Peer Reviewed
Endovascular Stent Placement for Transplant-Related Arterio-Enteric Bleed: A Case Report
Introduction. A 52-year-old woman with a history of kidney and pancreas transplantation (17 years prior), end-stage renal disease on dialysis, type 1 diabetes mellitus, hypertension, and class 2 pulmonary hypertension presented to the emergency department (ED) for abdominal distention and pain following surgical repair of a right tibial fracture one week ago.
History. The patient had a kidney and pancreas transplantation 17 years prior, end-stage renal disease on dialysis, type 1 diabetes mellitus, hypertension, and class 2 pulmonary hypertension. She presented to the ED after sustaining an open right tibial fracture from a fall. She required high-flow nasal cannula support for hypoxia and underwent open reduction and internal fixation of her right leg. Postoperatively, she developed increasing abdominal distention and pain. After 1 week, her hemoglobin level dropped to 7.0 g/dL, and she experienced a large bloody bowel movement during dialysis. Physical examination revealed pale, dry skin, a 3+ systolic murmur, and moderate abdominal distention. After multiple episodes of hematochezia, she was transferred to the intensive care unit, received four units of packed red blood cells, and ultimately developed hemorrhagic shock requiring intubation, vasopressors, and the massive transfusion protocol.
Diagnostic/Laboratory Testing. A computed tomography angiogram (CTA) of the abdomen and pelvis performed after initiating the massive transfusion protocol showed brisk arterial bleeding in the jejunal loops and a 1.6 × 0.9 cm pseudoaneurysm near the pancreatic transplant anastomosis (Figure 1).
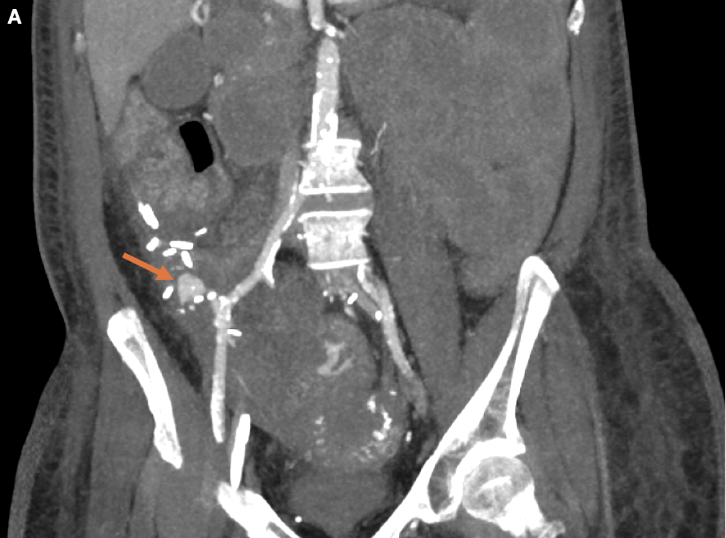
Figure 1. Thick multiplanar reformatted (MPR) coronal computed tomography (CT) in the arterial phase demonstrates a large pseudoaneurysm (arrow) arising from the pancreatic transplant arterial anastomosis.
The presence of an associated arterio-enteric fistula was confirmed with early arterial phase imaging, demonstrating contrast within the bowel lumen with associated pooling on delayed images (Figure 2). The patient’s lactate was elevated at 4.4 mmol/L, and her hemoglobin had decreased from a baseline of 9.4 to 7.0 g/dL.
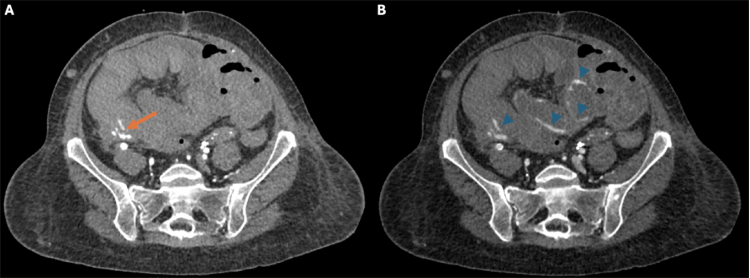
Figure 2. (A) Arterial phase axial image just inferior to the pseudoaneurysm shows active contrast extravasation (arrow) into a blood-filled right lower quadrant bowel loop. (B) Delayed image reveals pooling of contrast (arrowheads) as fresh blood surrounds hematoma filled loops of bowel.
Differential Diagnosis. In this patient with a history of pancreas and kidney transplantation who presented with severe hematochezia, an arterio-enteric fistula at the transplant anastomosis was the leading concern, supported by CT findings of active contrast extravasation into the bowel lumen. The pseudoaneurysm, also visualized on imaging, could have contributed to the development of the fistula by eroding into adjacent bowel loops, especially in the setting of local inflammation, infection, or compromised tissue integrity due to immunosuppression and prior surgical manipulation. Endoscopic evaluation excluded common causes of upper and lower gastrointestinal bleeding such as esophageal varices, peptic ulcers, and other mucosal lesions.
