Early use of corticosteroids can preserve muscle function in patients with Duchenne muscular dystrophy (DMD)1
Early use of corticosteroids can preserve muscle function in patients with Duchenne muscular dystrophy (DMD)1
This is a sponsored post written by Catalyst Pharmaceuticals, Inc.
DMD is a rapidly progressive, severely debilitating, rare disease of muscle wasting. The signs and symptoms of DMD include:
Progressive muscle damage in DMD is due to chronic inflammation rather than direct mechanical damage to muscle tissue.3,5,6 Muscle inflammation plays a central role in driving DMD progression by impairing muscle regeneration and increasing muscle degeneration and fibrosis, which ultimately lead to loss of muscle function and progression of DMD.3,5
Corticosteroids are recommended as part of the standard of care for patients with DMD because they can slow the decline in muscle strength and function. Therefore, guidelines recommend initiating corticosteroids before a substantial decline in physical health occurs.7 The earlier the better, as studies have shown that early treatment of corticosteroids in patients with DMD at 2 to 4 years of age may help prolong their ability to walk.1

AGAMREE® is a corticosteroid indicated for the treatment of Duchenne muscular dystrophy (DMD) in patients 2 years of age and older.
AGAMREE has proven efficacy
In clinical trials, it has demonstrated statistically significant improvements in muscle strength and motor function.8 AGAMREE was studied in a multicenter, randomized, double-blind, placebo- and active-controlled clinical trial that enrolled 121 boys aged 4 to <7 years of age with a confirmed DMD diagnosis who were ambulatory and who had not been previously treated with corticosteroids. Patients were randomized to treatment with AGAMREE 6 mg/kg/d (n=30), AGAMREE 2 mg/kg/d (n=30), prednisone 0.75 mg/kg/d (n=31), or placebo (n=30) for 24 weeks.8 After 24 weeks, patients on prednisone and placebo crossed over to receive AGAMREE at either 6 mg/kg/d or 2 mg/kg/d for an additional 24 weeks, making the total time 48 weeks. For patients electing not to continue treatment past the end of treatment Period 2, there was a 4-week dose-tapering period. Ninety-four percent of participants (n=114/121) continued from Period 1 to Period 2.9,10
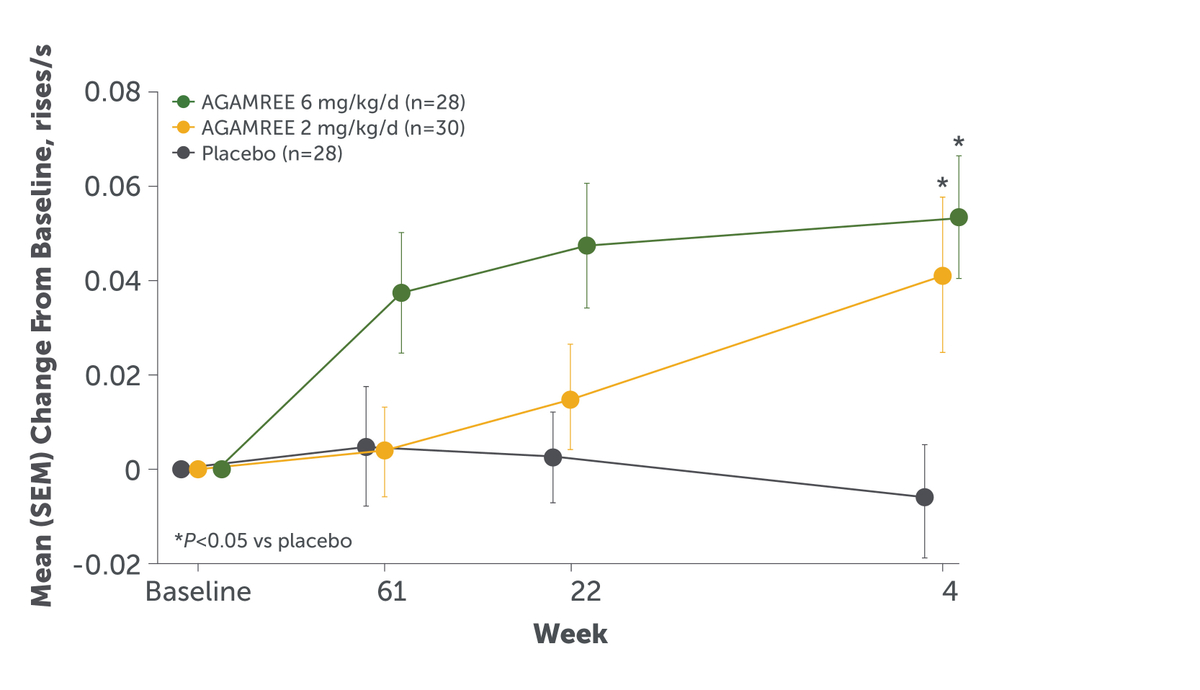
