Myxofibrous Tumor Progression to Osteosarcoma
Introduction. A 67-year-old man, monitored since October 2022 for an incidental left proximal femoral lesion, arrived as a transfer from an outside hospital where he presented with an inability to walk after feeling a “pop” and was found to have a pathologic left subtrochanteric fracture in December 2023.
History. The patient is a 67-year-old man with a history of chronic obstructive pulmonary disease, deep vein thrombosis (DVT), arthritis, Factor V Leiden, left knee replacement, and monostotic fibrous dysplasia of the left femur. In October 2022, the patient was found to have an incidental left femoral bone lesion on abdominal computed tomography (CT) during workup for abdominal pain at an outside facility. In December 2023, the patient sustained a left subtrochanteric femur fracture while walking during which he felt a “pop” and underwent open biopsy and cephalomedullary nail fixation at our institution. During the next 1.5 years, imaging revealed progressive disease of his left femur, resulting in the patient undergoing multiple biopsies and eventually total femur arthroplasty. His symptoms included pain and swelling of the left knee and hip.
Diagnostic testing. The patient’s initial radiographs from December 2023 demonstrated a lucent lesion (Figure 1) in the proximal femur with cortical thinning, later progressing to a subtrochanteric fracture (Figure 2) and cortical destruction with expansion (Figure 3)1,2, corresponding with biopsy proven fibrous dysplasia with secondary cystic changes.
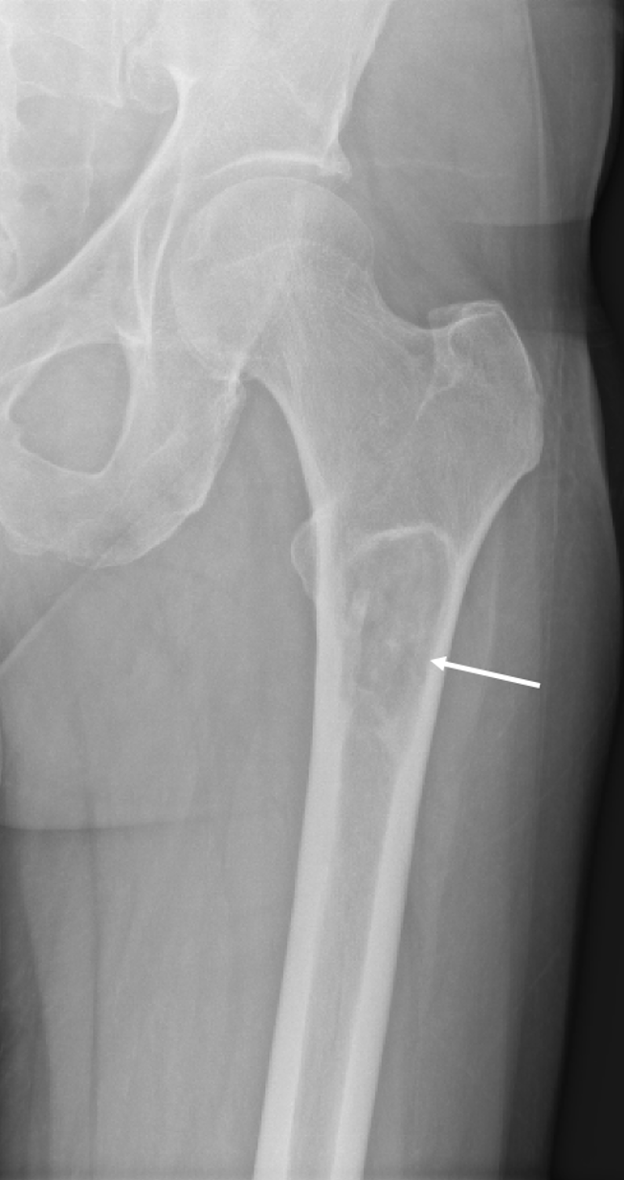
Figure 1. Initial plain radiograph of left proximal femur. Lucent lesion (white arrow) in the proximal left femur with cortical thinning.
