Peer Reviewed
Congenital Hemangioma: A Rare Vascular Tumor Fully Formed at Birth
Before reading further, take a moment to review the corresponding Photoclinic Challenge and test your clinical reasoning.
Introductory sentence. A male infant born at 40 weeks and 3 days' gestation was delivered via cesarean section to a primigravida mother because of nonprogressive labor.
History. The mother had initially presented for labor induction. Her prenatal course was notable for a resolved placenta previa. A second-trimester ultrasound demonstrated a posterior placenta near the cervical os, and a follow-up scan at 32 weeks confirmed resolution.
Non-invasive prenatal testing, including cell-free DNA screening for aneuploidy, was negative, and a Group B Streptococcus screening was also negative.
At birth, the infant had an uncomplicated postnatal transition with Apgar scores of 9 at both 1 and 5 minutes, requiring only routine newborn resuscitation. A physical examination demonstrated a 5 × 5 cm, well-circumscribed, violaceous lesion, with approximately 2 cm of elevation on the right posterior thigh, proximal to the popliteal fossa. The lesion displayed a central area of telangiectasia surrounded by a thin peripheral rim of pallor, which was warm to the touch and slightly compressible (Figure 1). The infant was otherwise hemodynamically stable and well-appearing.
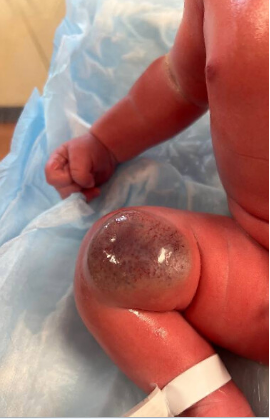
Figure 1. A 5 × 5 × 2 cm, well-circumscribed, violaceous lesion on the right posterior thigh, proximal to the popliteal fossa is shown. The lesion displayed a central area of telangiectasia surrounded by a thin peripheral rim of pallor, was warm to the touch, and slightly compressible.
Diagnostic testing. We used a grayscale ultrasound with multiple longitudinal and transverse real-time images with a color flow Doppler. The images demonstrated a soft-tissue mass within the subcutaneous soft tissues of the right medial distal thigh. The lesion was relatively isoechoic with mild heterogeneity and moderate vascularity. It measured 3.4 × 1.1 × 4.7 cm and demonstrated detectable venous and arterial waveforms without obvious extension beyond the subcutaneous tissue.
A complete blood count (Table 1) and coagulation studies (Table 2) were obtained. The institution’s D-dimer cutoff is 0.5 µg/mL, used to exclude deep vein thrombosis and pulmonary embolism. In neonates, D-dimer levels are physiologically higher than in adults and gradually decline with age.1 The infant’s partial thromboplastin time (PTT) was within the normal range. At the same time, prothrombin time (PT) and international normalized ratio (INR) were slightly elevated, which is consistent with the known characteristics of neonatal hemostasis as well as reduced levels of certain coagulation factors balanced by increased hematocrit, mean corpuscular volume, and elevated von Willebrand factor levels.2-3
Table 1. Complete Blood Count and Differential
|
Test |
Patient value |
Reference range* |
|
WBC x 103, µL |
16.78 |
8:04-15.40 |
|
RBC x 105, µL |
5.18 |
4.10-5.55 |
|
Hemoglobin, g/dL |
18.4 |
13.9-19.1 |
|
Hematocrit, % |
51.1 |
39.8-53.6 |
|
MCV, fL |
98.6 |
91.3-103.1 |
|
MCH, pg |
35.5 |
31.3-35.6 |
|
MCHC, g/dL |
36.0 |
33.0-35.7 |
|
RDW, % |
17.4 |
14.8-17.0 |
|
Platelet count x 103 |
219 |
218-419 |
|
Neutrophils, % |
62.8 |
|
|
Bands, % |
0.0 |
|
|
Lymphocytes, % |
28.1 |
|
|
Monocytes, % |
7.4 |
|
|
Eosinophils, % |
1.7 |
|
|
Basophils, % |
0.0 |
|
|
Segs + Bands x 103 |
10.54 |
1.60-6.06 |
|
Lymphocytes x 103 |
4.72 |
2.09-7.53 |
|
Monocytes x 103 |
1.24 |
0.52-1.17 |
|
Eosinophils x 103 |
0.29 |
0.12-0.66 |
|
Basophils x 103 |
0.00 |
0.02-0.11 |
*Reference ranges are from the institutional laboratory
Table 2. Coagulation Studies
|
Test |
Patient value |
Reference range* |
|
PT sec |
20.7 |
|
|
PTT sec |
24.2 |
24.0-34.3 |
|
INR |
1.8 |
|
|
D-dimer µg/ml |
1.35 |
< 0.50 |
*Reference ranges are from the institutional laboratory. No reference range was specified for PT or INR. Results may be affected by the patient’s elevated hematocrit.1-3
Abbreviations: WBC, white blood cell; RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; RDW, red cell distribution width; SEG, segmented neutrophil.
