Brexpiprazole Reduces Agitation Associated With Alzheimer Dementia, Japanese Study Finds
A recent multicenter clinical trial has confirmed that brexpiprazole, an antipsychotic medication already approved for agitation associated with Alzheimer dementia (AAD) in the United States, is effective and generally well tolerated in Japanese patients. This randomized, double-blind, placebo-controlled study is the first to examine brexpiprazole in an Asian population and offers insight into its clinical utility in a region where treatment options for AAD remain limited.
Agitation is among the most disruptive neuropsychiatric symptoms in Alzheimer disease, affecting approximately 45% of patients and often leading to caregiver distress, earlier institutionalization, and increased healthcare costs. In many countries, there are no approved treatments for AAD, leading to off-label antipsychotic use with questionable efficacy and significant safety concerns. Brexpiprazole, a serotonin-dopamine activity modulator, is currently the only agent approved by the FDA specifically for this indication.
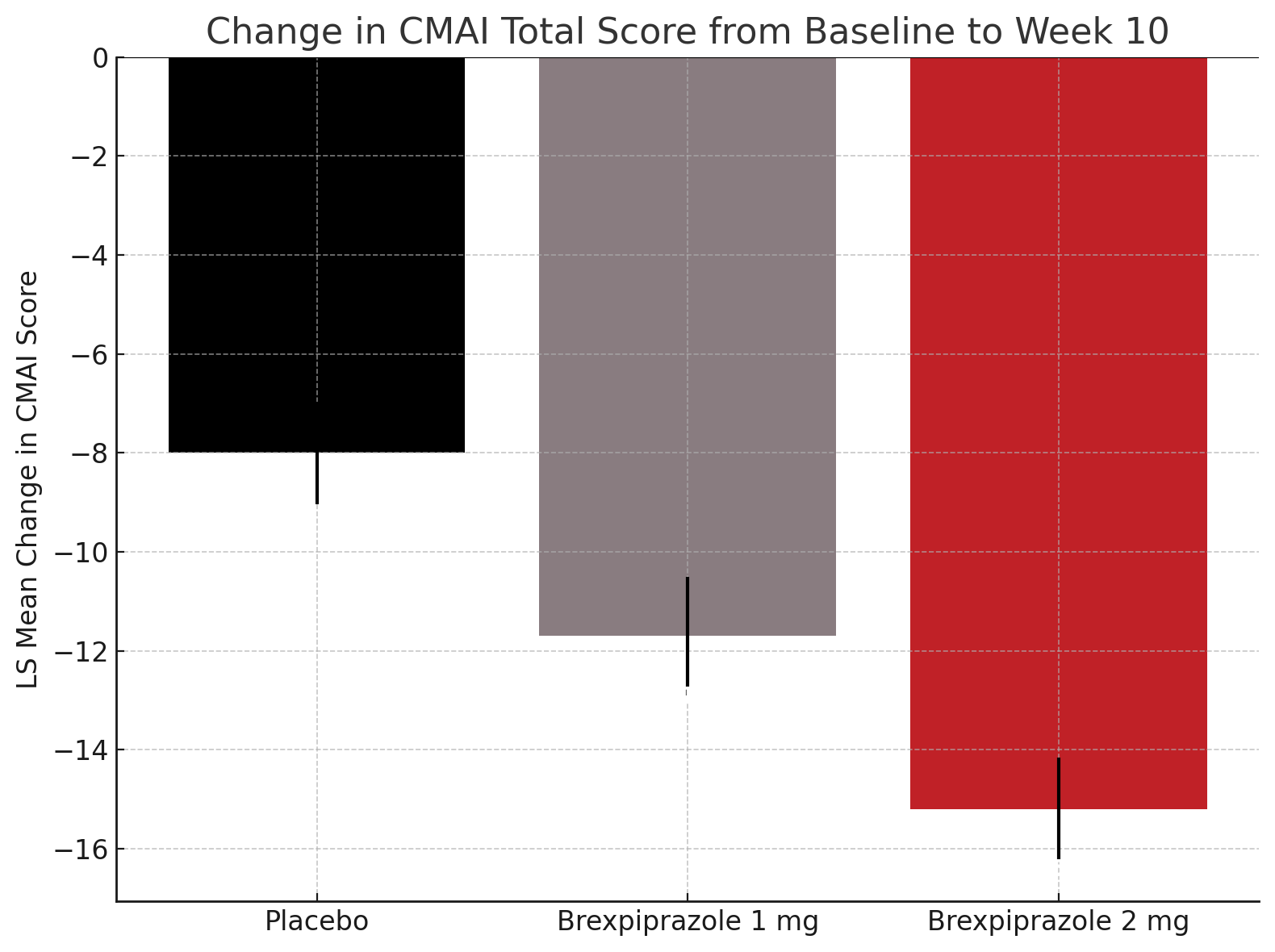
The current study enrolled 410 patients aged 55 to 90 years with probable Alzheimer disease and significant agitation symptoms, as measured by standardized neuropsychiatric inventories. Participants were randomized to receive either brexpiprazole 1 mg/day, brexpiprazole 2 mg/day, or placebo during a 10-week treatment period. The primary efficacy endpoint was the change in the Cohen-Mansfield Agitation Inventory (CMAI) total score from baseline to week ten. Both doses of brexpiprazole demonstrated statistically significant reductions in CMAI scores compared with placebo, indicating meaningful improvements in agitation symptoms. The 2 mg/day group achieved a least squares mean difference of -7.2 versus placebo (P < .0001), while the 1 mg/day group showed a difference of -3.7 (P = .0175). Improvements in agitation symptoms were observable from week 4 in the 2 mg group and from week 8 in the 1 mg group.
