Peer Reviewed
Acute Aortic Dissection in a Patient with Systemic Lupus Erythematosus on Corticosteroid Therapy
Introduction. A 29-year-old woman with systemic lupus erythematosus (SLE) and a history of hypertension presented to the emergency department with acute chest pain and lower extremity paresthesia.
History. The patient presented to the hospital with a complex medical history. She had been on long-term treatment with prednisone and hydroxychloroquine for her SLE, which included controlling systemic symptoms and managing lupus-related organ involvement. Additionally, she had a history of hypertension, necessitating ongoing management. Notably, she had previously experienced non-infective endocarditis, a complication often associated with SLE.
Upon presentation, the patient presented with several symptoms, including cough, lower extremity paresthesia, and sudden onset chest pain without radiation. She denied experiencing fever, fatigue, or joint pain at the time of presentation, though these hallmark SLE symptoms had been present intermittently in her history. She did not report any new rashes, including a butterfly rash (malar rash), which is pathognomonic for SLE.
The patient’s physical examination revealed hypertensive emergency with blood pressure measuring 185/120 mmHg. Her heart rate was elevated at 110 beats per minute, and the patient appeared distressed, clutching her chest. There was no pulse differential between the right and left upper extremities, but a new diastolic murmur was auscultated at the right upper sternal border, consistent with aortic regurgitation. No gallops were noted, and peripheral pulses were diminished in both lower extremities, raising concern for vascular compromise. The lungs were clear to auscultation, and no bruits were heard over the carotid or femoral arteries. These clinical findings, combined with the patient’s medical history, raised concerns regarding potential cardiovascular involvement, particularly given the increased risk of vascular complications in SLE patients. This comprehensive history provided valuable context for the subsequent diagnostic evaluation and treatment decisions.
Diagnostic Testing. Upon admission, several diagnostic tests were performed to evaluate the patient’s condition. Electrocardiogram (EKG) showed no evidence of myocardial infarction (MI). Chest X-ray revealed an enlarged upper mediastinum, and transthoracic echocardiography (TTE) confirmed mild aortic regurgitation and a small pericardial effusion.
An elevated erythrocyte sedimentation rate (ESR) of 60 mm/hour was also noted (normal level, > 20 mm/hour for women younger than 50 years of age), indicating underlying inflammation. Computed tomography angiography (CTA) confirmed the diagnosis of a Stanford Type A aortic dissection (AD), extending from the root of the aorta to the bilateral external iliac arteries. A transesophageal echocardiogram (TEE) was performed, which provided additional detail regarding the extent of the dissection and its effect on the aortic valve. Laboratory tests revealed a hemoglobin level of 7.5 g/dL (normal range, 12-16 g/dL in women, 13.5-17.5 g/dL in men), elevated C-reactive protein (CRP) of 31 mg/L (normal range, < 5 mg/L), and serological markers consistent with active SLE. These include positive anti-nuclear antibody, anti-double stranded DNA antibodies, and low complement levels C3–36 (normal range, 90-180 mg/dL), C4–13 (normal: 10-40 mg/dL). The combination of imaging studies and laboratory findings allowed for a comprehensive assessment of the patient’s condition, facilitating prompt diagnosis and initiation of appropriate treatment measures1.
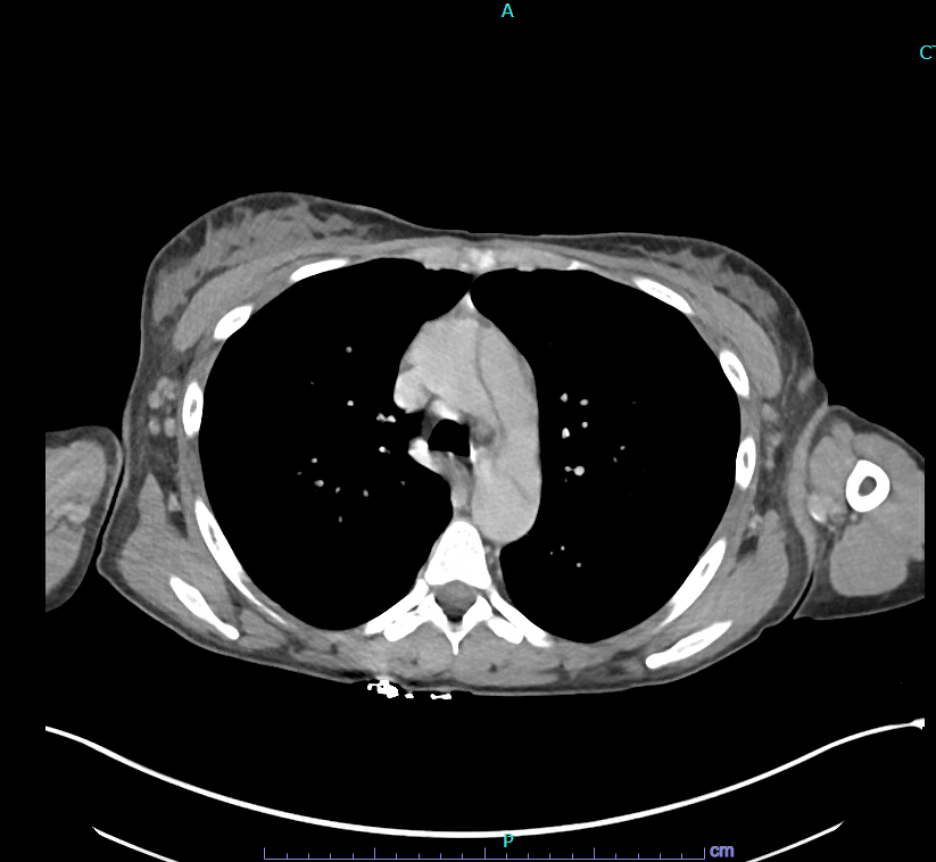
