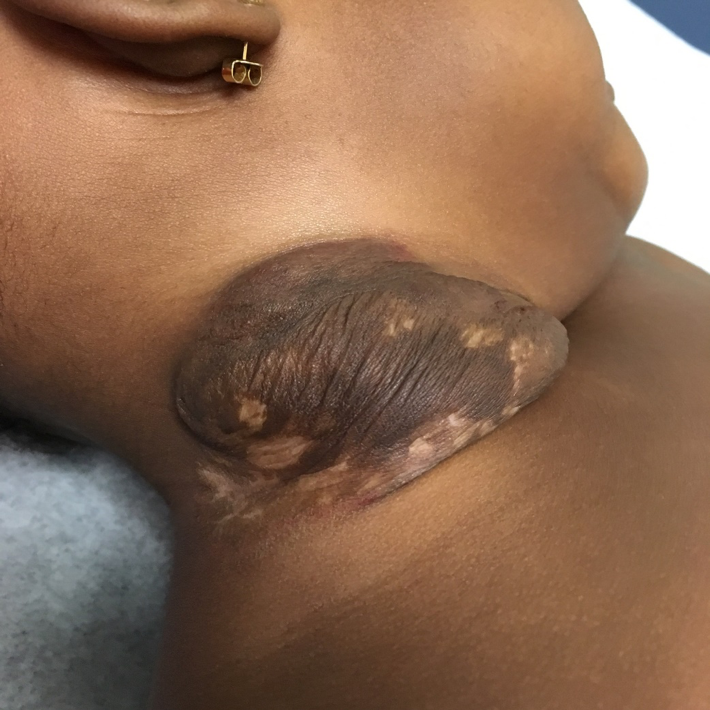
Figure 3. At 1 year of age, the patient’s hemangioma had resolved but with residual skin changes.
- de Graaf M, Breur JMPJ, Raphaël MF, Vos M, Breugem CC, Pasmans SGMA. Adverse effects of propranolol when used in the treatment of infantile hemangiomas: a case series of 28 infants. J Am Acad Dermatol. 2011;65(2):320-327.
- Darrow DH, Greene AK, Mancini AJ, Nopper AJ; Section on Otolaryngology–Head and Neck Surgery and Section on Plastic Surgery. Diagnosis and management of infantile hemangioma. Pediatrics. 2015;136(4):e1060-e1104.
- Smolinski KN, Yan AC. Hemangiomas of infancy: clinical and biological characteristics. Clin Pediatr (Phila). 2005;44(9):747-766.
- Shin HT, Orlow SJ, Chang MW. Ulcerated haemangioma of infancy: a retrospective review of 47 patients. Br J Dermatol. 2007;156(5):1050-1052.
- Kim HJ, Colombo M, Frieden IJ. Ulcerated hemangiomas: clinical characteristics and response to therapy. J Am Acad Dermatol. 2001;44(6):962-972.
- Morelli JG, Tan OT, Weston WL. Treatment of ulcerated hemangiomas with the pulsed tunable dye laser. Am J Dis Child. 1991;145:1062-1064.
- Mathew A, Eboh N, Brower GL. Infant with facial lesions and respiratory distress. Contemp Pediatr. 2016;33(11):7-38.
- Lv M-m, Fan X-d, Su L-x. Propranolol for problematic head and neck hemangiomas: an analysis of 37 consecutive patients. Int J Pediatr Otorhinolaryngol. 2012;76(4):574-57
- Léauté-Labrèze C, Dumas de la Roque E, Hubiche T, Boralevi F, Thambo J-B, Taïeb A. Propranolol for severe hemangiomas of infancy. N Engl J Med. 2008;358(24):2649-2651.
- Storch CH, Hoeger PH. Propranolol for infantile haemangiomas: insights into the molecular mechanisms of action. Br J Dermatol. 2010;63(2):269-274.
- Raphael MF, Breugem CC, Vlasveld FAE, et al. Is cardiovascular evaluation necessary prior to and during beta-blocker therapy for infantile hemangiomas? A cohort study. J Am Acad Dermatol. 2015;72(3):465-472.
- Drolet BA, Frommelt PC, Chamlin SL, et al. Initiation and use of propranolol for infantile hemangioma: report of a consensus conference. Pediatrics. 2013;131(1):128-140.
- Ng M, Knuth C, Weisbrod C, Murthy A. Propranolol therapy for problematic infantile hemangioma. Ann Plast Surg. 2016;76(3):306-310.
- Shah SD, Baselga E, McCuaig C, et al. Rebound growth of infantile hemangiomas after propranolol therapy. Pediatrics. 2016;137(4):e20151754.
- Nieuwenhuis K, de Laat PCJ, Janmohamed SR, Madern GC, Oranje AP. Infantile hemangioma: treatment with short course systemic corticosteroid therapy as an alternative for propranolol. Pediatr Dermatol. 2013;30(1):64-70.