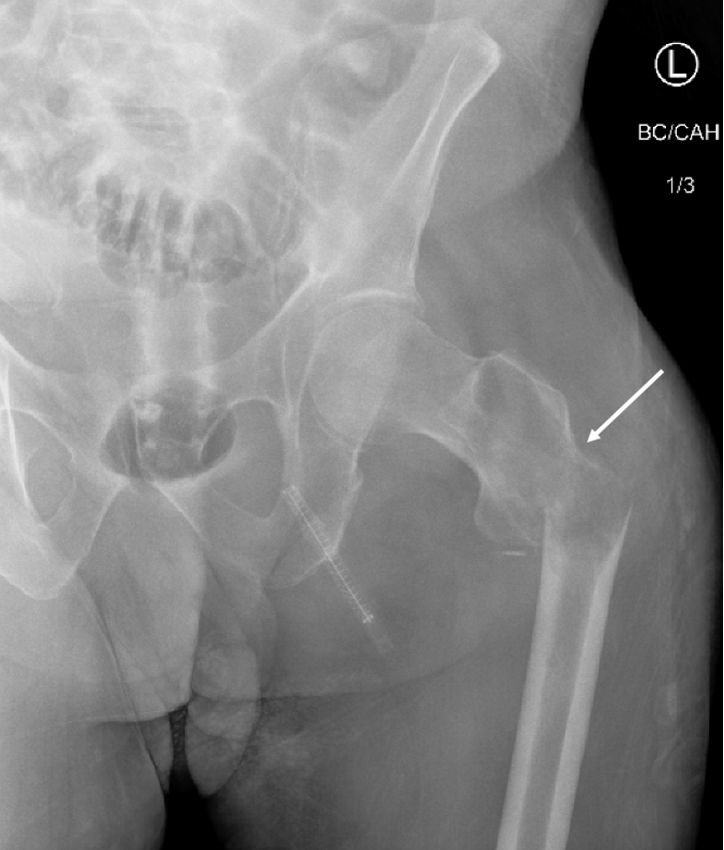
Figure 2. Plain radiograph of left proximal femur. Progression to displaced pathologic left subtrochanteric femur fracture with cortical destruction (white arrow).
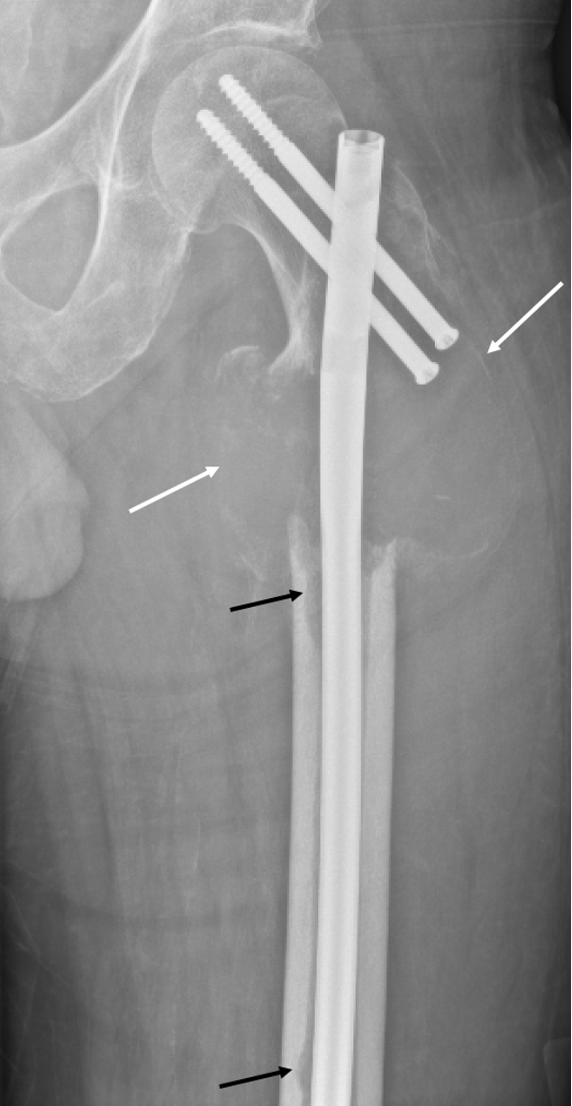
Figure 3. Plain radiograph of left proximal femur. The lesion has enlarged with increased periosteal bone formation and cortical destruction (white arrows). There are new lucent endosteal foci (black arrows) in the medial mid and distal femoral diaphyseal cortices. Postsurgical changes of intramedullary nailing with interlocking screw fixation of pathologic fracture of the subtrochanteric left proximal femur.