Figure 1. Computed tomography angiography (CTA) showing aortic dissection in a patient with systemic lupus erythematosus (SLE).
Differential diagnoses. In addition to AD, several differential diagnoses were considered given the patient’s presentation of acute chest pain and associated symptoms.
First, acute coronary syndrome (ACS), encompassing conditions such as MI and unstable angina, was a primary concern due to the abrupt onset of chest pain. Diagnostic evaluation typically involves an electrocardiogram (ECG) to detect characteristic ST-segment changes indicative of myocardial ischemia or infarction. Additionally, cardiac enzyme assays, such as troponin levels, aid in confirming myocardial damage. Accurate and prompt differentiation from ACS was crucial to avoid delays in the diagnosis and treatment of AD1,2.
Second, the possibility of pulmonary embolism (PE) was considered, particularly given the patient’s symptoms of chest pain and cough, as well as her history of systemic lupus erythematosus (SLE) and associated hypercoagulable state2. Imaging modalities such as computed tomography pulmonary angiography (CTPA) or ventilation-perfusion (V/Q) scan are typically employed to visualize pulmonary vasculature and confirm the presence of emboli. However, the absence of respiratory symptoms such as dyspnea or hemoptysis made PE less likely in this case.
Finally, myocarditis or pericarditis were also considered potential differential diagnoses, given the patient's history of SLE and the possibility of inflammatory cardiac involvement either as part of the disease process or as a consequence of treatment with medications like corticosteroids. Diagnostic evaluation may involve echocardiography to assess for myocardial inflammation or pericardial effusion, while cardiac magnetic resonance imaging (MRI) can provide additional details regarding myocardial tissue inflammation1,2. However, the absence of significant findings on echocardiography and the presence of characteristic imaging findings of AD made these diagnoses less likely in this case. Considering the patient's clinical presentation, imaging findings, and laboratory results, AD emerged as the most likely diagnosis, necessitating urgent intervention and management.
Treatment and management. Upon confirmation of AD, the patient underwent a multidisciplinary approach to management involving medical therapy and surgical intervention. Initially, esmolol and diltiazem infusion were initiated to control heart rate and blood pressure, aiming to reduce shear forces on the dissected aorta and minimize propagation of the dissection. Subsequently, the patient was transitioned to oral medications including diltiazem, carvedilol, hydralazine, isosorbide dinitrate, and minoxidil, with concurrent nitroprusside infusion as needed for blood pressure control.
Surgical intervention played a crucial role in the patient’s management. Aortic valve repair with commissural resuspension, replacement of ascending aortic hemi-arch, placement of a frozen elephant trunk in the descending aorta, and insertion of a subclavian arterial stent were performed to address the extensive involvement of the aorta and mitigate the risk of further complications such as rupture or malperfusion2.
In addition to pharmacological and surgical interventions, the patient received supportive care to manage complications arising during hospitalization, including hyperkalemic cardiac arrest, Haemophilus influenzae pneumonia, acute cerebral infarcts, and acute renal failure necessitating continuous renal replacement therapy (CRRT).
The pneumonia, caused by H. influenzae, was attributed to a combination of postoperative immunosuppression, chronic corticosteroid use, and impaired airway clearance during the prolonged intubation period following surgery.
