Generalized Eruptive Keratoacanthomas Arising in the Setting of Anti-Cancer Therapy: An Illustrative Case with Spontaneous Resolution
Introduction. A 61-year-old man undergoing treatment for metastatic urothelial carcinoma of the bladder with pembrolizumab and enfortumab vedotin-ejfv presented with new-onset painful nodules on the bilateral arms, legs, and feet.
History. The patient had a history of metastatic urothelial carcinoma of the bladder, initially diagnosed in March 2020. He began treatment with pembrolizumab (200 mg IV every 3 weeks) in May 2023, which he continued until February 2024. Due to concern for new lung metastases developing while on pembrolizumab, enfortumab vedotin-ejfv (1.25 mg/kg IV) was added beginning February 28, 2024, at which time both agents were administered. Subsequent doses of enfortumab vedotin-ejfv were given without pembrolizumab on March 13, March 26, and April 2, 2024. Six weeks after his last infusion of pembrolizumab and 8 days after his most recent dose of enfortumab vedotin-ejfv, he developed new-onset firm nodules on his extremities, and anticancer therapy was discontinued. He had no other notable medical history and had never developed a similar reaction.
The patient’s physical examination revealed firm, skin-colored to slightly hypopigmented verrucous papules diffusely spread across the bilateral arms (Figure 1A and B), legs, and feet. Due to the rapid progression and painful nature of the eruption, he was hospitalized for 8 days for a suspected severe cutaneous drug reaction.
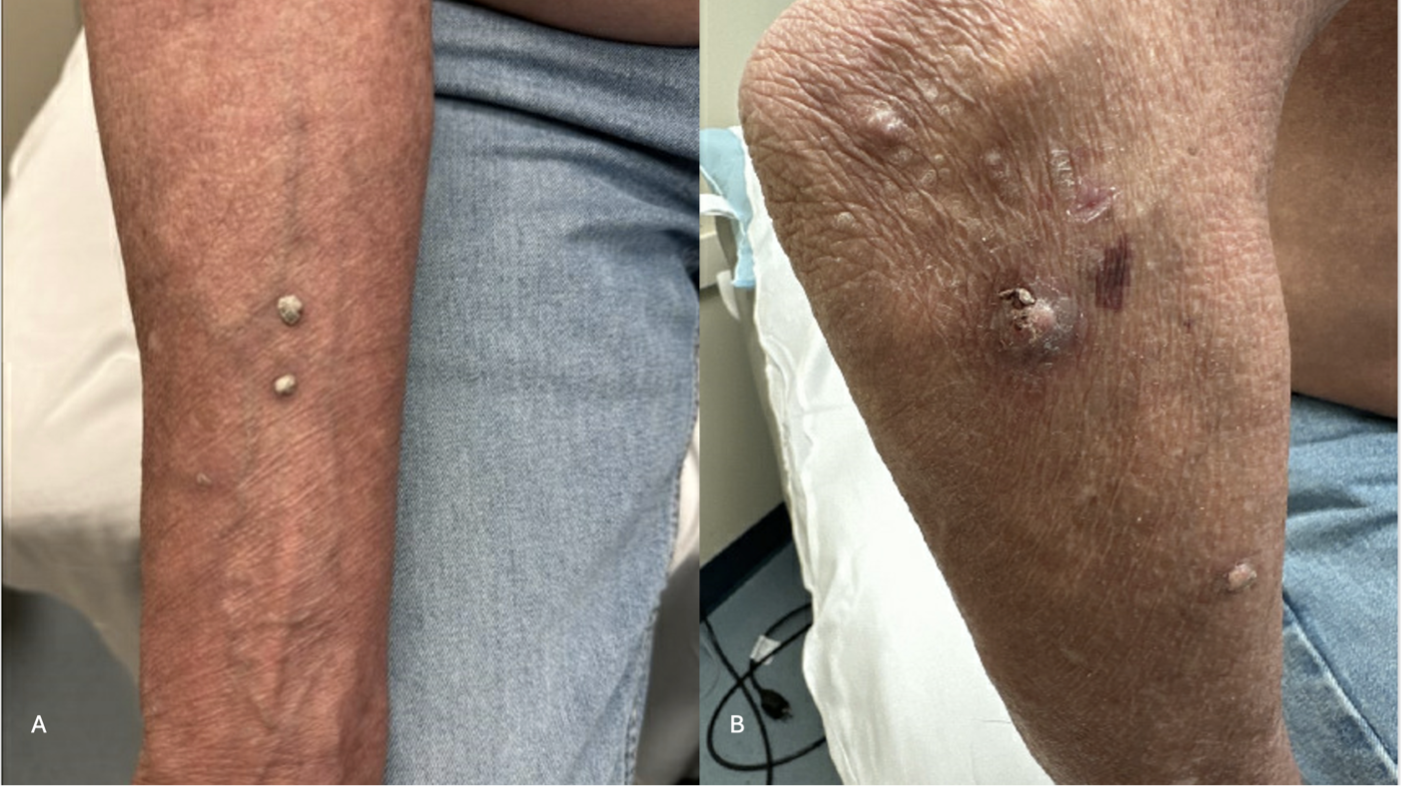
Figure 1A-B. (A) This image presents flesh to white-colored, firm verrucous papules of the right arm. (B) This image shows white-colored, firm verrucous papules on the patient’s legs and feet.