In July 2024, a follow-up magnetic resonance imaging (MRI) revealed a multilobulated destructive mass with hemorrhagic, cystic, and solid extraosseous components (Figure 4).2 The lesion extended into the femoral neck and proximal diaphysis, with T2-hyperintense cystic regions and internal septations.3 At this time, repeat biopsies showed a low-grade giant cell rich neoplasm.
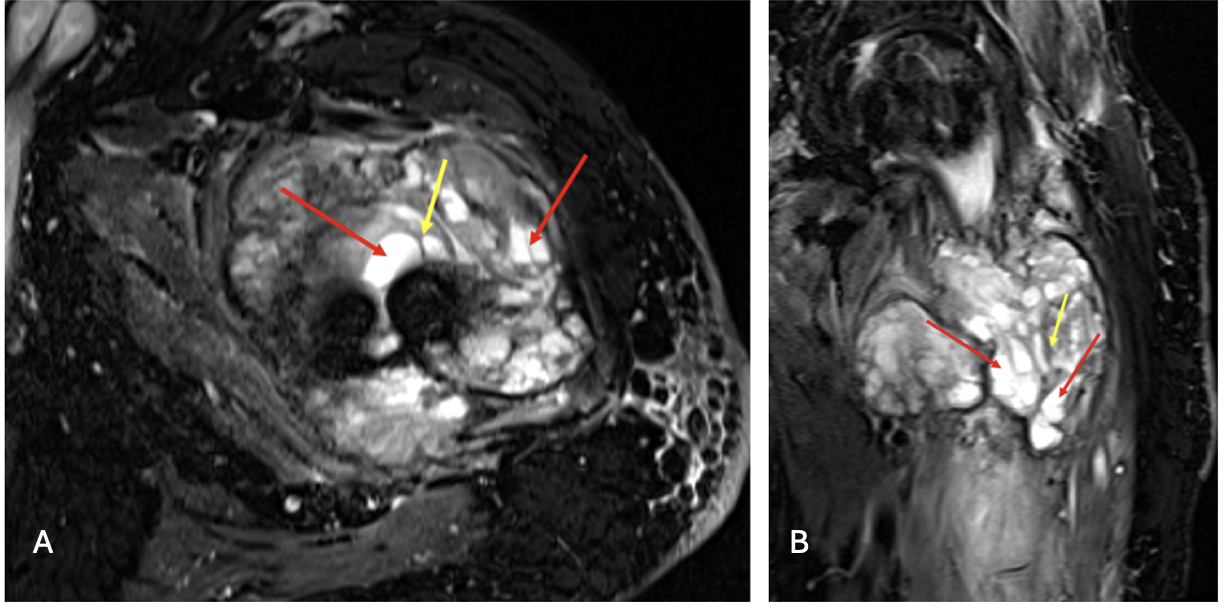
Figure 4A-B. MRI STIR WARP axial (a) and coronal (b) images of the left femur. Heterogeneous, multilobulated soft tissue mass at the fracture site with osseous involvement of the femoral neck, intertrochanteric region, and proximal diaphysis. Internal low-signal septations (yellow arrow) and T2-hyperintense cystic components (red arrows) are present, compatible with a giant cell–rich fibro-osseous neoplasm. New mass consistent with biopsy proven low giant cell rich neoplasm with GNAS mutation.
One year later, follow-up radiographs revealed an aggressive, permeative, marrow-replacing lesion in the distal femoral metadiaphysis, confirmed by CT to have cortical destruction and aggressive growth (Figure 5)4,5, consistent with low-grade sarcoma on biopsy.
