Peer Reviewed
Fat Embolism Syndrome After Corticosteroid Use in Sickle Cell Disease
Introduction. Managing patients with sickle cell disease (SCD) and its potential complications remains a challenge. In this inherited disorder, mutation of the beta globin gene results in hemoglobin S (HbS), which can polymerize and lead to red blood cell (RBC) sickling under hypoxic conditions.1 Patients with SCD may carry homozygous (HbSS) or heterozygous (non-HbSS) HbS mutation profiles. Hemolytic anemia and vaso-occlusion with subsequent ischemic damage are the classic pathologic features, but there are other poorly understood events including endothelial dysfunction, inflammation, hypercoagulability, and platelet activation.1,2 These processes lead to a wide range of acute and chronic complications involving multiple organ systems.
Fat embolism syndrome (FES) is a rare, high-risk complication of SCD that manifests with hypoxemia, neurologic impairment, and petechial rash.3 In patients with SCD, FES often occurs in the setting of a vaso-occlusive episode (VOE) and involves bone marrow necrosis, but other risk factors for development are unclear. Non-HbSS genotypes and human parvovirus B12 infection have been associated with FES in a prior meta-analysis.4 While corticosteroids have been controversial in the management of FES, the relationship between corticosteroid use and the development of FES is unknown especially in patients with SCD.
Here, we describe the case of a 28-year-old woman with HbSS SCD who received corticosteroids for allergy-like symptoms and later presented with a VOE and ultimately expired from FES. To our knowledge, this is the only reported case of FES after corticosteroid administration in a patient with the HbSS genotype. We will use this case to highlight corticosteroid use as a possible precipitant of FES in patients with SCD.
Case Description. A 28-year-old woman with HbSS SCD, bilateral avascular necrosis of the hips, and asthma initially presented to the emergency department with facial swelling and urticaria on her arms and wrists. She did not have any known cardiac comorbidities and an echocardiogram 2 years prior did not show evidence of right or left ventricular dysfunction. She received a 3-day course of prednisone 40 mg daily and an oral antihistamine. Her symptoms initially improved but then recurred after completing prednisone. She presented to the emergency department (ED) 6 days later where 10 mg of oral dexamethasone was given, and she was discharged with a 5-day course of prednisone 40 mg daily.
After 2 days, she again presented to the ED with significant pain in her hips and knees and was admitted for a suspected VOE. Complete blood count showed leukocytes of 11.6 g/dL, hemoglobin of 9.8 g/dL, and platelet count of 334,000/microL. She did not have signs or symptoms consistent with acute chest syndrome or laboratory evidence of hemolysis at time of presentation. She was provided pain control with intravenous hydromorphone and ketamine and hydrated with intravenous fluids.
On the second day of admission, she desaturated to 88% on room air and required two liters of supplemental oxygen by nasal cannula. She was afebrile and rapidly became tachycardic and tachypneic. She did not have any stridor, wheezing, rash, or urticaria at this time. Chest x-ray did not reveal any opacity or edema. Later in the afternoon, she became unresponsive without any improvement after naloxone administration. She was intubated for rapidly progressive encephalopathy and need for airway protection. Arterial blood gas after intubation did not reveal acute hypoxia or hypercapnia. Repeat complete blood count showed leukocytes of 14.6 g/dL, hemoglobin of 5.6 g/dL, and platelet count of 70,000/microL. The patient’s serum lactate dehydrogenase was elevated, and chest x-ray after intubation showed new poorly defined bilateral hazy airspace opacities. CT angiogram of her brain did not show any bleed or obvious ischemic event. She quickly developed shock and acute renal failure requiring vasoactive medications and dialysis. She was also started on empiric vancomycin and piperacillin–tazobactam. Simple transfusion of red blood cells was started. Her shock was suspected to be distributive in origin stemming from a rapidly progressive VOE. Central venous access was obtained, and emergent hemodialysis was initiated.
After 2 hours, she had cardiac arrest from refractory shock and expired after a course of cardiopulmonary resuscitation. Her postmortem autopsy showed evidence of systemic fat emboli. Specific findings included bilateral lungs with multifocal fat and bone marrow emboli, alveolar hemorrhage, and hemosiderin-laden macrophages. Bilateral kidneys showed diffuse intravascular fat emboli within the glomeruli. The bowels showed extensive enteric necrosis. The heart showed right ventricular dilation and left ventricular hypertrophy. Neuropathologic evaluation of the brain showed evidence of metabolic encephalopathy and acute hypoxic and ischemic changes, but no fat emboli were observed.
