Peer Reviewed
Disseminated Herpes Zoster: A “Can’t Miss” Diagnosis
Introduction. A 65-year-old woman presented to the dermatology clinic with ecchymotic lesions that had started 10 days prior in various areas, including the right lower back, right heel, central abdomen, and left forearm.
History. The patient had a 15-year history of tongue cancer with neck lymph node metastasis, along with follicular lymphoma in 2010 with progression in 2016 and 2022, non-small cell lung cancer (diagnosed in 2017) with metastases to the brain (diagnosed in 2021), and T-cell large granular lymphocyte leukemia (diagnosed in 2022).
At 1 month prior to developing lesions, she had completed a 3-month, weekly methotrexate and G-CSF regimen from which she developed mouth ulcers and saw little-to-no hematological improvement. In the preceding months, she was anemic due to her underlying leukemia, thrombocytopenic, and profoundly neutropenic. Her absolute neutrophil count in the month prior to presentation was 0.03-0.44 thousand/µL (normal range: 1.85-7.62 thousand/µL) with evidence of significant improvement post-methotrexate discontinuation. She was not actively taking any chemotherapy medications or immunosuppressive agents at the time of presentation to the clinic.
At the time of presentation, the patient had mild chronic fatigue due to her malignancy burden, but she was in no acute distress and had no systemic symptoms. Her physical examination demonstrated a 0.3 cm abdominal vesicle (Figure 1), 1.8 cm hemorrhagic plaque with minimal erythema on her lower back (Figure 2), and 1 cm violaceous plaque on her right medial foot (Figure 3). According to the patient the lesions were neither painful nor itchy, and they showed no signs of drainage.
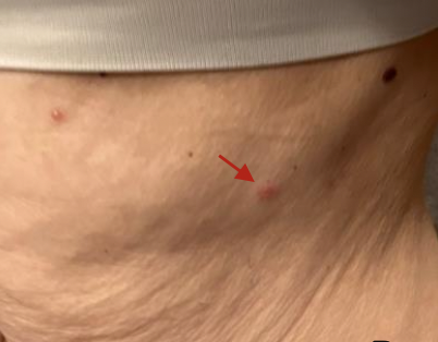
Figure 1. Left lateral abdomen is shown with 0.3 cm vesicle. An additional small vesicle can be seen to left of this lesion.
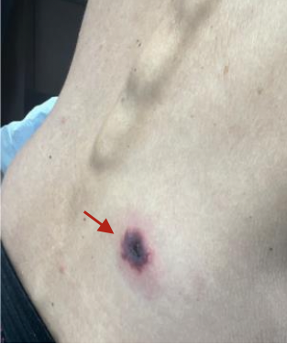
Figure 2. This image shows the patient’s back with 1.8 cm hemorrhagic plaque with central vesicles, along with scattered small vesicles visible on upper right and lower medial back.
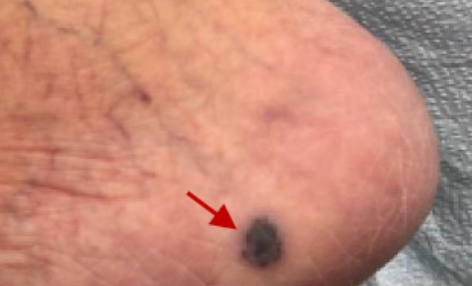
Figure 3. A right medial heel with 1 cm violaceous plaque is shown.
Diagnostic Testing. We performed punch biopsies, which showed ballooning degeneration, multinucleated keratinocytes, foci of basophilic eggshell chromatin at periphery of the nucleus, classic for viral keratinocyte infection (Figure 4). Varicella immunohistochemical stains were positive; herpes simplex virus 1 and 2 staining was negative. A viral culture was taken from the abdominal lesion, and a polymerase chain reaction test was performed, which detected varicella-zoster virus (VZV). Because VZV was detected in greater than two noncontiguous dermatomes, it was identified as disseminated herpes zoster.1
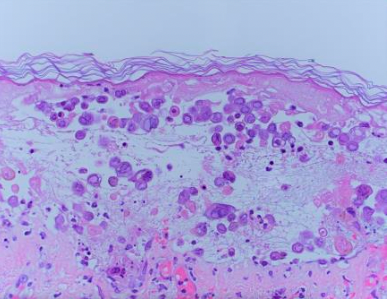
Figure 4. An hematoxylin and eosin stain from a punch biopsy showing classic viral change: ballooning degeneration, multinucleated keratinocytes, and foci of basophilic eggshell chromatin periphery of the nucleus.