Diagnostic testing. A biopsy of a nodule on the right forearm revealed keratinocytes with ample glassy cytoplasm extending into the dermis, nuclear pleomorphism, and neoplastic keratinocytes extending to the resection margin (Figure 2A and B). These findings supported a diagnosis of squamous cell carcinoma of the keratoacanthomatous type.
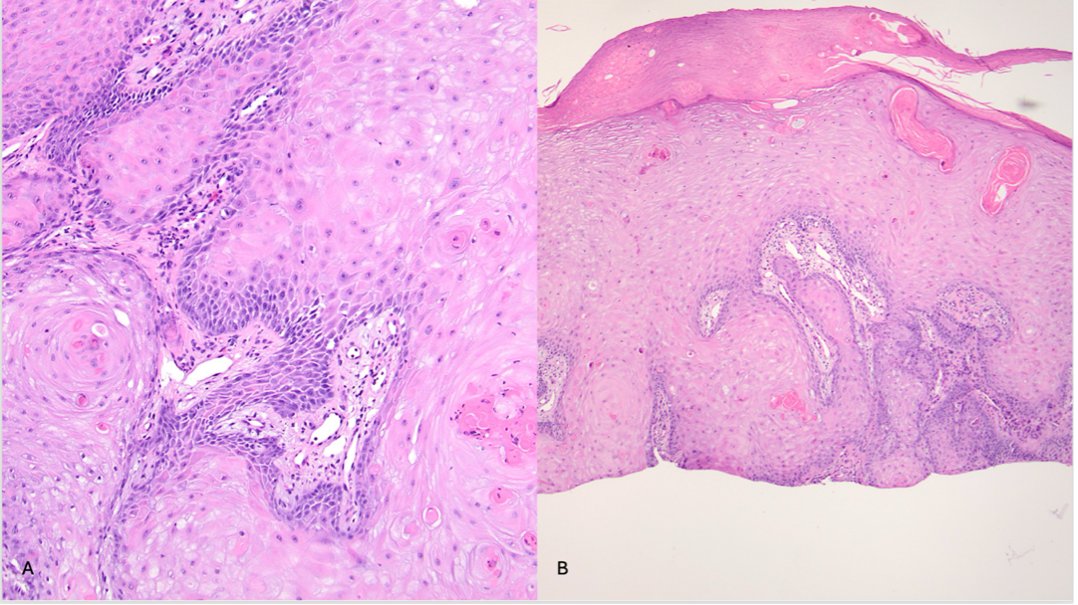
Figure 2A-B. The specimen reveals keratinocytes with ample glassy cytoplasm extending into the dermis. (A) Nuclear pleomorphism is encountered, with neoplastic keratinocytes extending to the margin of the resection. (B) The architecture favors a diagnosis of a squamous cell carcinoma of keratoacanthomatous type.
Based on the clinical course and temporal relationship with therapy, the final diagnosis was generalized eruptive keratoacanthomas (EKAs) secondary to enfortumab vedotin-ejfv and pembrolizumab.
Differential diagnoses. Given the clinical appearance and acute onset, the differential diagnoses included:
- Severe cutaneous drug reaction. This diagnosis was initially suspected due to the timing of systemic therapy, but biopsy findings favored keratoacanthomas.
- Squamous cell carcinoma (SCC). The patient’s biopsy supported keratoacanthomatous type SCC, though the eruptive and self-limited nature was more consistent with drug-induced EKAs.
- Other keratoacanthoma-predisposing conditions. We also considered other keratoacanthoma-predisposing conditions such as Muir-Torre syndrome, xeroderma pigmentosum, Ferguson-Smith syndrome, and Grzybowski syndrome. However, these were considered less likely given the absence of relevant history or features.
Treatment and management. During the patient’s 8-day hospitalization, anticancer therapy was withheld. The patient was treated with intravenous immunoglobulin (IVIG; 70 g IV every 24 hours for 4 doses), methylprednisolone (1,000 mg IV every 24 hours for 3 doses), triamcinolone ointment 0.1% (applied to affected skin three times daily for 3 days, then twice daily), and petrolatum as needed, and disease activity stabilized under this regimen. A management discussion was held involving the patient, the dermatologist, and the oncologist. Strategies considered included surgical removal, trialing cryotherapy, or intralesional corticosteroid (ILK) injections. Given the extensive number of lesions (approximately 30 or more), surgical excision was not feasible. Outside of continuing with the hospital treatment regimen, the patient opted for a “watchful waiting” approach to determine if the lesions would resolve after hospital discharge.
Outcome and follow-up. Two months following the initial eruption, the nodules spontaneously resolved without scarring. The patient was able to resume pembrolizumab therapy without recurrence of EKAs once the nodules resolved.