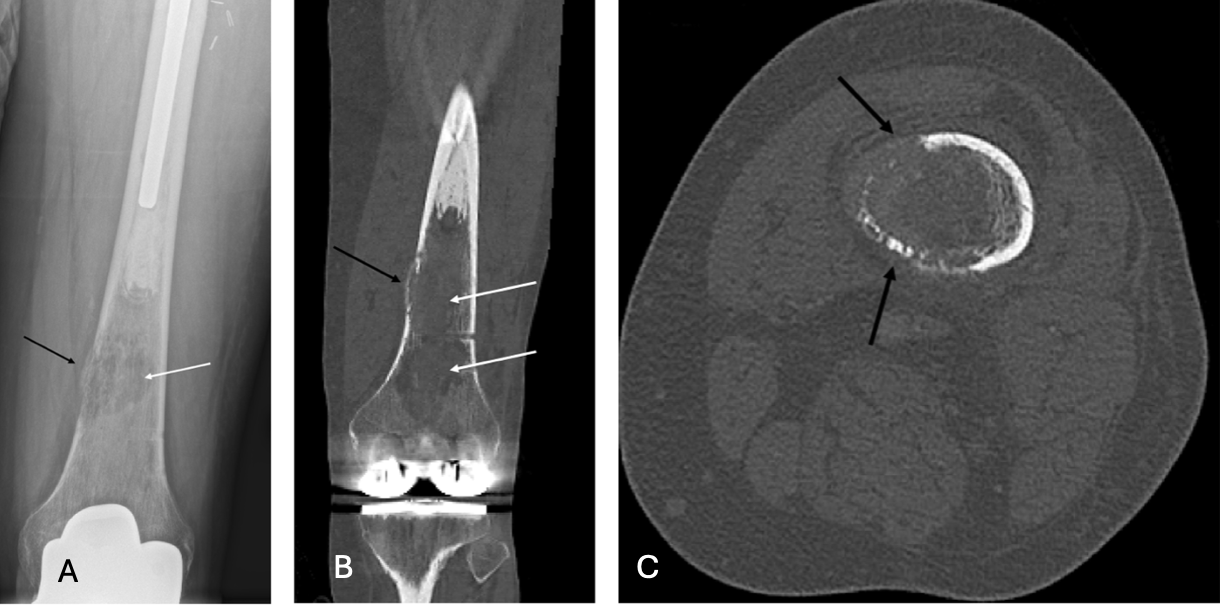
Figure 5A-C. Plain radiograph (a), coronal CT (b), and axial CT (c) of left distal femur. New lucent, aggressive marrow-replacing lesion (white arrow) of the distal femoral diametaphysis with cortical thinning and permeation (black arrow), corresponding with biopsy-proven low-grade osteosarcoma.
The diagnosis of LSMFT for the initial proximal femoral lesion was favored due to its typical location, mixed fibro-osseous imaging appearance, histologic heterogeneity, and presence of GNAS mutation. 2,3,8,9 Additionally, the reported malignant transformation rate of liposclerosing myxofibrous tumor (LSMFT) is approximately 10%–16% compared to the 0.5% risk seen in fibrous dysplasia, favoring the initial proximal femoral lesion to be LSMFT rather than fibrous dysplasia with subsequent biologic progression consistent with malignant transformation.5,10,11, 12,13 A combination of clinical presentation, imaging features, lesion location, histopathology, and exclusion of mimicking entities, which is discussed further below, led to the initial diagnosis of LSMFT.