Differential diagnoses. Our initial differential diagnoses included disseminated herpes zoster (DHZ), herpes zoster (HZ), or ecthyma. HZ was excluded as a possibility due to greater than two dermatomes involved. Ecthyma was considered due to the patient being immunocompromised and the presence of a vesicle. This potential diagnosis was ultimately excluded due to a lack of pus or drainage from the vesicles, which is expected with a bacterial infection such as ecthyma, as well as the supporting dermatopathological evidence.
Treatment and management. Although hospital admittance was an option, we decided to manage the patient as an outpatient with empirical treatment and close monitoring because she was presenting with few related symptoms and was otherwise in her normal state of health. We prescribed the patient 1000 mg oral valacyclovir three times daily for 10 days, and she was instructed to present to the emergency department (ED) if any signs of visceral dissemination arose, such as fever, chills, worsening skin lesions, or abdominal pain. We would follow-up with the patient in 2 weeks.
Outcome and follow up. At her 2-week follow-up, the patient’s lesions had largely resolved. She was then transitioned to valacyclovir 500 mg twice daily for suppressive therapy and given a referral to infectious disease, who recommended she remain on the same medication dosage and duration for 7 months for prevention.
The patient was again instructed to present to the ED if any signs of visceral dissemination arose, such as fever, chills, worsening skin lesions, or abdominal pain. At her next follow-up appointment 1 month later, the patient was clear of her previous lesions and had no new lesions. She was not undergoing chemotherapy at the time.
Discussion. DHZ occurs when there are more than 20 vesicles present in at least one dermatome outside the original, two or more contiguous dermatomes are involved, or there is visceral effect such as encephalitis, hepatitis, or pneumonitis.1
Advanced age, malignancy, immunosuppression, HIV, and/or chemotherapy are the greatest risk factors for more serious complications of HZ, including susceptibility to compounded bacterial infections, reactivation, post-herpetic neuralgia, scarring, encephalitis, central nervous system involvement, visceral involvement, and dissemination.2,3 Additionally, this patient population often presents with atypical cutaneous manifestations, specifically hemorrhagic lesions.4,5 Disseminated herpes zoster is one of the most common infections following hematopoietic cell transplantation. It is also seen in about 10% of immunocompromised patients with VZV. Visceral effects include VZV pneumonia, encephalitis, and hepatitis.6
Our patient was a 65-year-old immunocompromised woman with disseminated VZV who lacked the greater than 20 disseminated lesions and had no systemic symptoms. Because of the variability of clinical presentation in immunocompromised individuals, evaluation must be as timely and effective as possible, and treatment must correspond to the risk level of the presenting patient. Our case involves VZV infection and reactivation to DHZ in an immunocompromised patient with few related symptoms. We successfully treated the patient with corresponding non-surgical treatment due to her atypically mild presentation.
Although DHZ classically presents with widely disseminated, densely gathered lesions and a severe manifestation, our patient presented with only three scattered, hemorrhagic lesions that covered enough dermatomal area to be categorized as DHZ, but without serious symptoms or visceral involvement. Further, DHZ typically affects a singular dermatome then spreads, but our patient presented initially with multiple involved dermatomes.3,7 The concurrent presentation could have been simultaneous development or due to a delay in coming into the clinic. Regardless, it is imperative to note the challenge of diagnosing and treating atypical presentations of DHZ, especially in patients with tumor burden or those undergoing chemotherapy. Failure to recognize this atypical presentation could result in delayed diagnosis or mismanagement, which could be fatal.