Treatment and Management. After recognizing the critical nature of the bleed soon after the CT scan, there was immediate face-to-face coordination with anesthesia and the critical care team. The patient initially required resuscitation in the CT suite; however, with timely cooperation, she was brought to the interventional radiology suite within 30 minutes of completion of the CT scan. Under general anesthesia, right common femoral artery access was obtained via ultrasound guidance, and angiography confirmed rapid active bleeding from the arterial conduit of the pancreas transplant (Figure 3).
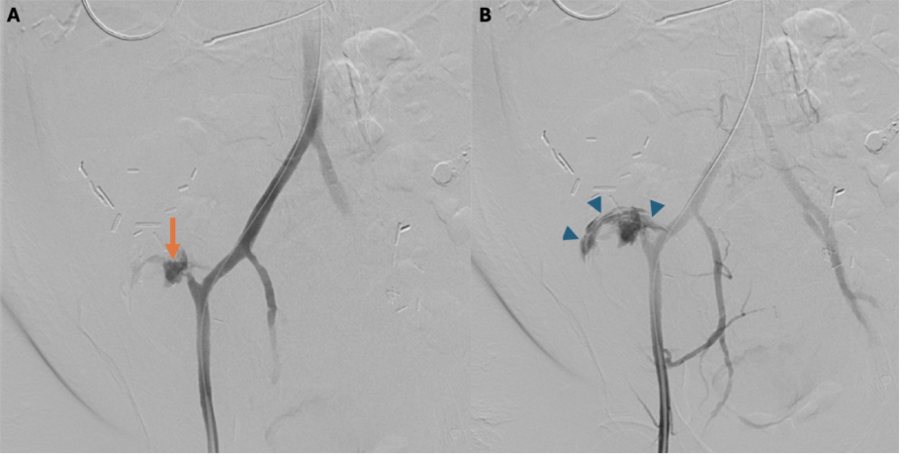
Figure 3. (A) Angiogram of the arterial conduit demonstrates the pseudoaneurysm (arrow). (B) A slightly delayed angiographic frame shows contrast extravasation (arrowheads) outlining the bowel lumen and confirming the presence of an arterio-enteric fistula.
A temporary balloon tamponade of the bleeding site was performed to stabilize the hemorrhage. An 8 mm × 4 cm Viabahn stent was then placed in the right external iliac artery. This effectively covered the arterial conduit and eliminated active bleeding on the final post-stent angiographic run (Figure 4). Closure of the right common femoral artery access was achieved with an 8 French AngioSeal device.
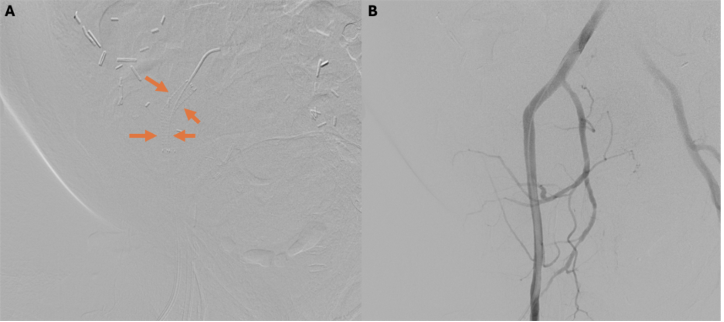
Figure 4. (A) Post-stenting angiogram of the right external iliac artery (arrow indicates stent). (B) Runoff image shows no residual contrast extravasation, confirming successful exclusion of the bleeding source.
Given the presence of an arterio-enteric fistula, the patient was continued on broad-spectrum antibiotics (e.g., vancomycin and cefepime). She ultimately received 12 units of packed red blood cells to maintain hemodynamic stability and resumed dialysis the following day.
Outcome and Follow-up. Following stent placement, the patient's bleeding resolved completely. Although she passed several melenic stools postprocedure as expected, she remained hemodynamically stable without requiring additional transfusions. CTA performed 1 week later showed no signs of active gastrointestinal (GI) bleeding, and both upper and lower endoscopies were unremarkable. She was discharged to a skilled nursing facility 10 days after the procedure.
At 1-week post-discharge, she returned to the ED with abdominal pain, hypotension, and altered mental status that progressed to obtundation, requiring intubation. Imaging revealed pneumatosis intestinalis concerning for bowel ischemia. Exploratory laparotomy demonstrated 70 cm of necrotic proximal small bowel, scattered areas of ischemia in other bowel segments, and a contained cecal perforation with retroperitoneal abscess. She underwent multiple re-explorations with initial discontinuity before eventual anastomosis. Surgical management was complicated by bleeding, which was managed conservatively. While initially requiring continuous renal replacement therapy (CRRT), she successfully transitioned back to intermittent hemodialysis.
Her hospital course was marked by progressive clinical deterioration. One month into readmission, she developed cholecystitis requiring percutaneous drainage, followed by respiratory failure necessitating reintubation. She was treated for Clostridioides difficile colitis and Klebsiella bacteremia secondary to pneumonia and ultimately required tracheostomy placement. Two months into the admission, she suffered cardiac arrest and was found to have an empyema. After extensive goals of care discussions with her family, supportive measures were withdrawn and the patient died.