Differential Diagnoses. Fibrous dysplasia, unicameral bone cyst, aneurysmal bone cyst, non-ossifying fibroma, and intraosseous lipoma are differential diagnoses to consider. Liposclerosing myxofibrous tumor (LSMFT) contains fibrous dysplasia-like features, making it difficult to distinguish from fibrous dysplasia.3 Some findings seen in fibrous dysplasia that help to discern these entities are the decreased sclerosis, increased uptake on bone scintigrams, and low to intermediate signal intensity on fluid-sensitive MRI sequences.3 Unicameral bone cysts may demonstrate proliferation of fibrous tissue with reactive bone during healing, which appear similar to LSMFT.17 However, unicameral bone cysts and similarly aneurysmal bone cysts are differentiated by their largely cystic components compared to the heterogeneous appearing LSMFT on MRI.3,18, 19 Intraosseous lipoma may share some features with LSMFT but is differentiated by the identification of fat on CT or MRI.3 Non-ossifying fibroma has a predilection for the distal femur and benign fibrous tissue features, which distinguishes it from LSMFT which typically occurs in the proximal femur and has multiple components on imaging.6 In addition, non-ossifying fibroma and aneurysmal bone cyst typically occur in the young population.6,19
Treatment and management. Following initial fixation, progressive disease prompted a proximal femur replacement, which was later converted to a total femur arthroplasty (Figure 6) due to distal extension and sarcomatous transformation. He was referred to radiation oncology for radiation therapy, with the first session in August 2024, and the therapy was completed in September 2024.
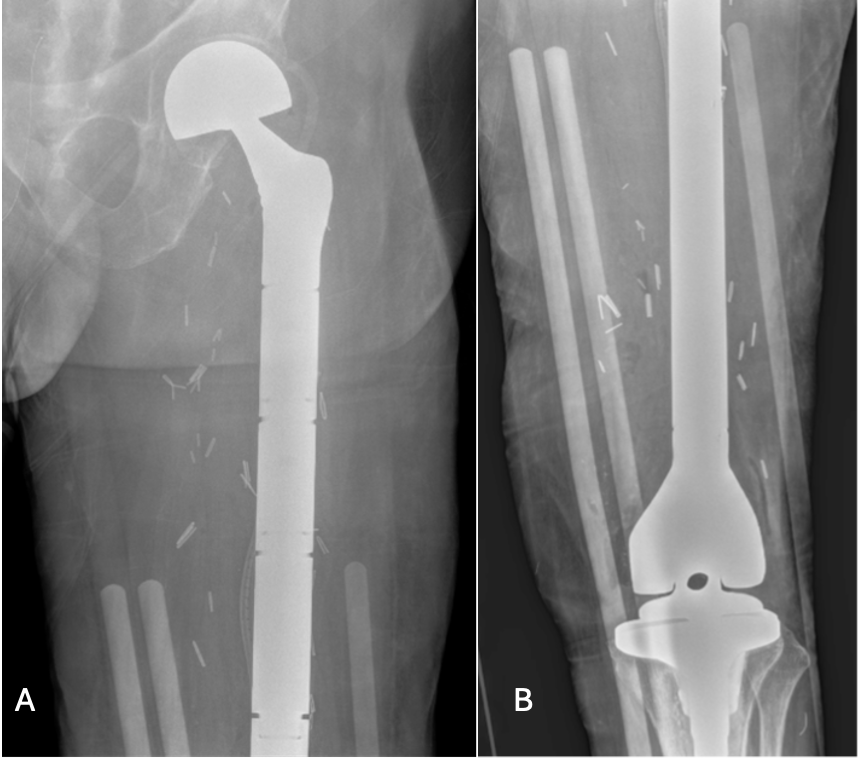
Figure 6A-B. Plain radiograph of left femur. Postsurgical changes of left femur resection with megaprosthesis placement and articulating tibial component.
Outcome and follow-up. The patient developed chronic draining sinus, infected prosthesis, and bilateral upper extremity DVT a few months after total left femur arthroplasty. The patient underwent explantation of the left femur hardware, debridement, and antibiotic spacer placement and later underwent left total femur arthroplasty. Patient completed a 15-day course of ciprofloxacin 500 mg twice daily prior to explantation and postoperatively started on cefepime 2g twice daily transitioning to Zosyn 4.5g every 6 hours due to pancytopenia for a combined total 7-day course with left knee joint cultures growing Pseudomonas aeruginosa. After discharge, patient completed a 42-day course of Zosyn 18g daily continuous infusion and was prescribed apixaban (Eliquis). Looking forward, the patient continues to follow up with orthopedic oncology and currently walks with a walker while working on mobility with home exercise with the goal to walk with a cane.