Discussion. This case describes a patient who received corticosteroids for a suspected allergic reaction and ultimately developed an acute VOE which rapidly progressed to FES and death. The pathophysiology of FES as it relates to corticosteroids in SCD is not well understood. In patients with SCD, it has been theorized that exposure to corticosteroids initially conveys cellular membrane-protective and systemic anti-inflammatory effects, and that when steroids are withdrawn, a rebound surge of the pro-inflammatory cascade may then precipitate a VOE.5 The most direct link between VOE and FES was proposed in 1987, in which sickled red blood cells lead to bone marrow necrosis, increased intramedullary pressure, and subsequent retrograde flow of necrotic bone marrow and fat into the intramedullary sinusoids and then the systemic circulation.6
Despite the mechanistic uncertainty, there is growing evidence that administering corticosteroids to patients with SCD may increase risk of a VOE.7,8,9 A 2022 meta-analysis found that 45% of 5151 patients with SCD admitted with VOE had been exposed to corticosteroids in the 5-day period prior to admission.10 In another meta-analysis of patients with SCD, FES was associated with non-HbSS hemoglobin genotypes and/or prior parvovirus B-19 infection.4 In this study, nearly all patients presented with a VOE and then rapidly deteriorated with encephalopathy, respiratory distress, and a significant drop in hemoglobin and platelets. Overall mortality from FES in this cohort was found to be 64% and both simple and exchange transfusion were associated with lower mortality. Our patient was not tested for parvovirus infection but did not have any symptoms consistent with a viral infection. Simple transfusion was initiated at the onset of shock, however she died before exchange transfusion could occur.
From review of the literature, we identified four additional cases of patients with SCD who developed FES in the setting of corticosteroid exposure (Table 1).11,12,13
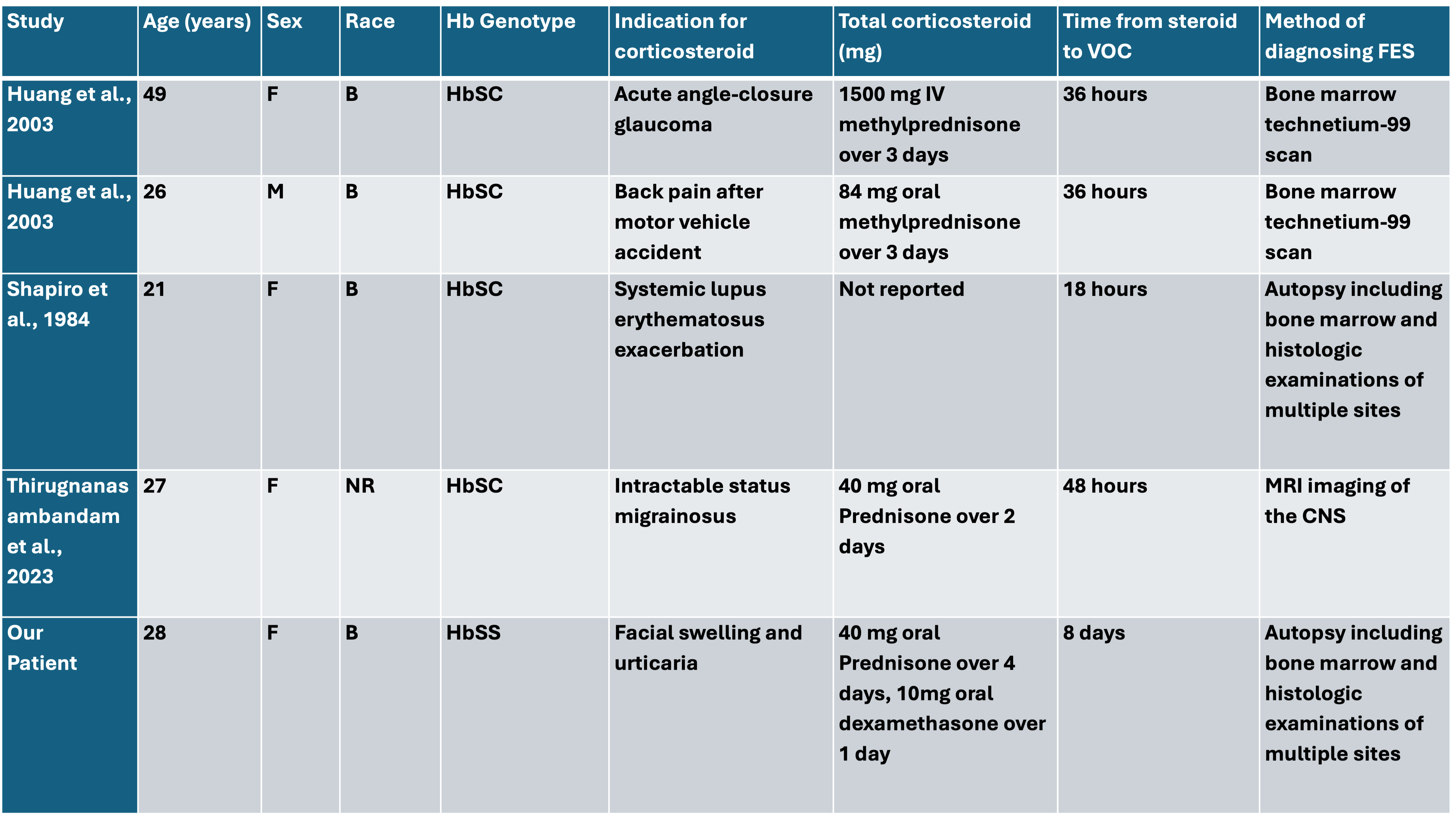
B = Black, F= female, Hb = hemoglobin, M=male.
