Peer Reviewed
A Rare Extra-Gonadal Sac Tumor Presenting as a Labial Mass in a Child
Introduction. A previously healthy 2-year-old girl presented to the primary care physician with a right anterior labial mass.
History. The patient’s parents reported that the mass was first noticed a few weeks ago following a mild trauma during playing, and they initially attributed the painful swelling to the incident. While the onset of the mass was linked to a specific traumatic event, the parents' concern grew as the lesion enlarged rather than resolved. The theory of a simple traumatic injury was subsequently dismissed following an ultrasound that characterized the mass as a 1.7 cm solid lesion in the right labia majora concerning for a lipoblastoma, prompting the referral to a pediatric surgeon.
The patient’s past medical history was unremarkable. She had been meeting developmental milestones and had no known family history of early-onset malignancies. There were no systemic symptoms such as fever, weight loss, or change in appetite. By the time she was evaluated in surgery clinic, she was no longer symptomatic. Her physical examination revealed a firm, non-mobile mass in the anterior right labia abutting the right crus of the clitoris and measuring approximately 2 cm. The mass was no longer painful during physical examination. There were no signs of skin changes, ulceration, or concern of overlying infection. No inguinal or generalized lymphadenopathy was present.
Diagnostic and laboratory testing. Contrast-enhanced MRI of the pelvis was obtained for better lesion characterization and surgical planning. MRI demonstrated a well-circumscribed, predominantly solid lesion with some cystic components, measuring 2.8 x 2.0 x 2.6 cm in the right labial region. The mass was homogenously hypointense on T1-weighted images and intermediate intensity T2-weighted images (Figure 1). Post-contrast imaging revealed heterogeneous, predominantly peripheral enhancement (Figure 2). Diffusion-weighted imaging (DWI) suggested high cellularity with diffuse diffusion restriction (Figure 3). T1 imaging suggested the absence of microscopic fat, hemorrhage, or calcifications (Figures 1 and 4). It appeared confined to the labia without deep pelvic infiltration. There were no suspicious satellite lesions or lymphadenopathy in imaging.
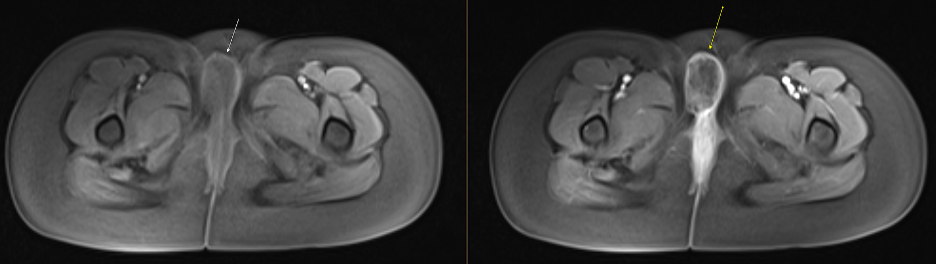
Figure 1. Pre (left) and Post-contrast (right) axial T1-weighted MR image reveals heterogeneous, predominantly peripheral enhancement of the lesion (arrow). Note the preserved tissue planes and absence of adjacent infiltration.
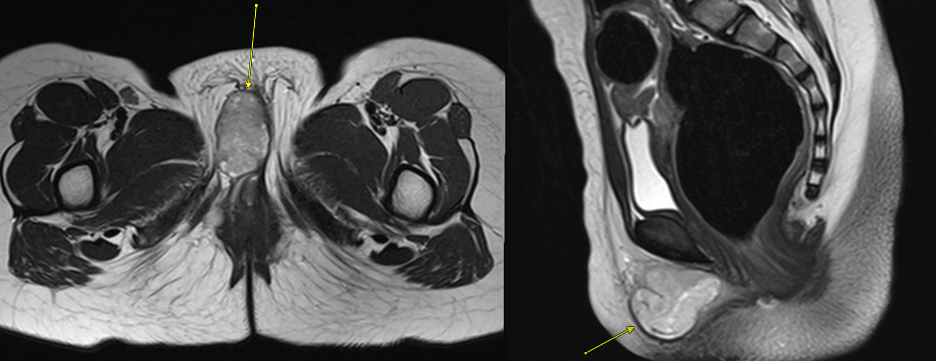
Figure 2. Pre-contrast axial T2-weighted MR image of the pelvis demonstrating a well-circumscribed, intermediate intensity lesion (arrow) within the right labia.