Discussion. Keratoacanthomas are squamoproliferative lesions with variable clinical presentations. While solitary lesions can be treated effectively with surgical excision or Mohs surgery, multiple keratoacanthomas may require alternative therapies such as topical 5-fluorouracil, cryotherapy, intralesional corticosteroids, or systemic agents. However, there are no consensus guidelines for the management of generalized EKAs.1
For patients with metastatic urothelial carcinoma of the bladder, platinum-based chemotherapy had long been the standard treatment. Recent head-to-head trials have demonstrated that the combined regimen of enfortumab vedotin-ejfv and pembrolizumab significantly improves progression-free and overall survival compared with chemotherapy.2 While this regimen offers a survival advantage, it has been associated with cutaneous adverse events, including severe skin reactions, maculopapular rash, pruritus, and alopecia.2
In this case, EKAs were developed following combination therapy with pembrolizumab and enfortumab vedotin-ejfv. Pembrolizumab, a PD-1 inhibitor, has been associated with keratoacanthomas in prior reports, likely due to immune-mediated dysregulation.3 Enfortumab vedotin-ejfv, a nectin-4–targeting antibody-drug conjugate, has also been associated with cutaneous eruptions, including toxic epidermal necrolysis, Stevens–Johnson syndrome, drug reaction with eosinophilia, and maculopapular rashes.4-6 In a retrospective review of 178 patients, 32.6% developed cutaneous eruptions of varying morphology, most commonly involving the distal extremities.6
This case highlights the importance of patient-centered management of dermatologic adverse events associated with cancer therapy. Although systemic and local treatments remain options, careful observation may be appropriate in select cases, as lesions can resolve spontaneously. In this patient, it is also possible that temporary discontinuation of anticancer therapy or the supportive treatments administered during hospitalization, including systemic corticosteroids and IVIG, contributed to stabilization and eventual resolution of the lesions. However, the relative contribution of these interventions versus the natural course of eruptive keratoacanthomas remains unclear. Early recognition and close monitoring are critical to prevent unnecessary treatment interruptions and to preserve patients’ quality of life during oncologic care.
Conclusion. Generalized eruptive keratoacanthomas are a rare cutaneous adverse event of enfortumab vedotin-ejfv and pembrolizumab therapy. Clinicians should be aware of this potential reaction and consider individualized, patient-centered approaches, including watchful waiting, when surgical options are not feasible.
AUTHORS
Kiera Murphy, BS1 • Rachel K. Anderson, BS2 • Alyssa N. Egland, CRNP2 • Thomas N. Helm, MD2
AFFILIATIONS
1Penn State College of Medicine, Hershey, PA
2Department of Dermatology, Penn State Milton S. Hershey Medical Center, Hershey, PA
CITATION
Murphy K, Anderson RK, Egland AN, Helm TN. Generalized eruptive keratoacanthomas arising in the setting of anti-cancer therapy: an illustrative case with spontaneous resolution. Consultant. Published online March 17, 2026. DOI: 10.25270/con.2026.03.000003
Received. September 24, 2025. Accepted. December 16, 2025.
DISCLOSURE
The authors report no relevant financial relationships.
ACKNOWLEDGMENTS
None.
CORRESPONDENCE
Kiera Murphy, BS, Penn State College of Medicine, 700 HMC Cres Road, Hershey, PA 17033 (Email: kmurphy9@pennstatehealth.psu.edu)
References
- Ambur A, Clark A, Nathoo R. An Updated Review of the Therapeutic Management of Keratoacanthomas. J Clin Aesthet Dermatol. 2022;15(11):30-36.
- Powles T, Valderrama BP, Gupta S, et al. Enfortumab Vedotin and Pembrolizumab in Untreated Advanced Urothelial Cancer. N Engl J Med. 2024;390(10):875-888. doi:10.1056/NEJMoa2312117
- Freites-Martinez A, Kwong BY, Rieger KE, Coit DG, Colevas AD, Lacouture ME. Eruptive Keratoacanthomas Associated with Pembrolizumab Therapy. JAMA Dermatol. 2017;153(7):694-697. doi:10.1001/jamadermatol.2017.0989
- Casale F, Roth G, Wanat K, Saab-Chalhoub M. Enfortumab Vedotin Drug Eruption: Cutaneous Adverse Events and Histopathologic Findings. Am J Dermatopathol. 2024;46(8):538-541. doi:10.1097/DAD.0000000000002750
- Wu S, Adamson AS. Cutaneous toxicity associated with enfortumab vedotin treatment of metastatic urothelial carcinoma. Dermatol Online J. 2019;25(2):13030/qt4j44w7w6. Published 2019 Feb 15.
- Malik R, Xiang DH, Riew GJ, et al. Characterization of adverse cutaneous effects in the setting of enfortumab vedotin for metastatic urothelial carcinoma: A retrospective review. J Am Acad Dermatol. 2024;91(4):753-755. doi: 10.1016/j.jaad.2024.06.037
©2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