Discussion. Liposclerosing myxofibrous tumor (LSMFT) is a rare fibro-osseous lesion characterized by myxoid, fibrous, lipomatous, and fibroxanthomatous components, often resembling fibrous dysplasia.1,2,6-8 It predominantly affects the intertrochanteric region of the femur, accounting for 80%–90% of cases.1,2,5 Etiology remains unclear, with degenerative changes or post-traumatic fibrous dysplasia suggested, supported by GNAS and TP53 mutations.3,9
The risk of malignant transformation in LSMFT is estimated at 10%–16%, though this may be overestimated due to referral and publication bias;5,10,11 some studies suggest a 0.5% risk, similar to fibrous dysplasia.10,12,13 Transformation is typically indolent over a prolonged period. Osteosarcoma is the most common post-transformation histology, though malignant fibrous histiocytoma, malignant fibroxanthoma, and high-grade spindle cell sarcomas are also reported.5,10-13,14
This case shows a possible stepwise progression from LSMFT to giant cell–rich fibro-osseous neoplasm to low-grade osteosarcoma. Transitional giant cell-rich phases are rarely reported, and, based on our research, only one similar case exists.2,13,15 The one similar case in the literature describes a patient who had LSMFT in the left femoral neck, with associated pathologic fracture, that evolved into benign giant cell reactive lesion and then into secondary osteosarcoma involving the proximal thigh.2 Sampling error or dual primary tumors is unlikely, given repeated histology and molecular testing.14,16 Needle biopsies have high diagnostic accuracy, with sensitivity up to 100% and specificity 90% for musculoskeletal malignancies.16
Histology can lag behind clinical and imaging findings, but relying solely on biopsy can miss malignant transformation. Aggressive clinical behavior (rapid growth, recurrent lesions, disproportionate pain) warrants suspicion even with benign histology. Long-term imaging surveillance and a low threshold for repeat biopsy and molecular profiling are crucial, especially with aggressive radiographic features like cortical destruction or extraosseous extension.5,6,14
Malignant transformation of LSMFT is rare but may follow a stepwise process through intermediate benign-appearing lesions. Multimodal assessment (radiology, pathology, molecular studies) is essential to guide timely intervention.5-7,14 Radiologists and clinicians should be aware of any changes in clinical symptoms or imaging findings that warrant suspicion for malignant transformation and initiation multimodal assessment.
AUTHORS
Kalyani N. Ballur, MD1, Oluwaferanmi T. Dada, BS2, Evan Maroun, MS3, Kevin Pierre, MD1, Troy Storey, MD1, Diego A L Garcia, MD¹
AFFILIATIONS
1Department of Radiology, University of Florida, Gainesville, Florida, USA
2University of Florida College of Medicine, Gainesville, Florida, USA
3Nova Southeastern College of Osteopathic Medicine, Fort Lauderdale, Florida, USA
CITATION
Ballur KN, Dada OT, Maroun E, Pierre K, Storey T, Garcia ALD. Myxofibrous tumor progression to osteosarcoma. Consultant. Published online April 22, 2026. DOI:10.25270/con.2026.04.000004
Received Dec. 21, 2025. Accepted Feb. 3, 2026.
DISCLOSURES
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS
None.