Results from this clinical trial demonstrated that AGAMREE significantly improved motor function as assessed by the Time to Stand Test (TTSTAND velocity).8,9 TTSTAND velocity measures the time required to stand to an erect position from a supine position (floor). For the primary endpoint, AGAMREE 6 mg/kg/d improved TTSTAND velocity at Week 24 vs placebo by 0.06 rises/second (P=0.002). AGAMREE 2 mg/kg/d also improved TTSTAND velocity at Week 24 vs placebo by 0.045 rises/second (P=0.017).8 Importantly, these improvements were clinically meaningful, as the minimal clinically important difference is greater than 0.02 rises/second.10,11 The clinically meaningful and statistically significantly improvements in TTSTAND velocity were maintained at 48 weeks among patients taking AGAMREE 6 mg/kg/d.10
Visit AGAMREEhcp.com to learn more
SEM, standard error of the mean.
References
- American Academy of Neurology. American Academy of Neurology Muscular Dystrophy Quality Measurement Set. American Medical Association. 2014.
- Duan D, Goemans N, Takeda S, Mercuri E, Aartsma-Rus A. Duchenne muscular dystrophy. Nat Rev Dis Primers. 2021;7(1):13.
- Miyatake S, Shimizu-Motohashi Y, Takeda S, Aoki Y. Anti-inflammatory drugs for Duchenne muscular dystrophy: focus on skeletal muscle-releasing factors. Drug Des Dev Therapy. 2016;10:2745-2758.
- Ohlendieck K, Swandulla D. Complexity of skeletal muscle degeneration: multi-systems pathophysiology
and organ crosstalk in dystrophinopathy. Pflugers Arch. 2021;473(12):1813-1839. - Grounds MD, Terrill JR, Al-Mshhdani BA, Duong MN, Radley-Crabb HG, Arthur PG. Biomarkers for Duchenne muscular dystrophy: myonecrosis, inflammation and oxidative stress. Dis Model Mech. 2020;13(2):dmm043638.
- De Paepe B, De Bleecker JL. Cytokines and chemokines as regulators of skeletal muscle inflammation: presenting the case of Duchenne muscular dystrophy. Mediators Inflamm. 2013;2013:540370.
- Bushby K, Finkel R, Birnkrant DJ, et al. Diagnosis and management of Duchenne muscular dystrophy, part 1: diagnosis, and pharmacological and psychosocial management. Lancet Neurol. 2010;9(1):77-93.
- AGAMREE (vamorolone) Oral Suspension [prescribing information]. Catalyst Pharmaceuticals, Inc.; 2024.
- Guglieri M, Clemens PR, Perlman SJ, et al. Efficacy and safety of vamorolone vs placebo and prednisone among boys with Duchenne muscular dystrophy: a randomized clinical trial. JAMA Neurol. 2022;79(10):1005-1014.
- Dang UJ, Damsker JM, Guglieri M, et al. Efficacy and safety of vamorolone over 48 weeks in boys with Duchenne muscular dystrophy: a randomized controlled trial. Neurology. 2024;102(5):e208112.
- Duong T, Canbek J, Birkmeier M, et al; CINRG-DNHS Investigators. The minimal clinical important difference (MCID) in annual rate of change of timed function tests in boys with DMD. J Neuromuscul Dis. 2021;8(6):939-948.
IMPORTANT SAFETY INFORMATION
Contraindications
AGAMREE is contraindicated in patients with known hypersensitivity to vamorolone or any of the inactive ingredients in AGAMREE.