Beyond SCD, several studies have explored corticosteroid exposure in patients with FES due to long bone fracture. In a 2009 meta-analysis including 398 patients, corticosteroid exposure reduced the risk of FES and hypoxia but not mortality.14 A subsequent 2011 meta-analysis found that among patients receiving corticosteroids after long bone fractures, the rate of FES was lower than in those who did not receive steroids. The authors noted poor overall quality of available evidence which limited any strong conclusions regarding steroid use.15
The diagnosis of FES is primarily clinical and should be suspected in patients who develop a new petechial rash with hypoxemia and/or neurologic impairment. Unfortunately, neurologic and pulmonary imaging findings are non-specific, and there are no reliable biomarkers for this disease. The management of FES is centered on supportive intensive care. There are no disease-specific treatments and there is insufficient evidence to support the use of commonly proposed treatments including anticoagulation and corticosteroids.16 In patients with hematologic abnormalities like thrombocytopenia, coagulopathy, and bone marrow injury, these interventions may even be harmful.17 Management of FES includes fluid resuscitation, vasoactive medications if needed, neurologic assessments with frequent exams or intracranial pressure monitoring, and lung-protective mechanical ventilation to support patients who develop acute respiratory distress syndrome.18 Simple and exchange transfusions may also benefit patients with FES and SCD.4 A recent retrospective study demonstrated a 100% survival rate in patients with SCD and FES (n = 18) who received both exchange transfusion and plasmapheresis, which may remove fat emboli that drive inflammation.19 Although survivorship bias may be a limitation, these results are very encouraging considering the high mortality of FES. While our case shows FES occurring in a patient with HbSS SCD after corticosteroid use, the diagnosis of FES is likely underdiagnosed and underreported making it difficult to determine the nature of this relationship. Our patient also had avascular necrosis, which we considered to be a form of long-bone damage in this clinical context, which may have also predisposed her to this event.
Conclusion. We present the unique case of a patient with HbSS SCD who received corticosteroids and then developed a VOE and FES. We share this case to highlight a rare complication of SCD that requires a high-index of clinical suspicion given the limitations in testing and lack of biomarkers for FES. It raises the possibility that steroids may be implicated in the precipitation of VOE and subsequent FES. It should be considered in the differential diagnosis in patients with SCD and respiratory failure, neurologic impairment, or shock after recent corticosteroid exposure. Although FES is rare and the relationship to corticosteroids remains unclear, it should be part of the risk-benefit analysis when considering corticosteroid use in patients with SCD.
