Peer Reviewed
Preventing Severe COVID-19 With Therapeutic Management
According to data provided by the Centers for Disease Control and Prevention on September 9, 2021, there were more than 40 million total cases in the United States with more than 109,000 new cases per day. There have been nearly 650,000 deaths recorded in the United States due to COVID-19 with more than 1000 new deaths recorded each day.
Beginning at the time the virus reached the United States in March 2020, there was a steady increase in the number of COVID-19 cases reported until the number peaked in the winter between November 2020 and January 2021. After that, there was a sharp decline in cases until June 2021. Now, due to the Delta variant, the cases have steadily increased, and as of September 2021, the number of cases reported are similar compared with the number of cases reported in fall 2020. This was the topic of discussion at this afternoon’s session at Practical Updates in Primary Care 2021.
Speaker, Poonam Mathur, DO, MPH, from the Institute of Human Virology at the University of Maryland School of Medicine in Baltimore, Maryland, spoke about strategies for the prevention of severe disease due to the virus that causes COVID-19.
“The purpose of monoclonal antibody therapy is to prevent the progression to severe disease,” Dr Mathur said.
Describing the Virology
Dr Mathur started the presentation by discussing the virology of COVID-19. She said it is caused by the SARS-CoV-2 virus. The RNA virus is mostly spread by respiratory droplets or aerosols during face-to-face contact. Spike protein on viral surface binds to ACE2 receptor on target cells, facilitating viral entry into host cells.
Then Dr Mathur talked about the viral targets of SARS-CoV-2 life cycle (Figure 1).
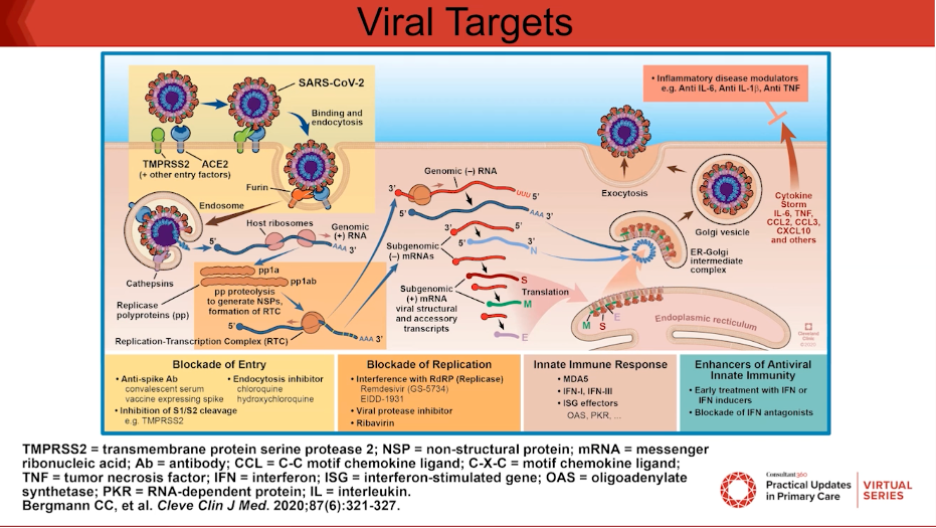
“At the point of entry, that is where we will see the monoclonal antibodies acting. For replication, we can administer remdesivir to stop that, and then there are immune modulators like baricitinib, which are Janus kinase 1 or 2 inhibitors that allow for alteration of the innate immune response,” Dr Mathur said.
Transmission Dynamics
Dr Mathur says that since SARS-CoV-2 is transmitted by airborne transmission, masks are very important to prevent transmission. The exposure is minimized when all parties are wearing masks.
After a person is exposed and infected with SARS-CoV-2, it can be up to 6 days before the person shows signs or symptoms. It has been hard to control the virus because of the long incubation period. This is different from other viruses that have caused an epidemic with fear of causing a pandemic, such as the Ebola virus.
Symptoms may include fever, cough, or shortness of breath. Some may progress to have severe illness or critical illness, which may be later in the course of the disease on days 7 to 14.
Disease Severity
Dr Mathur then outlined the different COVID-19 disease severity categories (Figure 2). The Chinese Centers for Disease Control and Prevention found that out of 44,672 cases, most of the cases were considered mild to moderate and less than 15% were considered severe and 6% were considered critical.

Monoclonal Antibody Therapies With Emergency Use Authorization
Dr Mathur says that many variants that can escape a single monoclonal antibody (mAb) are currently in circulation. The single mAbs, casirivimab and imdevimab, allow for many escape mutations, which theoretically, would predispose to treatment failure. However, when used in combination, there are very few escape mutations.
“It is thought that use of 2 monoclonal antibodies would be superior to just one,” Dr Mathur said.
Figure 3 outlines 3 mAb therapies with emergency use authorization (EUA) from the US Food and Drug Administration (FDA).
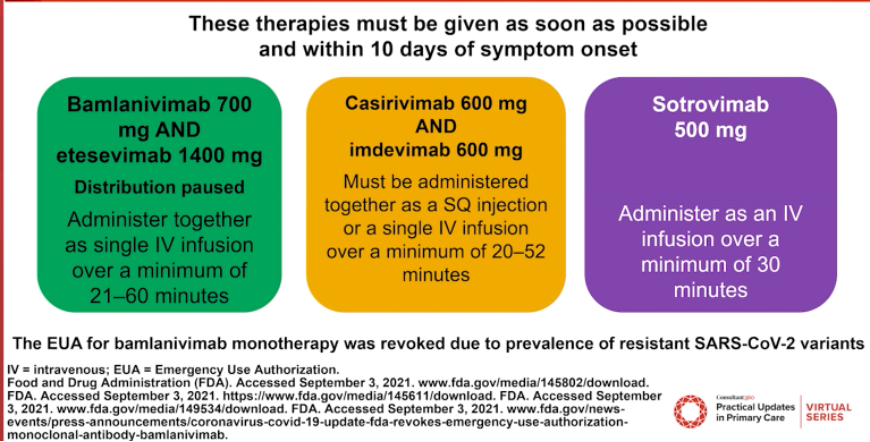
The FDA has determined a very specific population with mild to moderate COVID-19 is eligible for EUA of mAb therapy. Patients must be at least 12 years of age, weigh at least 40 kg, have positive results of direct SARS-CoV-2 viral testing, be within 10 days of symptom onset, and be at high risk of progressing to severe COVID-19 or hospitalization.
“These therapies may be associated with worse clinical outcomes in hospitalized COVID-19 patients requiring high-flow oxygen or mechanical ventilation,” Dr Mathur said.
According to Dr Mathur, the reason the duel mAb is thought to work better than the single mAb is because there are 2 potent antibodies that are binding to different parts of the receptor-binding domain of the spike protein, and they are supposed to prevent generation of escaping end-therapy failure.
Dr Mathur continued to speak about the trials that led to the EUA of mAb, the data that supports the use of mAbs, the reasons why distribution of bamlanivimad and esesevimab was paused, the dosing of the mAb therapy, ways to locate an infusion center for patients, and ways to talk to your patients about the administration of mAb therapy.
—Jessica Bard
Reference:
Mathur P. Therapeutic management of COVID-19: strategies for the prevention of severe disease. Presented at Practical Updates in Primary Care 2021; September 23-25, 2021; Virtual.


