Ask the Experts: Q&A With Douglas DiRuggiero, PA-C, DMSc
Veteran dermatology Physician Assistant Douglas DiRuggiero, PA-C, DMSc, shares practical insights on how he incorporates ZORYVE® (roflumilast) into the treatment of atopic dermatitis, seborrheic dermatitis, and plaque psoriasis. From patient-friendly formulations to Mr DiRuggiero's real-world experience across diverse populations, he discusses why formulation and tolerability are key to success in inflammatory skin disease management.
INDICATIONS
ZORYVE cream, 0.3%, is indicated for the topical treatment of plaque psoriasis, including intertriginous areas, in adult and pediatric patients 6 years of age and older.
ZORYVE cream, 0.15%, is indicated for the topical treatment of mild to moderate atopic dermatitis in adult and pediatric patients 6 years of age and older.
ZORYVE topical foam, 0.3%, is indicated for the treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older.
ZORYVE topical foam, 0.3%, is indicated for the treatment of plaque psoriasis of the scalp and body in adult and pediatric patients 12 years of age and older.
Please see additional IMPORTANT SAFETY INFORMATION at the end of the article.
This article is based on an interview between Leyla Gulen and Douglas DiRuggiero, PA-C, DMSc.
Leyla Gulen: I have here Douglas DiRuggiero, a physician assistant at Skin Cancer and Cosmetic Dermatology. Douglas, tell us about yourself and your practice.
Douglas DiRuggiero: It's an honor to be here. Thank you. I've been practicing medical and surgical dermatology for nearly
25 years in northwest Georgia. I typically see around 50 to 60 patients per day ranging in age from birth to 100 years old,
with diverse backgrounds, ethnicities, and various economic status. This is one of the many reasons why I love the
specialty of dermatology.
Ms Gulen: That's very impressive. Would you say you see a lot of patients with inflammatory dermatoses?
Mr DiRuggiero: Yes. Plaque psoriasis, seborrheic dermatitis, atopic dermatitis—people with these inflammatory conditions represent a large part of my practice. I'd say roughly 50%.
Ms Gulen: So, when it comes to treating these patients, how does a medication like ZORYVE (roflumilast) fit into your practice? Are there any nuances in how you use it?
Mr DiRuggiero: Yes. Each formulation of ZORYVE is specifically designed for each of its indications, so ZORYVE cream, the 0.3%—it's indicated for topical treatment of plaque psoriasis, including intertriginous areas in adult and pediatric patients 6 years of age or older. Meanwhile, for atopic dermatitis, the skin barrier has been disrupted and topicals can penetrate more easily. In this case, ZORYVE cream 0.15% is indicated for topical treatment of mild to moderate atopic dermatitis in adult and pediatric patients 6 years of age and older. As for seborrheic dermatitis, there is more scalp involvement, and a cream can be challenging to apply. ZORYVE foam 0.3% is indicated for treatment of seborrheic dermatitis in adult and pediatric patients 9 years of age and older. One important consideration, though, is that ZORYVE is contraindicated in patients with moderate to severe liver impairment.
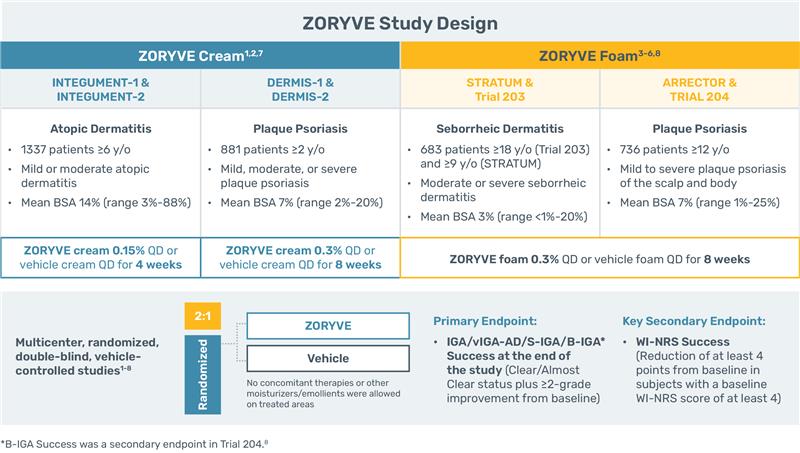
Ms Gulen: And you find those strengths reliably deliver the results you're looking for with each condition?
Mr DiRuggiero: Most definitely in my practice, the results I’ve seen in my patients on ZORYVE have been consistent with those in clinical trials. For instance, in atopic dermatitis clinical trials, twice as many patients achieved vIGA-AD Success at Week 4 with ZORYVE cream 0.15% (31%, n=884) vs vehicle (14%, n=453). Data are pooled analyses of the INTEGUMENT-1 and INTEGUMENT-2 studies. In seborrheic dermatitis clinical trials, 77% of patients achieved IGA Success at Week 8 with ZORYVE foam 0.3% (n=458) vs vehicle (53%, n=225). Data are pooled analyses of the STRATUM and Trial 203 studies. In plaque psoriasis cream clinical trials, six times more patients achieved IGA Success at Week 8 with ZORYVE cream 0.3% (40%, n=576) vs vehicle (7%, n=306). Data are pooled analyses of the DERMIS-1 and DERMIS-2 studies. In plaque psoriasis foam clinical trials, 64% of patients achieved S-IGA Success at Week 8 with ZORYVE foam 0.3% (n=481) vs vehicle (21%, n=255). Data are pooled analyses of the ARRECTOR and Trial 204 studies. ZORYVE is safe and well-tolerated across body locations—there are low rates of stinging or burning in each indication: 1.6% in atopic dermatitis, 1.3% in seborrheic dermatitis, and 0.4% in plaque psoriasis with ZORYVE cream 0.3%. Additionally, all formulations are not associated with folliculitis, atrophy, striae, or HPA-axis suppression. There were also low rates of discontinuation due to adverse events: in atopic dermatitis, 1.6% and 1.1% with ZORYVE and vehicle, respectively; in the seborrheic dermatitis trial, fewer than 1%; and in plaque psoriasis, 1%.