DHZ can be clinically diagnosed by visualizing skin lesions and/or assessing systemic symptoms. Common differentials for disseminated bullae include varicella, erysipelas, impetigo, ecthyma, enteroviral infection, and herpes simplex virus infections. Viral isolation and cultures, pathology slides, laboratory results, serological testing, and a thorough history can be obtained to make the correct diagnosis.8 Classic histopathological findings of DHZ lesions are destruction of epithelial cells notable for “ballooning, multinucleated giant cells and eosinophilic intranuclear inclusions.”8
Hemorrhagic lesions, present in our patient, are atypical and classically indicate the progression of reactivated varicella to DHZ in the setting of immunosuppression.9 Purpuric HZ, not seen in our patient, can cause painful, raised erythematous, dermatomal lesions in immunocompromised patients or those undergoing antiplatelet and/or anticoagulative therapy and may concomitantly present with elevated erythrocyte sedimentation rate, thrombocytopenia, leukocytosis, or positive anti-varicella zoster virus IgG and IgM.10 Bullous lesions, gangrenous lesions, or an erythematous plaque may also occur in immunosuppressed individuals.2 When the diagnosis is particularly difficult to ascertain, immunohistochemistry and histological visualization of viral invasion of the follicular and sebaceous epithelium rather than the epidermis can point clinicians to atypical HZ. Polymerase chain reaction testing of any obtainable scabs, vesicles, or macular and/or papular lesions present initially during disease onset have higher sensitivity and specificity than standard culture or shell viral, which consists of centrifugation of the specimen, followed by incubation for 1 day to 4 days, then immunofluorescent staining.9
Increased duration and dose of immunosuppression, disease severity, longer course of disease process, lowered level of immune recovery, and increased time to immune recovery post-treatment are all predictors of unfavorable outcomes in patients who are immunocompromised.7 In studies involving vulnerable patient populations, such as immunocompromised patients and patients who had undergone bone marrow transplants, intravenous acyclovir for localized and DHZ was shown to stop progression of HZ, decrease the viral replication, and prevent dissemination.11 Intravenous acyclovir is typically recommended for VZV infection with suspected visceral dissemination or in immunocompromised patients such as patients who recently received allogeneic hematopoietic stem cell transplant, contracted moderate-to-severe acute chronic graft-versus-host disease, or have received aggressive anti-rejection therapy. Managing DHZ includes 10 mg/kg (or 500 mg/mg2) every 8 hours until infection is clinically considered controlled, at which point antiviral medication can be administered orally.12 The patient’s anticoagulative therapy may be continued during treatment for purpuric HZ.13
Further clinical anecdotes support that famciclovir and acyclovir are safe and effective against VZV in immunocompromised hosts, although research trials on the subject are limited. Famciclovir and valacyclovir may be preferred due to ease of administration and higher plasma concentrations. Failure to respond to these may indicate the need for foscarnet or cidofovir which may be less susceptible to a mutated viral DNA polymerase.1 Greater length of delay of treatment after onset of symptoms is the main known risk factor progression of VZV to DHZ among the immunosuppressed population, emphasizing the importance of beginning antiviral treatment as soon as possible after symptom onset.6 Therefore, patients who are virally asymptomatic, like ours, need evaluation as soon as possible to prevent further complications or mortality.
Conclusion. It is imperative to consider the various presentations of reactivated VZV when treating patients with immunodeficiency. Our patient confirms the significance of an atypical presentation of DHZ without systemic symptoms or extensive cutaneous involvement. Management guidelines call for immediate antiviral therapy, especially due to the high risk of death associated with HZ in immunocompromised patients and the minimal risk of antiviral treatment. This patient was treated empirically with oral antivirals for 10 days, closely monitored, and continued on prophylactic antiviral agents for 7 months because she did not show signs of systemic involvement and was otherwise in her normal state of health.