Discussion. Vascular complications after transplantation occur in approximately 12–20% of patients and include aortic pseudoaneurysms, arteriovenous fistulas, vascular graft thrombosis, graft leaks, and venous outflow obstruction.1 Though many of these complications can be managed effectively, they are a cause of graft failure or rejection. Arterio-enteric fistulas, although rare, can be devastating in pancreatic transplant recipients, with only 28 cases documented in the past two decades.2 Pseudoaneurysms, which only occur in about 8% of pancreatic transplants, can also lead to significant risk of life-threatening hemorrhage.2
Possible causes in this case include pancreatic enzyme–related damage, local inflammation, and compromised vascular walls secondary to malpositioning or immunosuppression.3,4 Over time, a pseudoaneurysm adjacent to the pancreatic graft likely eroded into the bowel and precipitated a catastrophic bleed. Chronic immunosuppression and subclinical infections may have weakened the arterial wall, while the recent trauma and orthopedic surgery likely acted as a “second hit,” and unmasked/accelerating the subclinical pseudoaneurysm, and ultimately led to hemorrhage.
For arterio-enteric fistulas, antibiotic prophylaxis and regular imaging surveillance are essential, particularly in immunosuppressed transplant recipients who face increased infection risks and poor healing.5 In this case, despite successful initial hemostasis, the patient developed pneumatosis intestinalis, possibly from the combined effects of hemorrhagic shock, vasopressor therapy, immunosuppression, and altered vascular dynamics.
Efficient coordination of care and face-to-face communication among interventional radiology and anesthesia was particularly critical in this high-risk vascular emergency. Interventional radiology (IR) is integral to the management of acute GI bleeding when surgery is high-risk or when endoscopy is not feasible. Angiographic localization enables targeted endovascular treatment, such as transcatheter embolization or covered stent placement, using coils, plugs, balloons, or glue.6,7 This collaboration facilitated a timely intervention and prevented further deterioration.
Arterio-enteric fistulas are rare but potentially lethal complications in transplant recipients. This case demonstrates how endovascular stenting through interventional radiology can provide life-saving treatment while minimizing surgical trauma. Prompt intervention, facilitated by efficient interdisciplinary collaboration, was particularly important in managing this high-risk emergency. The patient's subsequent development of pneumatosis intestinalis, despite initial hemostatic success, suggests a need for vigilant post-procedure monitoring in immunosuppressed patients.
AUTHORS:
Jay Talati1 MD • Kevin Pierre2 MD • Gia Serrano3 • Chase Labiste2 MD • Jeffrey Vogel4 MD • Hugh Davis4 MD
AFFILIATIONS:
1University of Florida College of Medicine, Gainesville, FL
2University of Florida Department of Radiology, Gainesville, FL
3University of Florida College of Liberal Arts and Sciences, Gainesville, FL
4University of Florida Department of Radiology, Division of Vascular and Interventional Radiology, Gainesville, FL
CITATION:
Endovascular stent placement for transplant-related arterio-enteric bleed: A case report. Consultant. Published online November 1, 2025. doi:10.25270/con.2025.11.000002
Received March 19, 2025. Accepted June 16, 2025.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Jay Talati, MD, University of Florida College of Medicine, 1600 SW Archer Road, Gainesville, FL 32610 (jaytalati99@gmail.com)
References
- Hedegard W, Saad WEA, Davies MG. Management of vascular and nonvascular complications after renal transplantation. Tech Vasc Interv Radiol. 2009;12(4):240-262. doi:10.1053/j.tvir.2009.09.006.
- Leiberman D, et al. Radiological initial treatment of vascular catastrophes in pancreas transplantation: Review of current literature. Transplant Rev (Orlando). 2021;35(3):100624. doi: 10.1016/j.trre.2021.100624
- Srivastava A, et al. Vascular complication in live related renal transplant: An experience of 1945 cases. Indian J Urol. 2013;29(1):42-47. doi: 10.4103/0970-1591.109983
- Connolly JE, Kwaan JH, McCart PM, Brownell DA, Levine EF. Aortoenteric fistula. Ann Surg. 1981;194(4):402-412. doi: 10.1097/00000658-198110000-00004.
- Weise LB, et al. Iliac artery-enteric fistulas following failed pancreatic transplant. J Vasc Surg Cases Innov Tech. 2024;10(2):101427. doi: 10.1016/j.jvscit.2024.101427
- Villa M, et al. Arterio-enteric fistula in failed enteric-drained pancreas transplants: an impending danger. Int J Angiol. 2014;23(1):65-68. doi: 10.1055/s-0033-1349169
- David R, Trieu JA, Shokoohi S. GI hemorrhage from an arterio-enteric fistula from a failed pancreas allograft. ACG Case Rep J. 2022;9(7):e00822. doi: 10.14309/crj.0000000000000822
©2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


