Collagenase Clostridium Histolyticum Shows Efficacy in Subgroup of Patients with Plantar Fibromatosis
Key Highlights:
- Collagenase clostridium histolyticum (CCH) improved Foot Function Index (FFI) Total Pain and combined Pain and Difficulty scores compared with placebo in a post hoc subgroup of patients with plantar fibromatosis.
- Patients treated with CCH showed higher response rates for reduced nodule consistency and improved Clinical Global Impression of Change scores.
- The safety profile of CCH was consistent with previous use in Dupuytren contracture and Peyronie disease, with no serious treatment-related adverse events.
- These findings may support identifying patients more likely to benefit from CCH treatment and inform ongoing phase 3 research.
In a post hoc analysis of a phase 2 randomized controlled trial, collagenase clostridium histolyticum (CCH) demonstrated statistically significant treatment benefits compared with placebo in a selected subgroup of patients with plantar fibromatosis (PFI). Key findings included greater improvement in Foot Function Index (FFI) Total Pain scores, combined FFI Pain and Difficulty scores, nodule hardness, and patient-rated outcomes such as Clinical Global Impression of Change (CGIC) and nodule consistency.
Plantar fibromatosis, also known as Ledderhose disease, is a rare, benign connective tissue disorder marked by fibrous collagen nodules in the plantar fascia. These nodules can cause significant pain, difficulty with ambulation, and reduced quality of life. Given the limited nonsurgical treatment options for symptomatic PFI, the enzymatic activity of CCH—already approved for use in related fibroproliferative conditions such as Dupuytren contracture and Peyronie disease—has prompted interest in evaluating its efficacy in this setting.
This study involved a post hoc subgroup analysis from a larger phase 2 trial population of 172 patients. The subgroup excluded patients with either an extreme baseline Numeric Rating Scale (NRS) pain score of 10 or nodule consistency rated as “moderately firm.” This resulted in a subset of 126 patients (61 treated with CCH and 65 with placebo). Primary efficacy assessments included changes from baseline to day 57 in FFI subscale scores and nodule parameters, analyzed using ANCOVA and mixed models for repeated measures. Responders for nodule consistency and CGIC were also evaluated using the Cochran-Mantel-Haenszel test.
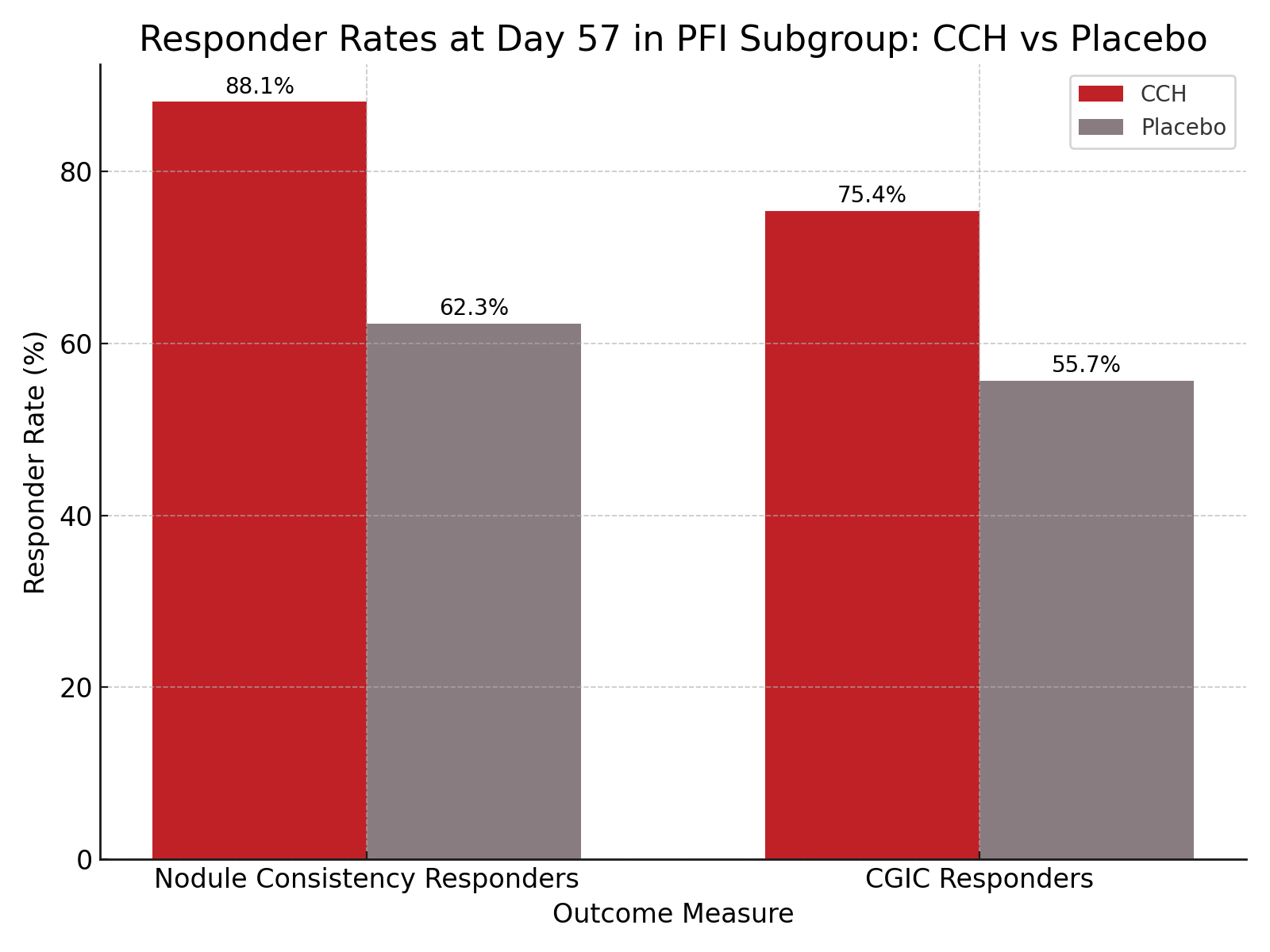
Patients in the CCH subgroup exhibited greater improvements in FFI Total Pain (LSM difference of 3.66; 95% CI, 0.80–6.53; P = .01) and combined Pain and Difficulty scores (LSM difference of 6.96; 95% CI, 1.05–12.87; P = .02) compared with placebo. Nodule consistency responders comprised 88.1% of the CCH group versus 62.3% in the placebo group (P = .001), while CGIC responders totaled 75.4% in the CCH group compared with 55.7% for placebo (P = .03). Improvements in nodule hardness were also superior in the CCH group.
Adverse events were consistent with the known safety profile of CCH, with the majority being mild to moderate injection site reactions. No serious adverse events or treatment-related complications were observed in the CCH arm.
“These post hoc results may help in identification of patients with PFI who are more likely to benefit from CCH treatment,” the study authors concluded.
Reference:
Anderson CJ, Gottlieb I, Levy J, et al. Collagenase clostridium histolyticum (cch) in patients with plantar fibromatosis (pfi): post hoc analysis of a phase 2, double-blind, randomized, placebo-controlled study. Presented at: The APMA 2025 Annual Scientific Meeting; July 24–27, 2025; Gaylord Texan, Dallas-Fort Worth, TX. https://www.apma.org/the-national/