The acute cerebral infarcts were likely embolic or hypoperfusion-related. Embolization from the disrupted aortic arch or intraoperative manipulation may have resulted in emboli to cerebral vessels. Alternatively, transient intraoperative hypotension and altered cerebral perfusion during aortic cross-clamping could have led to watershed infarcts. Neurologic symptoms postoperatively prompted neuroimaging, which confirmed multiple small cortical infarcts. These complications highlight the systemic impact of extensive Stanford Type A aortic dissection and the importance of aggressive perioperative monitoring in patients with complex multisystem disease such as SLE.
Post-discharge, the patient was maintained on an aggressive antihypertensive regimen consisting of carvedilol 12.5 mg twice daily, diltiazem extended-release 180 mg daily, hydralazine 25 mg three times daily, isosorbide mononitrate 30 mg daily, and minoxidil 5 mg daily. This combination targeted both heart rate and afterload reduction to minimize shear stress on the aortic wall and prevent recurrence. Hydroxychloroquine 400 mg daily was continued for SLE management, while prednisone was tapered gradually with a plan to transition to steroid-sparing immunosuppressants under rheumatology guidance.
A review of medications contraindicated in AD is crucial to avoid such drugs that can exacerbate the patient’s condition. For example, vasodilators like hydralazine can increase aortic wall stress and are avoided in acute AD. Similarly, beta-blockers with intrinsic sympathomimetic activity (e.g., pindolol) are contraindicated as they can increase heart rate and worsen dissection. Nonsteroidal anti-inflammatory drugs (NSAIDs), due to their impact on renal function and blood pressure, are avoided in patients with AD.
Comparing Type A and Type B AD treatment shows significant differences. Type A dissections, which involve the ascending aorta, are treated as surgical emergencies due to the high risk of complications like rupture, cardiac tamponade, and stroke. Surgical repair, often involving aortic valve repair or replacement and grafting of the aorta, is the definitive treatment. Medical therapy, including beta-blockers and vasodilators, is used initially to stabilize patients before surgery. Alternatively, Type B dissections, affecting the descending aorta, are typically managed medically unless complications arise. Blood pressure and heart rate control are central to preventing dissection progression. Medications like beta-blockers (e.g., esmolol) and vasodilators (e.g., nitroprusside) are used to manage Type B dissections conservatively. Endovascular or surgical intervention is reserved for complicated cases, such as those with organ malperfusion or rupture.
Outcome and follow-up. Long-term follow-up and monitoring were carefully structured to address both cardiovascular health and SLE-related complications.3 The patient was enrolled in coordinated outpatient care involving cardiology, cardiothoracic surgery, rheumatology, nephrology, and neurology. Cardiology follow-up was scheduled initially every 2 weeks for the first 2 months post-discharge, then monthly for the first year to monitor blood pressure control, assess aortic remodeling via imaging (CT angiography every 3–6 months), and titrate medications. Cardiothoracic surgery follow-ups were conducted at 1 month, 3 months, and 6 months to monitor surgical graft integrity and valve function via echocardiography.
Rheumatology followed her every 4 weeks to 6 weeks for the first 6 months to guide SLE management. Corticosteroids were tapered slowly and discontinued by 6 months, at which point mycophenolate mofetil 1 gram twice daily was initiated as a steroid-sparing agent. Hydroxychloroquine 400 mg daily was continued indefinitely.
Due to acute kidney injury requiring CRRT during hospitalization, nephrology monitored her renal recovery monthly for the first 6 months. Her creatinine gradually improved, and dialysis was not required long-term, but mild residual chronic kidney disease persisted, necessitating renal-protective strategies (e.g., ACE inhibitor avoidance, salt restriction). Antihypertensive medications were adjusted over time: minoxidil was discontinued after 3 months due to fluid retention, and isosorbide mononitrate was tapered off as blood pressure stabilized. Carvedilol was increased to 25 mg twice daily, and diltiazem was continued at 240 mg daily for combined rate and BP control.
Overall, the patient’s condition stabilized with multidisciplinary input, and she remained complication-free 18 months post-dissection, with preserved aortic graft integrity, well-controlled blood pressure, and quiescent SLE.
Discussion. AD is a rare, life-threatening cardiovascular complication in patients with SLE.3-5 The rarity of AD in patients with SLE presents challenges in understanding its pathogenesis and optimal management. Factors such as SLE-related vascular changes, hypertension, and corticosteroid therapy contribute to the risk of aortic complications. Early diagnosis and intervention are crucial for reducing mortality, highlighting the importance of suspicion for AD in hypertensive patients with SLE on long-term corticosteroids.