Imaging plays a critical role in the evaluation, providing information on lesion depth, vascular density and flow velocity, presence of arteriovenous shunts, and any calcifications.4-5
In this case, the grayscale and color Doppler ultrasound demonstrated typical features of congenital hemangioma noted on imaging: confinement to the subcutaneous fat, heterogeneous echotexture, moderate vascularity, and evidence of arterial-venous shunting.6 Although not routinely required, one can employ a magnetic resonance imaging (MRI) to characterize congenital hemangioma, particularly in cases where one is considering surgical excision.4
Differential diagnosis. When evaluating a suspected congenital hemangioma, the differential diagnosis includes other vascular tumors, vascular malformations, and soft tissue masses. Accurate distinction is essential, as these entities differ significantly in natural history, imaging features, and management strategies.4-10
A frequent diagnostic challenge is differentiating congenital hemangioma from infantile hemangioma. While infantile hemangioma may be present at birth or appear within the first few weeks of life, it follows a distinct growth pattern with proliferative, plateau, and involution phases.11-16 In contrast, congenital hemangioma is fully developed at birth and does not undergo postnatal proliferation.5 Immunohistochemical staining for glucose transporter-1 (GLUT-1) can aid diagnosis; it is consistently positive in infantile hemangioma and negative in congenital hemangioma .14
Tufted hemangioma is another rare vascular tumor that can be present at birth or in early infancy. It typically appears as a firm, red-to-violaceous nodule or plaque involving the skin and subcutaneous tissue, often associated with hypertrichosis.17 In contrast, congenital hemangioma presents as a soft, well-circumscribed mass. Kaposiform hemangioendothelioma (KHE) is a rare, locally aggressive vascular tumor that may present at birth as an ill-defined subcutaneous mass with overlying purpura or bruising. KHE is strongly associated with Kasabach-Merritt phenomenon (KMP), a consumptive coagulopathy characterized by profound thrombocytopenia.17-18 Our patient showed no evidence of thrombocytopenia or coagulopathy.
Pyogenic granuloma (lobular capillary hemangioma) is a benign capillary vascular tumor that typically arises on the skin or mucous membranes. While it can occur in infancy, it is most commonly present after 4 months of age and is particularly frequent in the second and third decades of life. Clinically, it often appears as a small, rapidly growing red papule with a friable surface. Following a proliferative phase lasting weeks to months, the lesion usually stabilizes. Histopathologic examination reveals lobulated capillary proliferation with a mixed inflammatory infiltrate, often including neutrophils.13-14,19 On immunohistochemistry, pyogenic granulomas are negative for GLUT1, a feature that distinguishes them from infantile hemangiomas.14 Clinical presentation and morphologic features further aid in distinguishing a pyogenic granuloma from a congenital hemangioma.19
