Peer Reviewed
Progressive Diffuse Calciphylaxis With Facial Involvement Despite Multimodal Treatment
Authors:
Pablo Michel, MD
University of Texas Health Science Center at San Antonio, Texas
Travis Vandergriff, MD; and Arturo Dominguez, MD
University of Texas Southwestern Medical Center, Dallas, Texas
Citation:
Michel P, Vandergriff T, Dominguez A. Progressive diffuse calciphylaxis with facial involvement despite multimodal treatment [published online July 10, 2018]. Consultant360.
A 49-year-old woman presented with a 1-month history of bilateral thigh bruising that had progressed to painful, indurated, necrotic ulcerations.
History. Her medical history included end-stage renal disease (ESRD) for which she was on hemodialysis (HD); diabetes mellitus; coronary artery disease; and mitral valve and aortic valve replacements. Her medications included insulin, aspirin, cinacalcet, simvastatin, and warfarin for anticoagulation started 1 year prior.
Physical examination. Physical examination revealed bilateral 10 × 15-cm ulcers in the inner thighs with extension to the adipose tissue that were covered by black eschar with peripheral induration, including a 5 × 3-cm satellite lesion in the left thigh (Figures 1 and 2).
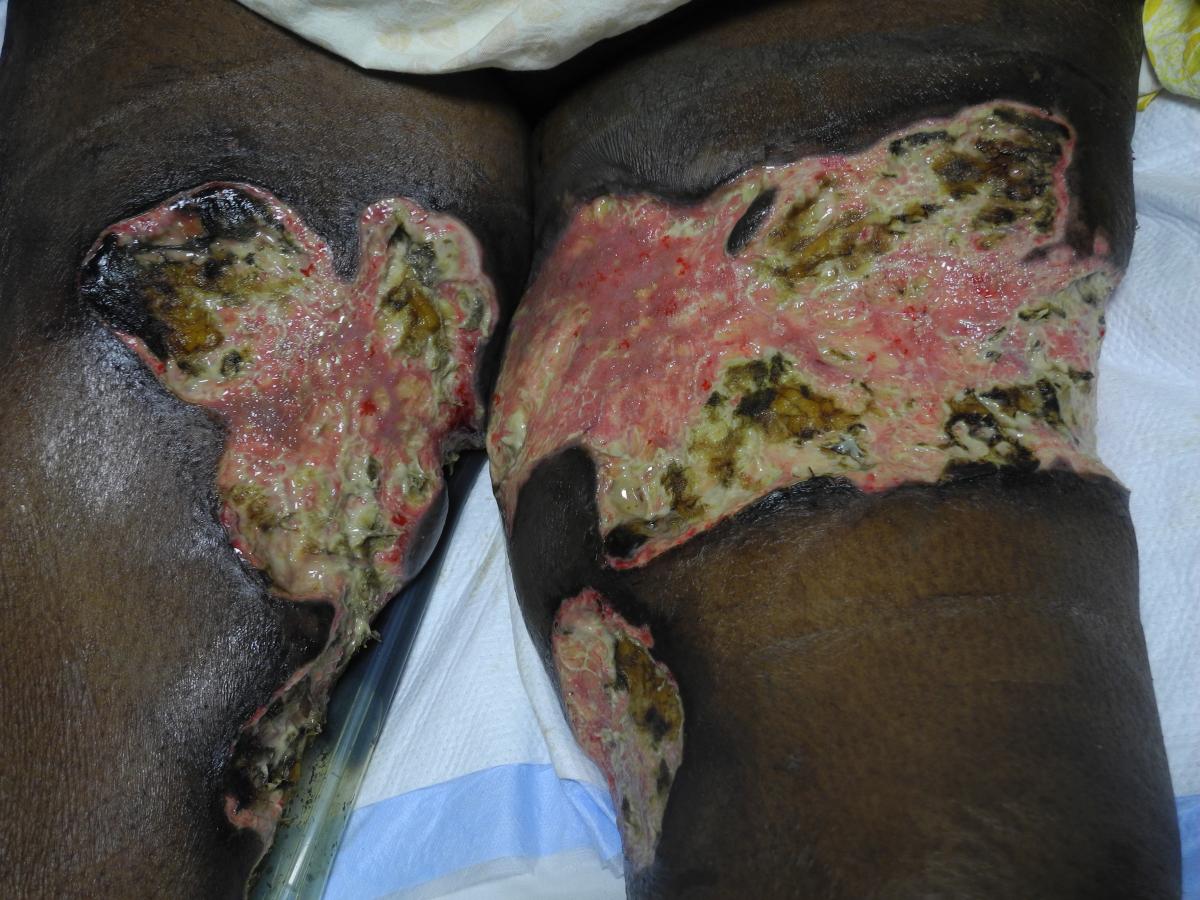
Figure 1. Deep ulcerations with well-defined borders in the anterior aspect of the thighs with fibrinogen at the base. Satellite ulceration in the medial aspect of left thigh can be observed.