AUTHORS:
Alexander O’Hara MD 1 • Daniel Sweeney MD 1 • Kirk Cahill MD 2 • Gail Kellberg APN 2 • Patrick Stiff MD 2
AFFILIATIONS:
1Department of Medicine, Loyola University Medical Center
2Division of Hematology/Oncology, Loyola University Medical Center
CITATION:
O'Hara A, Sweeney D, Cahill K, Kellberg G, Stiff P. Fat embolism syndrome after corticosteroid use in sickle cell disease. Consultant. Published online July 16, 2025. doi:10.25270/con.2025.07.000003
Received February 20, 2025. Accepted April 8, 2025
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Alexander O’Hara MD, 2160 S First Ave, Maywood IL 60153 (alexander.ohara@luhs.org)
References:
- Piel FB, Steinberg MH, Rees DC. Sickle cell disease. N Engl J Med. 2017;376(16):1561-1573. https://doi.org/10.1056/NEJMra1510865
- Connes P, Renoux C, Joly P, Nader E. Vascular pathophysiology of sickle cell disease. Presse Med. 2023;52(4):104202. https://doi.org/10.1016/j.lpm.2022.104202
- Novelli EM, Gladwin MT. Crises in sickle cell disease. Chest. 2016;149(4):1082-1093. https://doi.org/10.1016/j.chest.2015.12.016
- Tsitsikas DA, Bristowe J, Abukar J. Fat embolism syndrome in sickle cell disease. J Clin Med. 2020;9(11):3601. https://doi.org/10.3390/jcm9113601
- Tsitsikas DA, Gallinella G, Patel S, et al. Bone marrow necrosis and fat embolism syndrome in sickle cell disease: increased susceptibility of patients with non-SS genotypes and a possible association with human parvovirus B19 infection. Blood Rev. 2014;28(1):23-30. https://doi.org/10.1016/j.blre.2013.11.001
- Ballas SK. Corticosteroids and sickle cell disease. J Natl Med Assoc. 2009;101(3):283-285. https://doi.org/10.1016/S0027-9684(15)30921-4
- Simkin PA, Downey DJ. Hypothesis: retrograde embolization of marrow fat may cause osteonecrosis. J Rheumatol. 1987;14(5):870-872.
- Lanzkron S, Pecker L. Pain without gain: steroids and sickle crisis. Blood. 2022;139(26):3678-3679. https://doi.org/10.1182/blood.2022016201
- Griffin TC, McIntire D, Buchanan GR. High-dose intravenous methylprednisolone therapy for pain in children and adolescents with sickle cell disease. N Engl J Med. 1994;330(11):733-737. https://doi.org/10.1056/NEJM199403173301104
- Darbari DS, Castro O, Taylor JG, et al. Severe vaso-occlusive episodes associated with use of systemic corticosteroids in patients with sickle cell disease. J Natl Med Assoc. 2008;100(8):948-951. https://doi.org/10.1016/S0027-9684(15)31448-9
- Walter O, Cougoul P, Maquet J, et al. Risk of vaso-occlusive episode after exposure to corticosteroids in patients with sickle cell disease. Blood. 2022;139(26):3771-3777. https://doi.org/10.1182/blood.2022016268
- Thirugnanasambandam RP, Maraikayar FM, Liu M, et al. Fat embolization syndrome secondary to steroid treatment in a case of sickle cell vaso‐occlusive crisis. Case Rep Hematol. 2023;2023:5530870. https://doi.org/10.1155/2023/5530870
- Huang J, Gay R, Khella S. Sickling crisis, fat embolism, and coma after steroids. Lancet. 1994;344(8927):951-952. https://doi.org/10.1016/S0140-6736(94)92623-8
- Shapiro MP, Hayes JA. Fat embolism in sickle cell disease: report of a case with brief review of the literature. Arch Intern Med. 1984;144(1):181-182. https://doi.org/10.1001/archinte.1984.00350130191049
- Bederman SS, Bhandari M, McKee MD, Schemitsch EH. Do corticosteroids reduce the risk of fat embolism syndrome in patients with long-bone fractures? A meta-analysis. Can J Surg. 2009;52(5):386-393. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2769134/
- Sen RK, Tripathy SK, Krishnan V. Role of corticosteroid as a prophylactic measure in fat embolism syndrome: a literature review. Musculoskelet Surg. 2012;96:1-8. https://doi.org/10.1007/s12306-011-0157-7
- Habashi NM, Andrews PL, Scalea TM. Therapeutic aspects of fat embolism syndrome. Injury. 2006;37(4)(suppl):S68-S73. https://doi.org/10.1016/j.injury.2006.02.009
- Mellor A, Soni N. Fat embolism. Anaesthesia. 2001;56(2):145-154. https://doi.org/10.1046/j.1365-2044.2001.01716.x
- Kosova E, Bergmark B, Piazza G. Fat embolism syndrome. Circulation. 2015;131(3):317-320. https://doi.org/10.1161/CIRCULATIONAHA.114.009592
- Rasool A, Thomas S, Luqmani A, et al. Combined red cell and plasma exchange is associated with reduced mortality of fat embolism syndrome in sickle cell disease. Blood. 2024;144(suppl 1):2497.