Vascular malformations are a crucial differential consideration. Unlike congenital hemangiomas, which are fully formed vascular tumors at birth, vascular malformations are structural anomalies within vessel formations.6-7,20 They grow proportionally with the child and do not undergo spontaneous involution. On Doppler ultrasonography, venous, capillary, and lymphatic malformations typically demonstrate slow-flow characteristics, whereas arteriovenous malformations or fistulas present as fast-flow lesions.4-5 Histologically, venous and combined vascular malformations are negative for Wilms tumor 1 (WT-1), which contrasts with CHs since they generally stain positively for this marker.6
Treatment and management. Congenital hemangiomas are classified into 3 subtypes: rapidly involuting (RICH), non-involuting (NICH), and partially involuting (PICH). In our case, the lesion demonstrated early changes within 24 hours of birth, including increased peripheral pallor and decreased prominence of the central telangiectasia (Figure 2), consistent with the early involutional phase of RICH.5-9
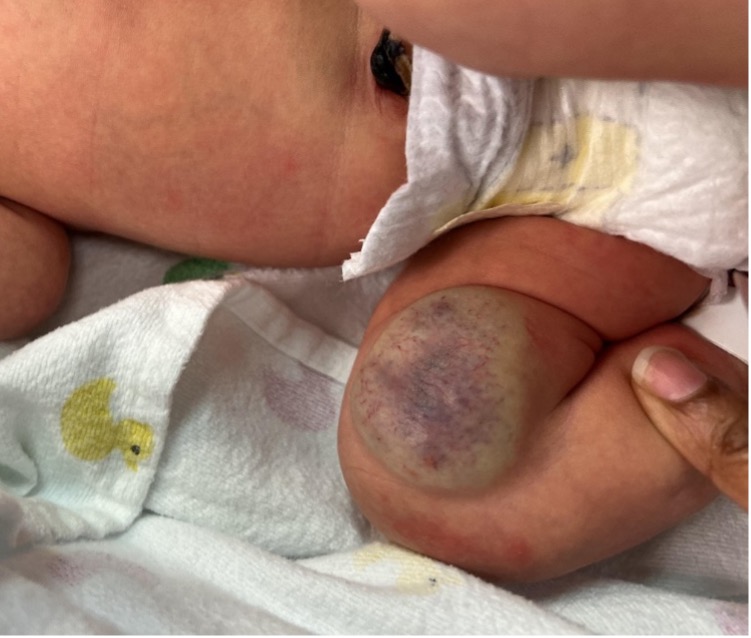
Figure 2. A clinical photograph of the infant’s lesion at 24 hours of life is shown. The lesion appeared smaller than at birth (5 × 4 × 1 cm), with reduced central telangiectasia and more prominent peripheral pallor. The mass remained soft and well circumscribed.
Management of the RICH phase is typically conservative, provided the infant is hemodynamically stable, and the lesion does not impair function or cause high-output cardiac failure.5-9
Outcome and follow-up. Our patient remained clinically well, and no intervention was required. The infant was followed by dermatology and has shown progressive spontaneous involution of the lesion without complications (Figure 3).
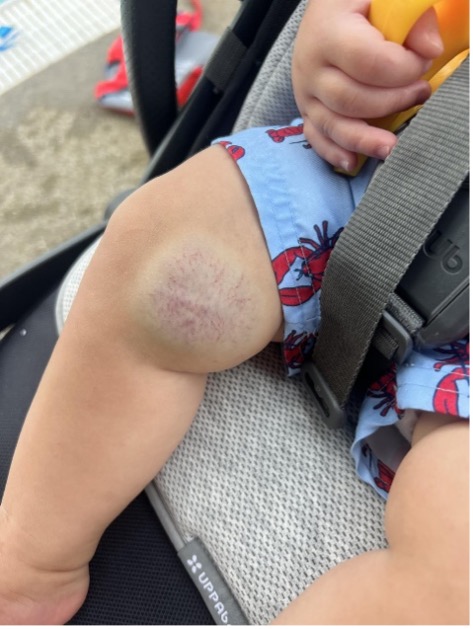
Figure 3. The RICH congenital hemangioma at 8 months of age, showing fading telangiectasia with decreased size of the lesion and blending with the surrounding skin.
RICH congenital hemangiomas usually regress completely by 12 to 14 months of age.5-6 Although ulceration is less common in congenital hemangioma than in infantile hemangioma, it can occur, particularly in areas subject to friction or trauma. Preventive measures, such as applying petroleum jelly, are recommended to maintain skin integrity.6,7 Close follow-up in the early weeks of life is advised to monitor for involution or complications, with visits spaced further apart as regression progresses.
