Noninvasive Glucose Monitor Demonstrates Accuracy in Patients With Diabetes Using Visible Light Technology
Key Highlights
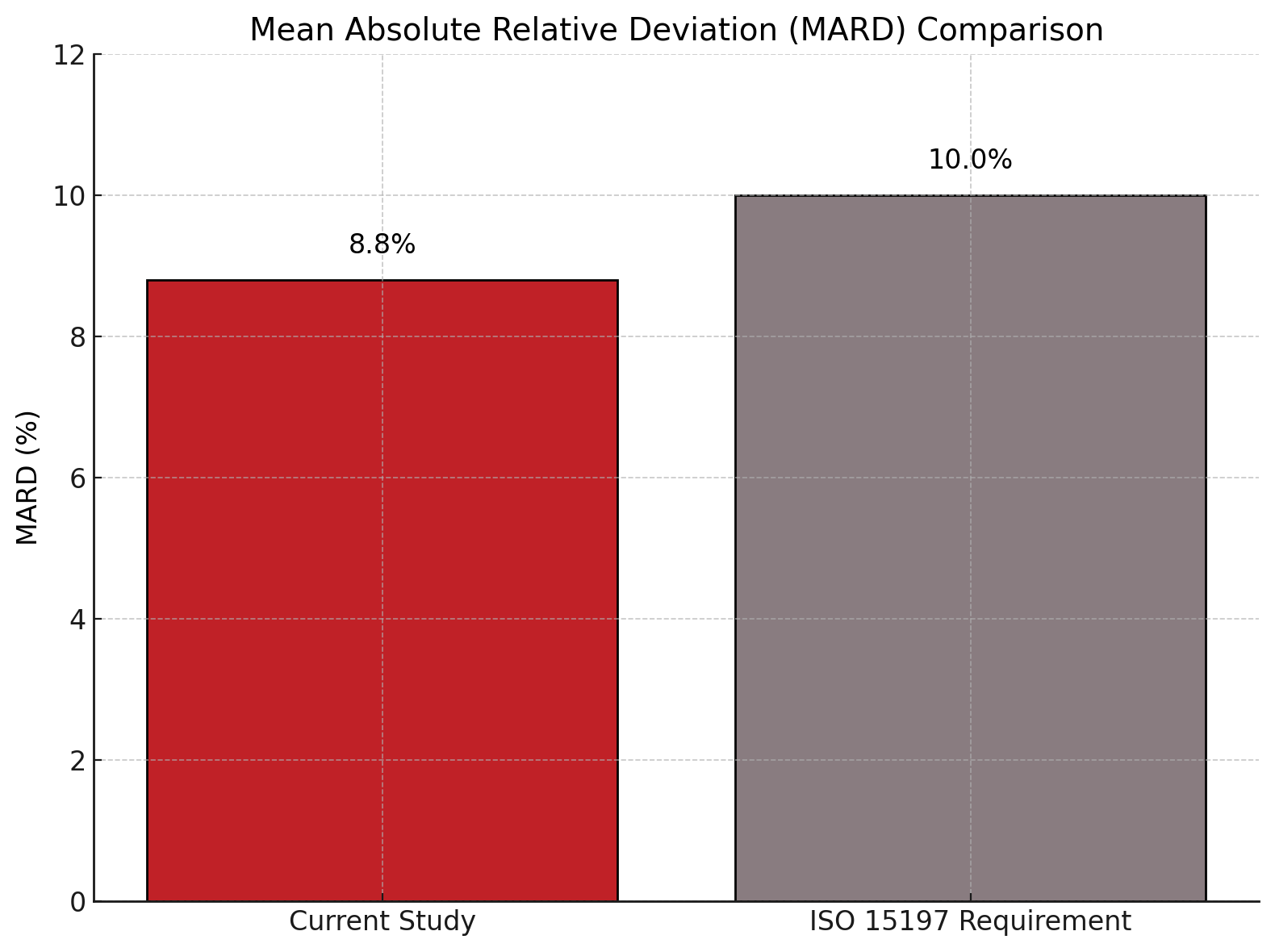
- The non-invasive glucose meter achieved a Mean Absolute Relative Deviation (MARD) of 8.8%.
- 100% of measurements fell within Zones A and B of the Consensus Error Grid, surpassing ISO 15197 standards.
- The device demonstrated clinical reliability in both fasting and postprandial glucose states.
- Calibration incorporated individual biometric characteristics such as fingertip thickness, skin condition, and BMI.
A novel non-invasive glucose monitoring device demonstrated high accuracy and clinical reliability in a study involving patients with diabetes presented at the American Diabetes Association 85th Scientific Session. With a MARD of 8.8% and 100% of measurements falling within the clinically acceptable Zones A and B on the Consensus Error Grid, the optical device shows promise as a viable alternative to current invasive methods, according to the study authors.
As diabetes prevalence is expected to reach 800 million globally by 2035, there is an urgent need for continuous glucose monitoring technologies that eliminate the discomfort and costs associated with traditional finger-prick and invasive sensors. While several non-invasive techniques have been introduced, their commercial viability has been hindered by insufficient accuracy.
In this study, researchers evaluated an optical glucose meter utilizing 660 nm visible light photoplethysmography (PPG) signals at the fingertip. A total of 28 patients with diabetes (13 men, 15 women) — including three with type 1 and 25 with type 2 diabetes — participated. Blood glucose measurements were taken in both fasting and postprandial states using the device and standard venous blood sampling. The calibration process accounted for individual parameters such as fingertip thickness, skin condition, and body mass index, employing tailored feature extraction to derive glucose estimations.
The device yielded a MARD of 8.8%, reflecting improved precision over previously reported non-invasive approaches. Furthermore, the Consensus Error Grid (Type 1) analysis showed that all data points (100%) were located within Zones A and B, satisfying and exceeding the ISO 15197 guideline that requires 99% of readings in these zones. These findings indicate the method’s strong clinical applicability.
“These results demonstrate the feasibility of non-invasive glucose measurement with accuracy comparable to venous blood glucose levels in subjects with diabetes, supporting the potential for commercialization with further research needed,” the study authors concluded.
Reference:
Kim CS, Yun JH, Song IK, Park HK. Development of noninvasive blood glucose measurement using visible light photoplethysmography signals in subjects with diabetes. Presented at: American Diabetes Association; June 20–23, 2025; Chicago, IL. https://professional.diabetes.org/scientific-sessions.