Secondary outcomes, including changes in CMAI subscale scores and global clinical impressions of severity and improvement, mirrored the primary results. Notably, the 1 mg/day dose, which had not shown consistent efficacy in previous global trials, demonstrated statistically significant improvements across multiple measures in this study. These findings suggest that lower doses may be effective in certain patient populations, particularly among older and lower-weight individuals, as was the case in this cohort.
The study also closely evaluated safety outcomes. Treatment-emergent adverse events (TEAEs) occurred in 76.8% of patients in the 1 mg group, 84.6% in the 2 mg group, and 73.8% in the placebo group. While the overall incidence of adverse events was higher than in prior global studies, most were mild to moderate in severity. Common TEAEs included somnolence, bradykinesia, insomnia, and muscle rigidity. Extrapyramidal symptoms (EPS) occurred more frequently in patients receiving brexpiprazole than placebo, but these were generally not severe, and scores on EPS-specific rating scales indicated minimal clinical impact. Two deaths occurred in the 1 mg group—due to cardiac arrest and aspiration pneumonia—but investigators determined these were unrelated to the study medication.
The patient population in this trial was distinct from those in earlier studies. Japanese participants were on average older (around 80 years old) and had a lower body weight (approximately 48 kg) compared with Western cohorts. These demographic differences may account for the higher rates of reported TEAEs, though the overall benefit-risk profile of brexpiprazole remained favorable. The study also included patients across a wide spectrum of care settings, including hospitals, nursing homes, and home care, enhancing the generalizability of its findings within Japan.
The researchers noted that while the 10-week treatment period was sufficient to demonstrate efficacy, longer-term data are still needed to evaluate the durability of response and risks such as tardive dyskinesia. An extension study spanning 24 weeks is currently underway and is expected to provide further insight into brexpiprazole’s long-term safety and effectiveness in this population.
Reference:
Nakamura Y, Adachi J, Hirota N, et al. Brexpiprazole treatment for agitation in Alzheimer's dementia: A randomized study. Alzheimer’s Dement. 2024;20(11):8002-8011. doi:10.1002/alz.14282