Warnings & Precautions
- Alterations in Endocrine Function: Corticosteroids, such as AGAMREE, can cause serious and life-threatening alterations in endocrine function, especially with chronic use. Monitor patients receiving AGAMREE for Cushing’s syndrome, hyperglycemia, and adrenal insufficiency after AGAMREE withdrawal. In addition, patients with hypopituitarism, primary adrenal insufficiency or congenital adrenal hyperplasia, altered thyroid function, or pheochromocytoma may be at increased risk for adverse endocrine events. Acute adrenal insufficiency can occur if AGAMREE is withdrawn abruptly, and could be fatal. The risk of adrenal insufficiency is reduced by gradually tapering the dose when withdrawing treatment. For patients already taking corticosteroids during times of stress, the dosage may need to be increased.
- Immunosuppression and Increased Risk of Infection: Use of corticosteroids, including AGAMREE, increases the risk of new infection, exacerbation of existing infections, dissemination, and reactivation or exacerbation of latent infection and may mask some signs of infection; these infections can be severe, and at times fatal. Tell patients and/or caregivers to inform their healthcare provider if the patient has had recent or ongoing infections or has recently received a vaccine. Advise patients taking AGAMREE to avoid exposure to chickenpox or measles and to alert their healthcare provider immediately if they are exposed.
- Alterations in Cardiovascular/Renal Function: Monitor for elevated blood pressure and monitor sodium and potassium levels in patients chronically treated with AGAMREE.
- Gastrointestinal Perforation: Use of corticosteroids increases the risk of gastrointestinal perforation in patients with certain gastrointestinal disorders, such as active or latent peptic ulcers, diverticulitis, fresh intestinal anastomoses, and non-specific ulcerative colitis. Signs and symptoms of gastrointestinal perforation may be masked.
- Behavioral and Mood Disturbances: Potentially severe psychiatric adverse reactions may occur with systemic corticosteroids, including AGAMREE, and may include hypomanic or manic symptoms (eg, euphoria, insomnia, mood swings) during treatment and depressive episodes after discontinuation of treatment. Encourage patients to seek medical attention if psychiatric symptoms develop.
- Effects on Bones: Prolonged use of corticosteroids, such as AGAMREE, can lead to osteoporosis, which can predispose patients to vertebral and long bone fractures. Monitor bone mineral density in patients on long-term treatment with AGAMREE.
- Ophthalmic Effects: The use of corticosteroids, such as AGAMREE, may increase the risk of cataracts, ocular infections, and glaucoma. Monitor intraocular pressure if treatment with AGAMREE is continued for more than 6 weeks.
- Vaccination: Do not administer live-attenuated or live vaccines to patients receiving AGAMREE. Administer live-attenuated or live vaccines at least 4 to 6 weeks prior to starting AGAMREE.
- Effects on Growth and Development: Long-term use of corticosteroids, including AGAMREE, can have negative effects on growth and development in children.
- Thromboembolic Events: Observational studies have shown an increased risk of thromboembolism. Use AGAMREE with caution in patients who have or may be predisposed to thromboembolic disorders.
Adverse Reactions
The most common adverse reactions (>10% for AGAMREE and greater than placebo) are cushingoid features, psychiatric disorders, vomiting, weight increased, and vitamin D deficiency.
Use in Specific Populations
- In patients with mild to moderate hepatic impairment, the recommended daily dose of AGAMREE is 2 mg/kg, preferably with a meal, up to a maximum daily dosage of 100 mg for patients weighing more than 50 kg.
- When used concomitantly with strong CYP3A4 inhibitors, the maximum recommended daily dose of AGAMREE is 4 mg/kg, preferably with a meal, up to a maximum daily dosage of 200 mg for patients weighing more than 50 kg.
- The safety and effectiveness of AGAMREE have not been established in pediatric patients below the age of 2 years.
INDICATIONS AND USAGE
AGAMREE is a corticosteroid indicated for the treatment of Duchenne muscular dystrophy (DMD) in patients 2 years of age and older.
Please see full Prescribing Information for additional Important Safety Information.
To report SUSPECTED ADVERSE REACTIONS, contact Catalyst Pharmaceuticals, Inc. at 1-844-347-3277 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.

AGAMREE is a registered trademark of Santhera Pharmaceuticals (Schweiz) AG.
© 2025 Catalyst Pharmaceuticals, Inc. All Rights Reserved.
AGA-0671-1 November 2025