In this case, a 29-year-old woman with SLE on prednisone and hydroxychloroquine, hypertension, and history of non-infective endocarditis presented with sudden-onset chest pain, cough, and lower extremity paresthesia. CTA revealed Stanford A AD extending from the root of the aorta to the bilateral external iliac arteries. Following medical stabilization with esmolol and diltiazem, the patient underwent complex surgical intervention, including aortic valve repair, replacement of ascending aortic hemi-arch, placement of frozen elephant trunk in descending aorta, and insertion of a subclavian arterial stent.
Despite these efforts, the patient’s hospital course was complicated by hyperkalemic cardiac arrest, H. influenza pneumonia, acute cerebral infarcts, and acute renal failure requiring CRRT. Post-discharge, she was maintained on an aggressive antihypertensive regimen to prevent recurrence, along with hydroxychloroquine therapy, while efforts were made to minimize corticosteroid usage given the recent AD.
Complications of AD are diverse and severe, including stroke, MI, limb ischemia, and organ ischemia or infarction. In this case, acute cerebral infarcts demonstrated the risk of stroke due to compromised blood flow to the brain1. Similarly, MI can result from malperfusion of the coronary arteries, while limb ischemia or infarction occurs when the dissection extends to the aortic branches supplying the extremities. Organ ischemia, including renal or bowel infarction, may develop due to compromised blood supply, further complicating the patient’s prognosis.
Beyond AD, other pathologies of the aorta, such as aneurysms, mural thrombi, and syphilitic aortitis, should also be considered. Aortic aneurysms represent localized dilations of the aorta, which may rupture if untreated, while mural thrombi are clots that form within the aortic wall, potentially leading to embolization1,2. Syphilitic aortitis, now rare due to advances in antibiotic therapy, can cause characteristic ‘tree-barking’ of the aorta, resulting from chronic inflammation and scarring of the vessel wall. A notable consideration is painless AD, which can occur when blood flow to spinal arteries, such as the artery of Adamkiewicz, is obstructed2. This can result in spinal cord ischemia and paralysis, without the hallmark symptom of chest pain. In such cases, the absence of pain can delay diagnosis, underscoring the need for vigilance in patients with SLE or other risk factors for aortic disease, particularly when they present with neurological symptoms or signs of spinal cord ischemia.
Conclusion. In this patient, chronic SLE-related inflammation, routine corticosteroid use, and hypertension likely contributed to the development of AD. Corticosteroids are known to impair connective tissue repair, which may weaken the aortic wall, while hypertension increases the mechanical stress on the aorta, promoting dissection. Given the high mortality of untreated AD, prompt diagnosis and aggressive management are essential, especially in hypertensive SLE patients. Reducing steroid doses and introducing other immunosuppressives early may help mitigate the risk of aortic complications, highlighting the importance of a multidisciplinary approach in managing these complex cases.
AUTHOR:
Divya Shah MD1
AFFILIATIONS:
1University of Arizona College of Medicine–Phoenix, Phoenix, AZ
CITATION:
Shah D. Acute aortic dissection in a patient with systemic lupus erythematosus on corticosteroid therapy. Consultant. Published online July 10, 2025. doi:10.25270/con.2025.07.000002
Received April 13, 2024. Accepted December 20, 2024.
DISCLOSURES:
The author reports no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Divya Shah, MD, University of Arizona College of Medicine–Phoenix, 475 N 5th St, Phoenix, AZ 85004 (shahdivya28@gmail.com)
References
- Aoyagi S, Akashi H, Otsuka H, Sakashita H, Okazaki T, Tayama AK. Acute type A aortic dissection in a patient with systemic lupus erythematosus. Jpn Heart J. 2002;43(5):567-571. doi:10.1536/jhj.43.567
- Wang SH, Chang YS, Liu CJ, Lai CC, Chen TJ, Chen WS. Incidence and risk analysis of aortic aneurysm and aortic dissection among patients with systemic lupus erythematosus: a nationwide population-based study in Taiwan. Lupus. 2014;23(7):665-671. doi:10.1177/0961203314523868
- Yuan SM. Aortic aneurysm and dissection in systemic lupus erythematosus. Z Rheumatol. 2019;78(3):287-294. doi:10.1007/s00393-018-0555-x
- Nor MA, Ogedegbe OJ, Barbarawi A, et al. Systemic lupus erythematosus and cardiovascular diseases: a systematic review. Cureus. 2023;15(5):e39284. doi:10.7759/cureus.39284. PMID: 37346216
- Chaaban N, Kshatriya S. Spontaneous coronary artery dissection with systemic lupus erythematosus. Ochsner J. 2022;22(4):353-355. doi:10.31486/toj.22.0003. PMID: 36561102