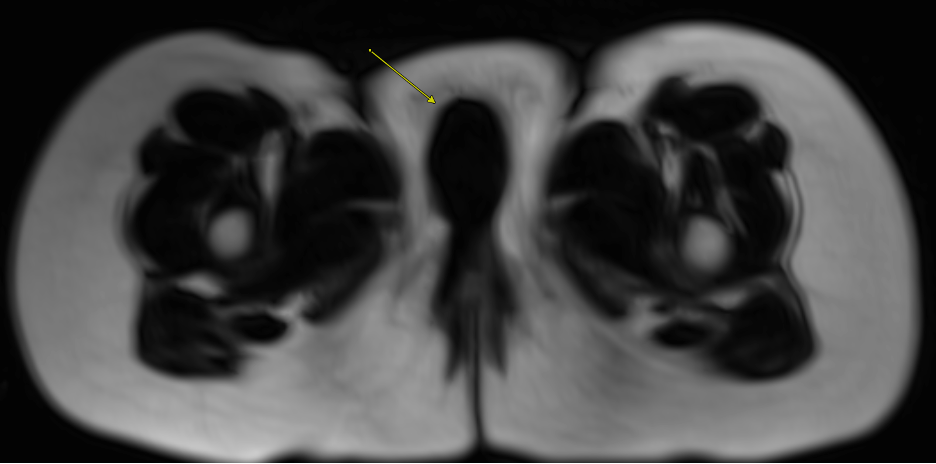
Figure 3. Pre-contrast axial T1-weighted Dixon "fat only" MR image demonstrating absence of fat signal within the right labial mass (arrow).
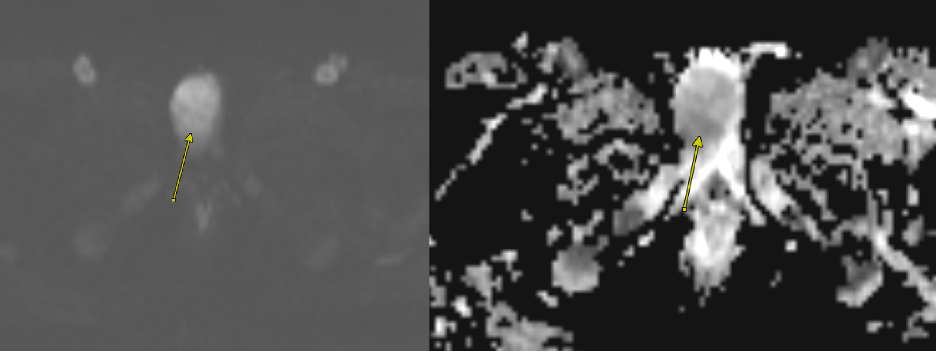
Figure 4. Axial diffusion-weighted imaging (DWI; left) and apparent diffusion coefficient (ADC; right) images show diffusion restriction within the mass (arrow). This finding suggests high cellularity and raises concern for malignancy.
Baseline laboratory evaluation, including serum tumor markers, was performed. Alpha-fetoprotein (AFP) was markedly elevated at approximately 1285 ng/mL (normal: <10 ng/mL for age), and lactate dehydrogenase (LDH) was mildly elevated. Beta-human chorionic gonadotropin (β-hCG) was within normal limits. These findings raised suspicion for a malignant neoplasm of germ cell origin.
Differential diagnosis. Differential diagnosis for a pediatric labial mass includes both benign and malignant lesions. Initially, lipoma or lipoblastoma was considered given the location and ultrasonography findings, but the physical examination raised concerns for malignancy including rhabdomyosarcoma and germ cell tumor. This was further supported by the MRI characteristics and markedly elevated AFP. Angiofibroblastoma and other mesenchymal tumors were also entertained but were less likely in the presence of significant AFP elevation and diffusion restriction. Lipomas and lipoblastomas were largely excluded considering the lack of fat content in imaging. A germ cell tumor, particularly a yolk sac tumor, became the leading concern.
Treatment and management. Tumor resection was planned for both diagnostic and treatment purposes. On the day of surgery, the evaluation began with a diagnostic cystourethroscopy to assess potential involvement of the genitourinary tract. This demonstrated no urethral or bladder mucosal abnormalities, limiting the resection to the right labia only. The tumor was well-encapsulated but firmly adherent to surrounding muscle fibers. A rim of tissue was resected en bloc with the mass to achieve clean margins, though the medial margin was particularly challenging due to proximity to the urethra and clitoris. Frozen section confirmed malignant cells. Additional margins were taken, all of which remained free of tumor, though a focal positive margin persisted medially.