Intervention is reserved for complications such as ulceration, bleeding, pain, or lesions in trauma-prone or functionally significant locations. Surgical excision, sometimes preceded by embolization, is an effective treatment.5-7 Pulsed dye laser and sclerotherapy may improve residual telangiectasia, and surgical removal of atrophic skin following involution may be considered for cosmetic reasons.5-7
Partially involuting and non-involuting congenital hemangioma can also be conservatively managed in asymptomatic patients. Importantly, pharmacologic therapies, such as beta-blockers (eg, propranolol) and corticosteroids, are ineffective in CH and are not recommended.5-8
Discussion Congenital hemangiomas are rare, benign vascular tumors that are fully developed at birth, with their true incidence remaining unknown. Recent studies have demonstrated somatic activating mutations in the GNAQ and GNA11 genes across all congenital hemangioma subtypes, suggesting that additional modifying factors influence differences in postnatal behavior.21 RICH congenital hemangiomas may occasionally be detected by a prenatal ultrasound, but they are most often diagnosed at birth. The earliest diagnosis was made at 12 weeks of gestation.4-6 Besides gestational age, fetal position, location, size, and the sonographer’s skill level determine the prenatal detection. Lesions typically occur on the head, neck, and lower extremities, usually as solitary masses measuring from a few centimeters to over 10 cm in size.4-6 Multifocal congenital hemangioma are exceedingly rare. Although uncommon, visceral involvement, such as hepatic RICH congenital hemangiomas, has been reported. Hepatic lesions are usually asymptomatic, but, when symptomatic, they may manifest with anemia, transient thrombocytopenia, or hypofibrinogenemia due to the tumor’s vascular nature.10 Unlike other vascular tumors such as KHE, RICH congenital hemangiomas are not typically associated with KMP.16,17 The natural history of RICH congenital hemangiomas is characterized by rapid involution, with up to 80% reduction in size reported within the first 2 years of life.6-8
Infantile hemangiomas are the most common benign vascular tumors of infancy, affecting approximately 4% to 5% of infants, with a higher prevalence in preterm infants, females, and those of Caucasian descent. These tumors are typically absent at birth or present as a subtle patch and enter a rapid proliferative phase during the first few weeks to months of life.11-17
Recognizing this difference is crucial for accurate diagnosis and effective management. Management of congenital hemangioma depends on several factors, including the lesion size, location, clinical subtype, and the presence of complications, such as thrombocytopenia or high-output cardiac failure. Accurate diagnosis of congenital hemangioma versus infantile hemangioma is crucial, as congenital hemangioma does not respond to beta-blocker therapy, and its natural history and treatment considerations differ. Congenital hemangiomas have distinct histopathologic features and immunohistochemical profiles that can aid in diagnosis when the clinical presentation and imaging findings are inconclusive.5-7,15-16
AUTHOR:
Anuja Sriparameswaran, MD, FAAP, IBCLC, MPH1
AFFILIATIONS:
1Nationwide Children’s Hospital, Department of Hospital Pediatrics, 700 Children’s Drive, Columbus, OH 4320
CITATION:
Sriparameswaran A. Congenital hemangioma: a rare vascular tumor fully formed at birth. Consultant. Published online January 6, 2026; DOI:10.25270/con.2026.01.000004
Received: September 2, 2025. Accepted: October 29, 2025
DISCLOSURES:
The authors report no relevant financial relationships. Informed consent from the infant’s family was obtained for all the images.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Anuja Sriparameswaran, MD, FAAP, IBCLC, MPH. Nationwide Children’s Hospital. 5475 Harlem Road, New Albany, OH, 43054 (email: Anuja.Sriparameswaran@osumc.edu)
Reference
- Khalilov Zİ, Ünsal A, Altuntaş N. The D-dimer reference intervals in healthy term newborns. Transfus Apher Sci. 2022;61(6):103493. doi:10.1016/j.transci.2022.103493
