Peer Reviewed
Atrophoderma of Pasini and Pierini: A Therapeutic Challenge
Introduction. A 14-year-old girl presented to the dermatology clinic with hyperpigmented, depressed lesions on her bilateral upper extremities and trunk that had been present for 4 years, with more recent darkening.
History. The patient reported that her lesions were asymptomatic and had never been hard, scaly, or erythematous. She noted darkening of the lesions on her trunk. She denied recent use of new medications or personal care products and reported no household contacts with similar findings. She had no known drug allergies; she took no medications and had a history of seasonal allergies. On examination, our patient presented with hyperpigmented lesions with well defined “cliff-drop” borders, meaning the lesions varied in diameter yet all had an abrupt transition between the normal and affected skin at the entirety of their edges, ranging from 2-8mm in depth. The lesions were distributed bilaterally on her trunk and upper extremities.1
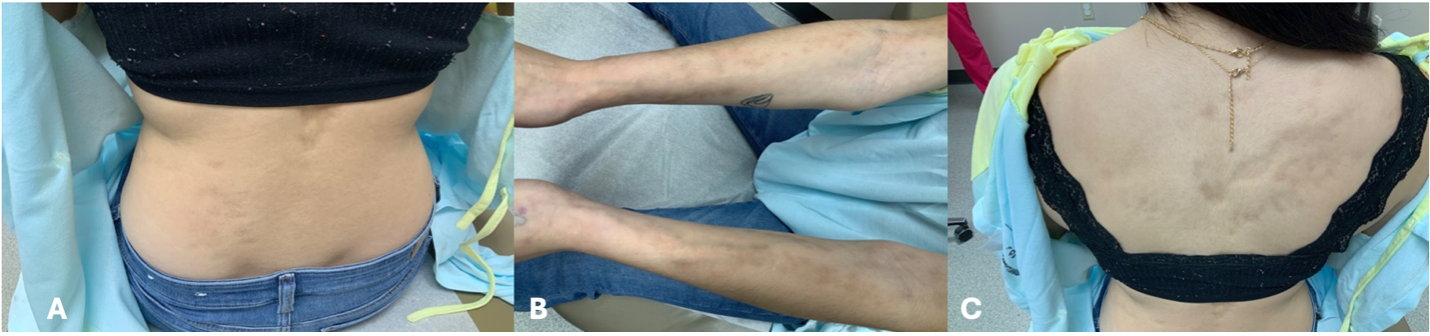 Figure 1A-C. Lesion presentation of suspected atrophoderma of Pasini and Pierini is shown.
Figure 1A-C. Lesion presentation of suspected atrophoderma of Pasini and Pierini is shown.
Diagnostic examination. We diagnosed the patient with Atrophoderma of Pasini and Pierini (APP), based on the patient’s history and our clinical examination. APP is a cutaneous condition characterized by dermal atrophy resulting from dermal collagen disorganization1. Although an infrequent finding, APP commonly presents in adolescence or early adulthood, though cases have also been reported in older adults, with a documented female-to-male ratio of 6:1.3 We did not perform any additional diagnostic testing at the time of the initial presentation given the observational findings are pathognomonic to APP.
Differential diagnosis. The differential diagnosis included several diagnoses with similar cutaneous dermatologic presentations, which were ultimately ruled out based on specific features of the patient’s physical presentation (Table 1). Anetoderma was considered due to the lesions' atrophic nature and pigment changes.9 However, the sharply demarcated cliff-drop borders made this diagnosis less likely, as anetoderma typically presents with localized areas of flaccid skin. In addition, Atrophoderma of Moulin (LAM), which presents with similar atrophic band-like lesions, was also considered.6 However, LAM presents with lesions following lines of Blaschko unilaterally, and our patient had a bilateral distribution of atrophic lesions.6 Furthermore, a diagnosis of lupus panniculitis was ruled out due to the lack of painful subcutaneous nodules. While nodules can lead to atrophy or scarring, our patient’s lesions were too diffuse to be explained by a single (or several) focal nodules, and the lack of active nodules made this diagnosis less likely.10 Lastly, linear localized morphea was explored, as this diagnosis can present, with hyper- and hypopigmented lesions distributed similarly. However, we ruled out this diagnosis because there was no dermal thickening or sclerosis, which are characteristic of linear localized morphea.