Pathologic examination of the resected specimen confirmed a yolk sac tumor (3.5 cm) with focal lymphovascular invasion. Immunohistochemical staining was positive for cytokeratin AE1/AE3, SALL4, and glypican-3, all supportive of a yolk sac tumor. The tumor was negative for OCT-4 and CD30, which helped exclude other germ cell tumor subtypes. No additional germ cell components (such as dysgerminoma or embryonal carcinoma) were definitively identified. Given that no gonadal lesion was found and imaging ruled out a pelvic or abdominal primary site, an extragonadal primary was favored.
Staging workup included a contrast-enhanced CT of the chest, abdomen, and pelvis, which showed no evidence of metastatic disease. The lesion was therefore considered Stage II extragonadal germ cell tumor, given microscopic residual disease (positive margin) without nodal involvement or metastases.
After multidisciplinary discussion in pediatric solid tumor board, the patient underwent a port placement and began adjuvant chemotherapy, following Children’s Oncology Group (COG) guidelines for standard-risk extragonadal germ cell tumors. The family was offered enrollment in a clinical trial (AGCT1531) comparing standard cisplatin-based therapy with alternative chemotherapy approaches. Ultimately, they proceeded with adjuvant chemotherapy (carboplatin, etoposide, and bleomycin), administered in cycles every 21 days, for a planned total of four cycles.
Outcome and follow-up. Following surgical resection, the patient recovered well. Postoperative pain was managed with oral analgesics, and she was discharged home the following day. The surgical incision healed appropriately without infection or dehiscence, and the child resumed normal activities.
Baseline tumor markers post-surgery remained elevated, but were expected to decline with effective chemotherapy. The patient successfully underwent port placement and initiated her chemotherapy regimen within a few weeks of surgery. No immediate severe adverse events were noted during the first cycle. Per the AGCT1531 Standard Risk 1 protocol, the patient will undergo 5 years of post-therapy surveillance. Monitoring is most intensive during the first 2 years with frequent tumor markers and imaging, after which the frequency will decrease.1
Discussion. Extragonadal germ cell tumors (GCTs) are an uncommon subset of GCTs arising outside the gonads. They account for less than 3% of all GCTs in children under 15 years of age.2 These lesions can occur anywhere along the body's midline. The most common locations include the mediastinum, retroperitoneum, and brain, while the pineal gland and sacrococcygeal area are rarer. Isolated cases have been reported in the bladder, prostate, vulva, placenta, pelvis, uterus, kidney, and nasal sinuses.3
The embryologic origin of these tumors is attributed to abnormal migration of primordial germ cells. Early in fetal development, these cells originate near the yolk sac and travel along the dorsal mesentery to the gonadal ridges. Any misrouting or entrapment along this path can result in ectopic germ cells that may later undergo neoplastic transformation. In the vulvar or labial region, germ cells sequestered within the labioscrotal swellings may give rise to such tumors.4-6
Pediatric labial masses include both benign (e.g., lipoblastoma, hemangioma) and malignant lesions (e.g., soft tissue sarcomas, GCTs). Imaging with ultrasonography, CT, or MRI helps characterize these masses, and serum tumor markers often guide the differential diagnosis.7,8 Elevated AFP is classically associated with yolk sac tumors (endodermal sinus tumors) and embryonal cell tumors, whereas beta-human chorionic gonadotropin (β-hCG) is more indicative of choriocarcinoma, dysgerminoma, or mixed GCTs.8 Ultimately, histopathologic evaluation remains the gold standard for a definitive diagnosis.9
In this case, markedly elevated AFP levels and immunohistochemical positivity for SALL4 and glypican-3, coupled with the absence of OCT-4 and CD30, confirmed the diagnosis of a yolk sac tumor.8,10
Management of extragonadal GCTs generally involves surgical resection followed by adjuvant chemotherapy.6 In the Children’s Oncology Group (COG) protocols, standard-risk GCTs typically undergo four cycles of postoperative chemotherapy.11 Prognosis is generally favorable, with a 5-year relative survival exceeding 90% in the United States.12 Despite the high curability rate, even in cases of recurrent or metastatic disease, continued surveillance of tumor markers during and after therapy is performed to monitor treatment response and detect early recurrence.13 Our patient underwent surgical resection, followed by port placement, and is currently undergoing adjuvant chemotherapy.
Conclusion. In this case report, we reviewed the diagnosis and management of a rare extragonadal GCT located in the right labia majora of a 2-year old girl. The diagnosis of yolk sac tumor was established with a combination of imaging, tumor marker testing, and ultimate resection. While she had a focal positive margin medially, a radical resection was not indicated as these tumors respond well to neoadjuvant chemotherapy. Follow-up imaging and tumor marker surveillance will guide further management and treatment assessment.