- Davenport P, Sola-Visner M. Hemostatic challenges in neonates. Front Pediatr. 2021;9:627715. doi:10.3389/fped.2021.627715
- Gorio C, Molinari AC, Martini T, Ferretti A, Albrici G, Carracchia G, Ierardi A, Leotta M, Portesi N, Sacco M, et al. Hemostasis Laboratory Diagnostics in Newborns. Journal of Clinical Medicine. 2025; 14(14):5068. https://doi.org/10.3390/jcm14145068
- Dubois J, Patriquin HB, Garel L, Powell J, Filiatrault D, David M, Grignon A. Soft-tissue hemangiomas in infants and children: diagnosis using Doppler sonography. AJR Am J Roentgenol. 1998;171(1):247-252 (US)
- Boon LM, Enjolras O, Mulliken JB. Congenital hemangioma: accelerated involution evidence of. J Pediatr. 1996;128(3):329-335. (US histopathology)
- Frieden IJ, Adams DM. Congenital hemangiomas: Rapidly involuting congenital hemangioma (RICH), noninvoluting congenital hemangioma (NICH), and partially involuting congenital hemangioma (PICH). In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated July 18, 2025. Accessed August 21, 2025. https://www.uptodate.com
- Enjolras O, Mulliken JB, Boon LM, Wassef M, Kozakewich HP, Burrows PE. Noninvoluting congenital hemangioma: a rare cutaneous vascular anomaly. Plast Reconstr Surg. 2001;107(7):1647-1654. doi:10.1097/00006534-200106000-00002
- Nasseri E, Piram M, McCuaig CC, Kokta V, Dubois J, Powell J. Partially involuting congenital hemangiomas: a report of 8 cases and review of the literature. J Am Acad Dermatol. 2014;70(1):75-79 DOI: 10.1016/j.jaad.2013.09.018
- Funk T, Lim Y, Kulungowski AM, et al. Symptomatic congenital hemangioma and congenital hemangiomatosis associated with a somatic activating mutation in GNA11. JAMA Dermatol. 2016;152(9):1017-1021. doi:10.1001/jamadermatol.2016.2365
- Lewis D, Vaidya R. Congenital and Infantile Hepatic Hemangioma. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. Updated June 12, 2023. Accessed August 24, 2025 https://www.ncbi.nlm.nih.gov/books/NBK518988/
- Metry DW. Infantile hemangiomas: Epidemiology, pathogenesis, clinical features, and complications. In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated November 25, 2024. Accessed August 21, 2025. https://www.uptodate.com
- Metry DW. Infantile hemangiomas: Management. In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated December 17, 2024. Accessed August 21, 2025. https://www.uptodate.com
- Léauté-Labrèze C, Harper JI, Hoege PH. Infantile haemangioma. Lancet. 2017;390(10089):85-94. doi:10.1016/S0140-6736(16)00645-0 (pyogenic granuloma)
- North PE, Waner M, Mizeracki A, Mihm MC Jr. GLUT1: a newly discovered immunohistochemical marker for juvenile hemangiomas. Hum Pathol. 2000;31(1):11-22.
- Martinez-Perez D, Fein NA, Boon LM, Mulliken JB. Not all hemangiomas look like strawberries: uncommon presentations of the most common tumor of infancy. Pediatr Dermatol. 1995;12(1):1-6
- Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170(3):e12804. doi:10.1111/bjd.12804
- Adams DM, Frieden IJ. Tufted angioma, kaposiform hemangioendothelioma (KHE), and Kasabach-Merritt phenomenon (KMP). In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated February 28, 2025. Accessed August 21, 2025. https://www.uptodate.com
- Lyons LL, North PE, Lai FM-M, Stoler MH, Folpe AL, Weiss SW. Kaposiform hemangioendothelioma: a study of 33 cases emphasizing its pathologic, immunophenotypic, and biologic uniqueness from juvenile hemangioma. Am J Surg Pathol. 2004;28(5):559-568. doi:10.1097/00000478-200405000-00001
- Lawley LP. Pyogenic granuloma (lobular capillary hemangioma). In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated May 29, 2024. Accessed August 21, 2025. https://www.uptodate.com
- Boon LM, Vikkula M, Seront E. Venous malformations. In: Levy ML, Corona R, eds. UpToDate. Waltham, MA: Wolters Kluwer. Updated July 12, 2024. Accessed August 21, 2025. https://www.uptodate.com
- Ayturk UM, Couto JA, Hann S, et al. Somatic activating mutations in GNAQ and GNA11 are associated with congenital hemangioma. Am J Hum Genet. 2016;98(5):789-795. doi:10.1016/j.ajhg.2016.03.009
©2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