CORRESPONDENCE:
Kalyani N. Ballur, MD, Department of Radiology, University of Florida, 1600 SW Archer Rd, Gainesville, FL 32608 (email: kalyani.ballur@ufhealth.org)
References
- Regado ER, Garcia PB, Caruso AC, et al. Liposclerosing myxofibrous tumor: A series of 9 cases and review of the literature. J Orthop. 2016;13(3):136-139. Published 2016 Mar 26. doi:10.1016/j.jor.2016.03.003
- Campbell K, Wodajo F. Case report: two-step malignant transformation of a liposclerosing myxofibrous tumor of bone. Clin Orthop Relat Res. 2008;466(11):2873-2877. doi:10.1007/s11999-008-0362-9
- Murphey MD, Carroll JF, Flemming DJ, Pope TL, Gannon FH, Kransdorf MJ. From the archives of the AFIP: benign musculoskeletal lipomatous lesions. Radiographics. 2004;24(5):1433-1466. doi:10.1148/rg.245045120
- Matsunobu T, Maekawa A, Jotatsu M, Makihara K, Hisaoka M, Iwamoto Y. Malignant transformation of giant cell tumor of bone 7 years after initial surgery: a case report and literature review. JBJS Case Connect. 2021;11(2):e20.00417. Published 2021 Apr 20. doi:10.2106/JBJS.CC.20.00417
- Kransdorf MJ, Murphey MD, Sweet DE. Liposclerosing myxofibrous tumor: a radiologic-pathologic-distinct fibro-osseous lesion of bone with a marked predilection for the intertrochanteric region of the femur. Radiology. 1999;212(3):693-698. doi:10.1148/radiology.212.3.r99se40693
- Deel C, Hassell L. Liposclerosing myxofibrous tumor: a review. Arch Pathol Lab Med. 2016;140(5):473-476. doi:10.5858/2014-0503-RS
- Zhang M, Zhang D, Yu W, Wang C. Liposclerosing myxofibrous tumor of the distal femur: A case report. Front Surg. 2023;9:1009975. Published 2023 Jan 6. doi:10.3389/fsurg.2022.1009975
- Pena-Burgos EM, Serra Del Carpio G, Tapia-Viñe M, et al. Liposclerosing myxofibrous tumor: a separated clinical entity? Diagnostics (Basel). 2025;15(5):536. Published 2025 Feb 22. doi:10.3390/diagnostics15050536
- Matsuba A, Ogose A, Tokunaga K, et al. Activating Gs alpha mutation at the Arg201 codon in liposclerosing myxofibrous tumor. Hum Pathol. 2003;34(11):1204-1209. doi:10.1016/s0046-8177(03)00430-1
- Boughzala-Bennadji R, Stoeckle E, Le Péchoux C, et al. Localized myxofibrosarcomas: roles of surgical margins and adjuvant radiation therapy. Int J Radiat Oncol Biol Phys. 2018;102(2):399-406. doi:10.1016/j.ijrobp.2018.05.055
- Wilson DAJ, Gazendam A, Visgauss J, et al. Designing a rational follow-up schedule for patients with extremity soft tissue sarcoma. Ann Surg Oncol. 2020;27(6):2033-2041. doi:10.1245/s10434-020-08240-z
- Gilkey FW. Arch Pathol Lab Med. 1999;123:1069–1074. PMID: 10484560
- Marui T, Yamamoto T, Yoshihara H, Kurosaka M, Mizuno K, Akamatsu T. De novo malignant transformation of giant cell tumor of bone. Skeletal Radiol. 2001;30(2):104-108. doi:10.1007/s002560000305
- Matsuyama A, Yonemitsu N, Hayashida S, Watanabe K, Sugihara H, Inokuchi A. Case of postradiation osteosarcoma with a short latency period of 3 years. Pathol Int. 2003;53(1):46-50. doi:10.1046/j.1440-1827.2003.01427.x
- Milgram JW. Malignant transformation in bone lipomas. Skeletal Radiol. 1990;19(5):347-352. doi:10.1007/BF00193088
- Mitsuyoshi G, Naito N, Kawai A, et al. Accurate diagnosis of musculoskeletal lesions by core needle biopsy. J Surg Oncol. 2006;94(1):21-27. doi:10.1002/jso.20504
- Dattilo J, McCarthy EF. Liposclerosing myxofibrous tumor (LSMFT), a study of 33 patients: should it be a distinct entity? Iowa Orthop J. 2012;32:35-39.
- Evans J, Shamrock AG, Blake J. Unicameral bone cyst. [Updated 2023 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470587/
- Nasri E, Reith JD. Aneurysmal bone cyst: a review. J Pathol Transl Med. 2023 Mar;57(2):81-87. doi: 10.4132/jptm.2023.02.23.
©2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