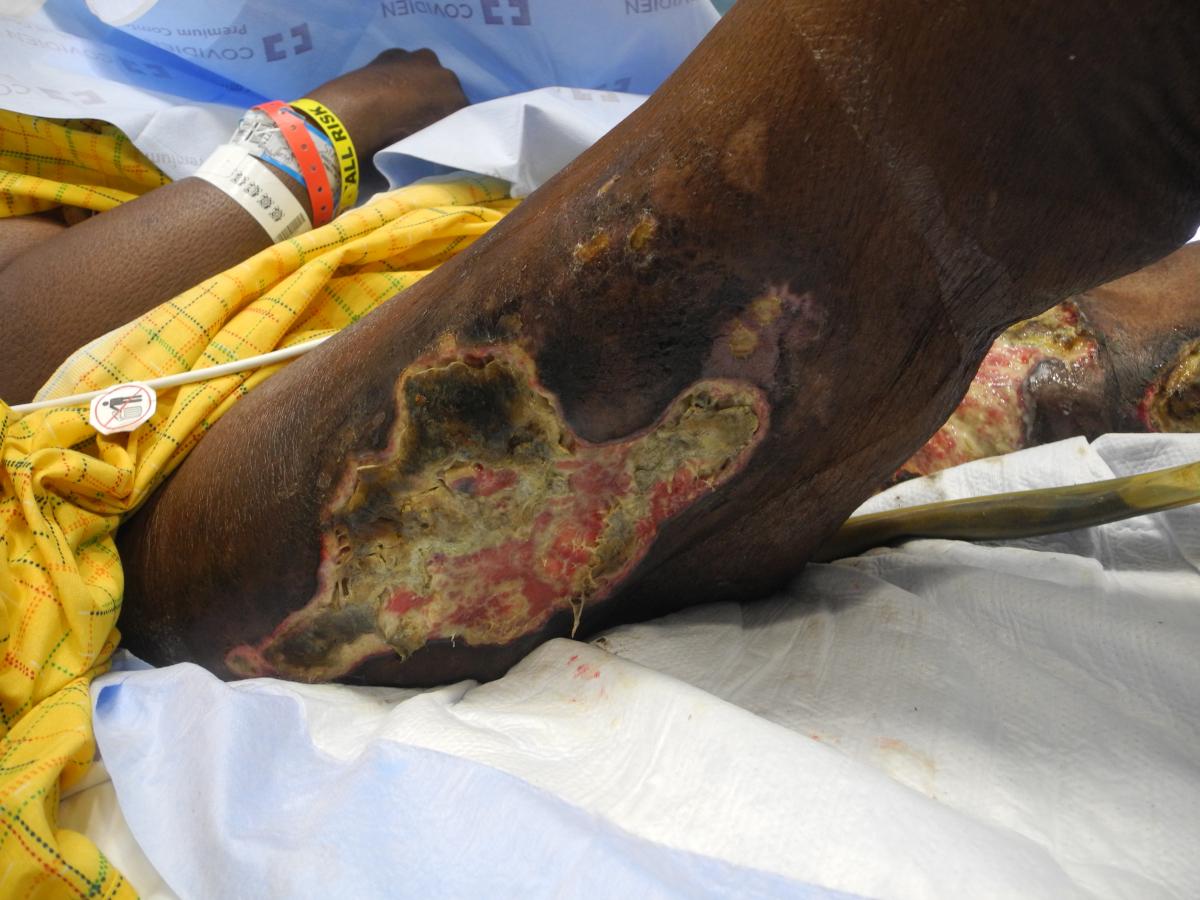
Figure 2. The lateral aspect of the right thigh with a large ulceration covered by black eschar.
At an outside facility, the patient had been treated with antibiotics for presumed bilateral thigh cellulitis. Because of a lack of clinical improvement, biopsies from both thighs had been taken, revealing thrombotic vasculopathy without evidence of calcification. Warfarin-induced skin necrosis (WISN) had been suspected, and anticoagulation subsequently had been discontinued. Treatment with vitamin K (menaquinone) and heparin had been initiated, but her wounds had failed to heal over the course of a month, prompting the transfer to our center.
Diagnostic tests. At presentation, laboratory studies demonstrated anemia of chronic disease, leukocytosis (26,000 cells/µL), hypoalbuminemia, a normal parathyroid hormone level, and a normal calcium-phosphate product. Levels of D-dimer, lupus anticoagulant, anti β2-glycoprotein, and anticardiolipin antibodies were all within normal limits. Cryoglobulins and cryofibrinogens were negative, as well. Antinuclear antibodies were positive in a 1:1280 dilution with a speckled pattern. Protein C activity was 34% (reference range, 83%-138%) despite weeks of vitamin K supplementation.
Because of the patient’s multiple risk factors, clinical presentation, and histopathologic findings, a diagnosis of calciphylaxis was suspected. The patient received 3 surgical debridements with subsequent placement of a vacuum-assisted wound closure device. Intravenous sodium thiosulfate, 25%, 3 times per week with hemodialysis was initiated, and enoxaparin was started as a long-term anticoagulant.