Table 1. Distinguishing APP from similar skin disorders
|
Diagnosis |
Morphology |
Color |
Distribution |
|
APP1,5 |
Sharply demarcated non-indurated, depressed plaques |
Hyperpigmented, brown, gray, violaceous |
Trunk, upper, and lower extremities |
|
Anetoderma1 |
Well-circumscribed, atrophic, flaccid, wrinkled/herniated plaques or nodules |
Flesh-colored, gray, white, brown, blue |
Trunk and proximal extremities |
|
LAM6 |
Linear patches or plaques |
Hyperpigmented |
Blaschko lines |
|
Lupus panniculitis8 |
Nodules, plaques |
Erythematous, violaceous, skin-toned |
Adipose dense body areas |
|
Linear localized morphea7 |
Band-like indurated plaques |
Hyper/hypo pigmented, erythematous |
Head, neck, upper and lower extremities |
Treatment and management. Although there is no standardized treatment for APP, some cases have been shown to improve with doxycycline3. However, we elected to start the patient on calcipotriene 0.005% topical cream to be applied once daily to the affected areas; the patient preferred to trial topical options and avoid oral medications at the time. The plan included consideration of doxycycline, hydroxychloroquine, or methotrexate if no improvement was observed. Additional case reports and literature support the use of doxycycline 200 mg daily to prevent the appearance of new lesions, as well as evidence supporting the use of topical corticosteroids and hydroxychloroquine in preventing disease progression1. Although some studies have detected Borrelia burgdorferi in patients with APP, the evidence for a causal relationship remains limited and inconclusive. Further research is needed to clarify this potential association before confidently using tetracyclines as a treatment approach.1
Outcome and follow-up. We subsequently lost the patient to follow-up. Three years later, she returned to the clinic with the same chief complaint. She reported little-to-no improvement with the use of calcipotriene 0.005% topical cream, as originally prescribed.
Upon re-examination, the hyperpigmented lesions with cliff-drop borders appeared unchanged in shape, size, and distribution. There had been no progression or onset of new lesions. We did not obtain laboratory testing after her initial visit, and we did not order any new laboratory tests. Although our patient had a biopsy confirmation of APP at another clinic, those results were not available for this report. Since the patient’s condition was stable, and further biopsy would not change the treatment plan agreed on, we did not perform an additional biopsy. Often, an initial biopsy of a suspected APP lesion is performed to rule out similar conditions that may be more progressive and require aggressive treatment.
Follow-up for the patient was as needed, as her lesions were stable. The overall prognosis for her APP indicates that lesions often stabilize over time, with spontaneous resolution reported infrequently. Additionally, the patient did not desire to pursue further treatment options at the time that had been initially discussed including doxycycline, hydroxychloroquine, and methotrexate for which there is no standardized dosing in this diagnosis.
Discussion: Atrophoderma of Pasini and Pierini is a rare condition characterized by sharply demarcated lesions with classic cliff-drop borders. Despite these characteristic features, APP remains under-recognized and often causes diagnostic confusion among clinicians and pathologists due to its unclear etiology and variable histopathologic findings.
In our patient’s case, the lesions remained stable over 3 years with no progression or improvement, even with a trial of topical calcipotriene. This outcome aligns with previously documented cases, indicating that APP often demonstrates a prolonged course with eventual stabilization but rarely undergoes spontaneous resolution or significant response to specific therapies.
The etiology of APP remains poorly understood. Histological findings documented in prior reports show flattening of the rete ridges, fragmentation of collagen fibers,4 and dermal thinning.6 Notably, Buechner and Rufi conducted a study evaluating a possible trigger of APP, examining 26 patients with APP for Borrelia burgdorferi antibodies. They found that 53% had positive IgG titers (compared to the 14% of the control group), suggesting a potential association.5 Although causality cannot be inferred from this single study, this finding supports further investigation.
A variety of treatment modalities have been trialed with inconsistent results. Topical corticosteroids, such as those used in our case, have shown limited effectiveness and do not consistently alter disease progression.5 In patients with a suspected B. burgdorferi involvement, oral doxycycline at the dose 200mg per day has shown some promise in preventing the appearance of new lesions, although results remain inconclusive.5 Other therapies, including hydroxychloroquine or Q-switched alexandrite laser treatments, have demonstrated potential benefit in select cases but lack large-scale evidence-based support.