AUTHORS:
Aspen Trautz, MD¹, Sarah Ahmed, MD¹, Claire Rose Kissinger BS², Carla Shiller, MD¹, Mary Miller Braden MD¹
AFFILIATIONS:
¹Department of Dermatology, St. Luke’s University Health Network, Bethlehem, PA
²Lewis Katz School of Medicine at Temple University/St. Luke’s University Health Network, Bethlehem, PA
CITATION:
Trautz A, Ahmed S, Kissinger CR, Shiller C, Braden MM. Disseminated herpes zoster: a “can’t miss” diagnosis. Consultant. Published online August 27, 2025. DOI: 10.25270/con.2025.08.000002
Received: January 3, 2025 Accepted: July 3, 2025.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Claire Rose Kissinger, BS, Lewis Katz School of Medicine at Temple University/SLUHN, 340 E North St. Bethlehem PA, 18018 (Email: tup48401@temple.edu)
References
- Bollea-Garlatti ML, Bollea-Garlatti LA, Vacas AS, Torre AC, Kowalczuk AM, Galimberti RL, Ferreyro BL. Clinical characteristics and outcomes in a population with disseminated herpes zoster: a retrospective cohort study. Actas Dermosifiliogr. 2017;108(2):145-152. doi:10.1016/j.ad.2016.10.009
- Lewis DJ, Schlichte MJ, Dao H Jr. Atypical disseminated herpes zoster: management guidelines in immunocompromised patients. Cutis. 2017;100(5):321-330.
- Leung J, Harpaz R, Baughman AL, et al. Evaluation of laboratory methods for diagnosis of varicella. Clin Infect Dis. 2010;51(1):23-32. doi:10.1086/653112
- Nair PA, Patel BC. Herpes zoster. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023–. Updated April 3, 2023. Accessed August 25, 2025. https://www.ncbi.nlm.nih.gov/books/NBK441824/
- Cvjetković D, Jovanović J, Hrnjaković-Cvjetković I, Brkić S, Bogdanović M. Reaktivacija herpes zoster infekcije varicela-zoster virusom [Reactivation of herpes zoster infection by varicella-zoster virus]. Med Pregl. 1999;52(3-5):125-128.
- Yagi T, Karasuno T, Hasegawa T, et al. Acute abdomen without cutaneous signs of varicella zoster virus infection as a late complication of allogeneic bone marrow transplantation: importance of empiric therapy with acyclovir. Bone Marrow Transplant. 2000;25(9):1003-1005. doi:10.1038/sj.bmt.1702340
- Serota FT, Starr SE, Bryan CK, Koch PA, Plotkin SA, August CS. Acyclovir treatment of herpes zoster infections: use in children undergoing bone marrow transplant. JAMA. 1982;247(15):2132-2136.
- Gnann JW Jr. Varicella-zoster virus: atypical presentations and unusual complications. J Infect Dis. 2002;186(suppl 1):S91-S98. doi:10.1086/342963
- McAdam AJ, Riley AM. Developments in tissue culture detection of respiratory viruses. Clin Lab Med. 2009;29(4):623-634. doi:10.1016/j.cll.2009.07.009
- Veraldi S, Beretta AE, Spigariolo CB. Four cases of hemorrhagic herpes zoster associated with dabigatran therapy. Dermatol Ther. 2022;35(5):e15413. doi:10.1111/dth.15413
- Balfour HH, Bean B, Laskin OL, et al. Acyclovir halts progression of herpes zoster in immunocompromised patients. N Engl J Med. 1983;308(24):1448-1453. doi:10.1056/NEJM198306163082402
- Ljungman P, Lonnqvist B, Ringdén O, Skinhøj P, Gahrton G. A randomized trial of oral versus intravenous acyclovir for treatment of herpes zoster in bone marrow transplant recipients. Bone Marrow Transplant. 1989;4(6):613-615.
- Sowell J, Reynolds H, Sowell J. Hemorrhagic herpes zoster with contralateral multidermatomal distribution associated with rivaroxaban: an unusual presentation. JAAD Case Rep. 2021;9:31-33. doi:10.1016/j.jdcr.2020.12.038
©2025 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