Ms Gulen: Now, you mentioned that ZORYVE has both cream and foam formulations. Is that important?
Mr DiRuggiero: Absolutely. I mean, formulation matters. If a patient finds their topical medication inconvenient or messy, or if they're juggling multiple products at once, adherence goes way down. It's helpful to have a topical that can be used across multiple parts of the body and a once-daily, patient-friendly formula. ZORYVE is all of these things. Additionally, as a steroid-free therapy, I'm not worried about skin atrophy or HPA-axis suppression.
Ms Gulen: You also mentioned the broad age range of patients you see daily. Do you have any concerns or considerations for younger patients with plaque psoriasis, seborrheic dermatitis, and atopic dermatitis?
Mr DiRuggiero: Yeah, I mean, I manage a large pediatric population of patients—many within the previously discussed age indications for ZORYVE. And in this group, formulation and tolerability really matter. I want topical therapies, especially for younger patients and with those who have sensitive skin, that is skin friendly and non-irritating. So it's reassuring that ZORYVE has no penetrating enhancers, no ceramide-stripping properties, no fragrances, ethanol, or propylene glycol, and, in fact, contains a novel, water-based emollient formulation to maintain moisture in the skin.
Ms Gulen: Well, thanks for sharing your thoughts with us again, Douglas.
IMPORTANT SAFETY INFORMATION
ZORYVE is contraindicated in patients with moderate to severe liver impairment (Child-Pugh B or C).
Flammability: The propellants in ZORYVE foam are flammable. Avoid fire, flame, and smoking during and immediately following application.
The most common adverse reactions (≥1%) for ZORYVE cream 0.3% for plaque psoriasis include diarrhea (3.1%), headache (2.4%), insomnia (1.4%), nausea (1.2%), application site pain (1.0%), upper respiratory tract infection (1.0%), and urinary tract infection (1.0%).
The most common adverse reactions (≥1%) for ZORYVE cream 0.15% for atopic dermatitis include headache (2.9%), nausea (1.9%), application site pain (1.5%), diarrhea (1.5%), and vomiting (1.5%).
The most common adverse reactions (≥1%) for ZORYVE foam 0.3% for seborrheic dermatitis include nasopharyngitis (1.5%), nausea (1.3%), and headache (1.1%).
The most common adverse reactions (≥1%) for ZORYVE foam 0.3% for plaque psoriasis include headache (3.1%), diarrhea (2.5%), nausea (1.7%), and nasopharyngitis (1.3%).
Please see full Prescribing Information for ZORYVE cream and full Prescribing Information for ZORYVE foam.
For more dermatologic disorders content, visit the Disease State Hub.
B-IGA=Investigator's Global Assessment-Body; BSA=body surface area; HPA=hypothalamic-pituitary-adrenal; IGA=Investigator's Global Assessment; QD=once daily; S-IGA=Investigator's Global Assessment-Scalp; vIGA-AD™=validated Investigator Global Assessment-Atopic Dermatitis; WI-NRS=Worst Itch Numeric Rating Scale; y/o=years old.
References
-
Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328(11):1073-1084.
-
ZORYVE® (roflumilast) cream. Prescribing information. Arcutis Biotherapeutics, Inc; 2024.
-
Blauvelt A, Draelos ZD, Gold LS, et al. Roflumilast foam 0.3% for adolescent and adult patients with seborrheic dermatitis: a randomized, double-blinded, vehicle-controlled, phase 3 trial. J Am Acad Dermatol. 2024;90(5):986-993.
-
ZORYVE® (roflumilast) foam. Prescribing information. Arcutis Biotherapeutics, Inc; 2025.
-
Data on File. Arcutis Biotherapeutics, Inc.
-
Gooderham MJ, Alonso-Llamazares J, Bagel J, et al. Roflumilast foam, 0.3%, for psoriasis of the scalp and body: the ARRECTOR phase 3 randomized clinical trial. JAMA Dermatol. 2025;161(7):698-706.
-
Simpson EL, Eichenfield LF, Alonso-Llamazares J, et al. Roflumilast Cream, 0.15%, for atopic dermatitis in adults and children: INTEGUMENT-1 and INTEGUMENT-2 randomized clinical trials. JAMA Dermatol. 2024;160(11):1161-1170.
-
Kircik LH, Alonso-Llamazares J, Bhatia N, et al. Once-daily roflumilast foam 0.3% for scalp and body psoriasis: a randomized, double-blind, vehicle-controlled phase IIb study. Br J Dermatol. 2023;189(4):392-399.
Marketed by Arcutis Biotherapeutics, Inc. and Kowa Pharmaceuticals America, Inc.
© 2025 Arcutis Biotherapeutics, Inc. All rights reserved. US-COM-PTF-00654 09/25