AUTHORS:
Kevin Pierre MD1 • Anaiya Meikle2 • Ana Ruzic Do MD3 • Heather Liebe MD3 • Ensar Yekeler MD4 • Evelyn Anthony MD4 • Mariana Rodero Cardoso MD4 • Priya Sharma MD4
AFFILIATIONS:
1University of Florida Department of Radiology
2University of Florida College of Liberal Arts and Sciences
3University of Florida Department of Surgery, Division of Pediatric Surgery
4University of Florida Department of Radiology, Division of Pediatric Radiology
CITATION:
Pierre K, Meikle A, Ruzic Do A, Liebe H, Yekeler E, Anthony E, Rodero Cardoso M, Sharma P. A rare extra-gonadal sac tumor presenting as a labial mass in a child. Consultant. Published online August 27, 2025. doi: 10.25270/con.2025.08.000001
Received April 16, 2025. Accepted May 8, 2025.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Anaiya Meikle, University of Florida College of Liberal Arts and Sciences, 330 Newell Dr, Gainesville, FL 32603 (email: anaiya.meikle@ufl.edu)
References
- Children's Oncology Group. Active surveillance, bleomycin, etoposide, carboplatin or cisplatin in treating pediatric and adult patients with germ cell tumors. ClinicalTrials.gov; 2024. Accessed August 6, 2025. https://clinicaltrials.gov/study/NCT03067181
- Kaatsch P, Häfner C, Calaminus G, Blettner M, Tulla M. Pediatric germ cell tumors from 1987 to 2011: incidence rates, time trends, and survival. Pediatrics. 2015;135(1):e136-143. doi:10.1542/peds.2014-1989
- Ronchi A, Cozzolino I, Montella M, Panarese I, Zito Marino F, Rossetti S, et al. Extragonadal germ cell tumors: Not just a matter of location. A review about clinical, molecular and pathological features. Cancer Med. 2019;8(16):6832–40. doi:10.1002/cam4.2195
- Sadiq Q, Khan FA. Germ cell seminoma. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023. Accessed April 01. http://www.ncbi.nlm.nih.gov/books/NBK559241/
- De Felici M, Klinger FG, Campolo F, Balistreri CR, Barchi M, Dolci S. To be or not to be a germ cell: the extragonadal germ cell tumor paradigm. Int J Mol Sci. 2021;22(11):5982. doi:10.3390/ijms22115982
- Oosterhuis JW, Looijenga LHJ. Human germ cell tumours from a developmental perspective. Nat Rev Cancer. 2019;19(9):522–37. doi:10.1038/s41568-019-0178-9
- Gupta S, Paul P, Mitra PK, Kundu P, P Francis S. Advances in diagnosis, treatment, and prognosis of yolk sac tumors: a comprehensive review. International Journal of Science and Healthcare Research. 2023;8(2):456–462. doi:10.52403/ijshr.20230258
- Jamshidi P, Taxy JB. Educational case: yolk sac (endodermal sinus) tumor of the ovary. Acad Pathol. 2020;7:2374289520909497. doi:10.1177/2374289520909497
- Dalal PU, Sohaib SA, Huddart R. Imaging of testicular germ cell tumours. Cancer Imaging. 2006;6(1):124–34. doi:10.1102/1470-7330.2006.0020
- Sharma R, Khera S, Sinha A, Yadav T. Pure yolk sac tumor of sacrococcygeal region. Autopsy Case Rep. 2021;11:e2021287. doi:10.4322/acr.2021.287
- Bhuta R, Shah R, Gell JJ, Poynter JN, Bagrodia A, Dicken BJ, et al. Children’s Oncology Group’s 2023 blueprint for research: germ cell tumors. Pediatr Blood Cancer. 2023;70(Suppl 6):e30562. doi:10.1002/pbc.30562
- Poynter JN, Amatruda JF, Ross JA. Trends in incidence and survival of pediatric and adolescent germ cell tumors in the United States, 1975-2006. Cancer. 2010;116(20):4882–91. doi:10.1002/cncr.25454
- Pedrazzoli P, Rosti G, Soresini E, Ciani S, Secondino S. Serum tumour markers in germ cell tumours: From diagnosis to cure. Crit Rev Oncol Hematol. 2021;159:103224. doi:10.1016/j.critrevonc.2021.103224
©2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