Prognostically, APP tends to follow a slow, indolent course. Lesions often evolve over 10-20 years, then stabilize, with minimal risk of systemic involvement or serious health consequences. However, the cosmetic impact can be significant, and treatment responses are variable. Our case contributes to the growing body of literature by highlighting a stable disease course over 3 years with no progression, despite minimal therapeutic intervention.
Ultimately, APP remains a poorly understood condition with no standardized treatment guidelines. Further research is essential to clarify its pathogenesis, explore potential triggers, and identify effective management protocols.
AUTHORS:
Kisa M. Seerveld OMS-II1, Michelle Gallagher DO1
AFFILIATIONS:
1Michigan State University College of Osteopathic Medicine, East Lansing, MI
CITATION:
Seerveld KM, Gallagher M. Atrophoderma of Pasini and Pierini: A Therapeutic Challenge. Consultant. Published online: March 23, 2026. DOI: 10.25270/con.2026.03.000005
DISCLOSURES:
The authors report no financial disclosures.
ACKNOWLEDGMENTS:
None.
CORRESPONDENCE:
Kisa M. Seerveld OMS-II, Michigan State University College of Osteopathic Medicine 965 Wilson Rd, East Lansing, MI 48824 (Email: seerveld@msu.edu)
References
- Cook JC, Syed HA, Puckett Y. Anetoderma. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2025. Updated April 6, 2025. Accessed March 17, 2026. https://www.ncbi.nlm.nih.gov/books/NBK560605/
- Gambichler T, Zimmer S, Käsbauer S, Stücker M, Bechara FG. Disseminierte Atrophodermia Pasini et Pierini in blaschkoidem Erscheinungsbild (disseminated atrophoderma of Pasini and Pierini with blaschkoid appearance). Dermatologie (Heidelb). Published online May 21, 2025. doi:10.1007/s00105-025-05503-2
- González-Morán A, López-Navarro N, de Argila D. Atrofodermia idiopática de Pasini y Pierini. Estudio de cuatro casos (idiopathic atrophoderma of Pasini and Pierini: study of 4 cases). Actas Dermosifiliogr. 2005;96(5):303-306. doi:10.1016/s0001-7310(05)75059-6
- Gupta M, Kaushik A, Roy K, Meena D. Idiopathic atrophoderma of Pasini and Pierini in a zosteriform pattern on face. Indian J Paediatr Dermatol. 2025;26(1):26. doi:10.4103/ijpd.ijpd_16_24
- Litaiem N, Idoudi S. Atrophoderma of Pasini and Pierini. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2025. Updated August 7, 2023. Accessed March 17, 2026. https://www.ncbi.nlm.nih.gov/books/NBK519069/
- Muntyanu A, Redpath M, Roshdy O, Jfri A. Idiopathic atrophoderma of Pasini and Pierini: case report and literature review. Clin Case Rep. 2019;7:258-263.
- Nanchaipruek Y, Thanomkitti K. Linear atrophoderma of Moulin: a case report. Thai J Dermatol. 2024;40(4):99-102. Accessed March 17, 2026. https://he02.tci-thaijo.org/index.php/TJD/article/view/269386
- Pathak R, Shah R, Agarwal A, et al. A clinical case of idiopathic atrophoderma of Pasini and Pierini with literature review. Case Rep Dermatol Med. 2025;2025:1-5. doi:10.1155/crdm/8886954
- Penmetsa GK, Sapra A. Morphea. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2025. Updated August 7, 2023. Accessed March 17, 2026. https://www.ncbi.nlm.nih.gov/books/NBK559010/
- Rangel LK, Femia AN, Vleugels RA. Clinical characteristics of lupus erythematosus panniculitis/profundus: a retrospective review of 61 patients. JAMA Dermatol. 2020;156(11):1264-1266. doi:10.1001/jamadermatol.2020.2797
- Tan SK, Tay YK. Linear atrophoderma of Moulin. JAAD Case Rep. 2016;2(1):10-12. doi:10.1016/j.jdcr.2015.10.005
©2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Consultant360 or HMP Global, their employees, and affiliates.