Our dermatopathologist’s review of the initial skin biopsy findings as well as of the debridement tissue revealed dermal necrosis, intravascular hyaline thrombi, vascular calcification, and extravascular soft tissue calcification (Figure 3), findings consistent with calciphylaxis.

Figure 3. Arteriole in the subcutis with mural calcification and thrombus (hematoxylin-eosin, original magnification ×400).
Treatment with sodium thiosulfate, intravenous bisphosphonates, and maggot debridement therapy permitted significant improvement initially. Of note, ongoing vitamin K supplementation was not administered during the hospitalization. Despite medical management, however, the patient developed new lesions, as well as evidence of continued ischemia and thrombosis around existing ulcers. Palliative care and discontinuation of hemodialysis were discussed with the patient and her family; however, they wished to continue aggressive medical management.
After 3 months, the ulcers continued to expand, and new lesions developed in atypical locations, including the scalp, face, knees, and toes (Figures 4 and 5). The patient had several episodes of gastrointestinal tract bleeding and septic shock, and she died 6 months after admission.
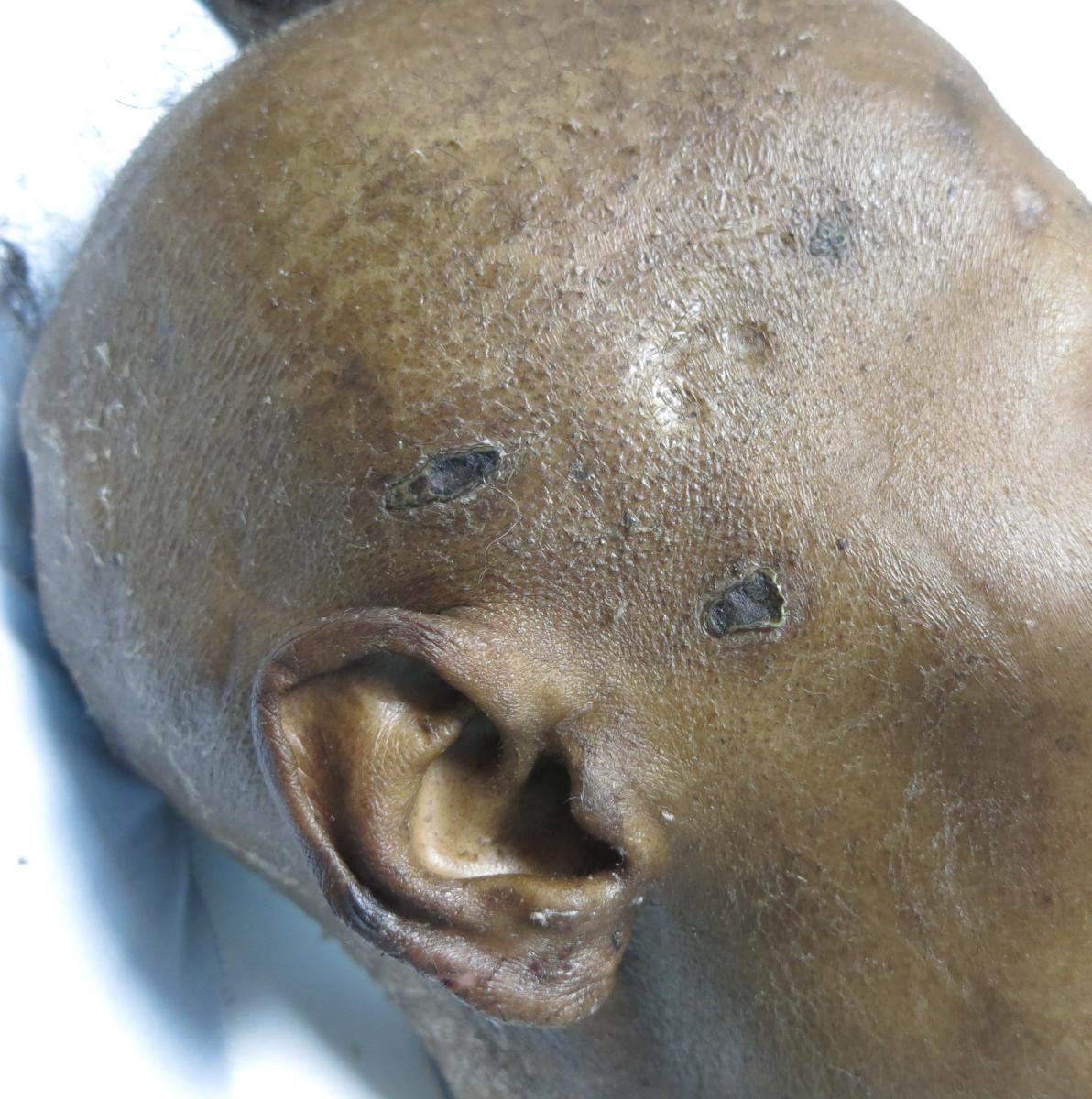
Figure 4. Well-demarcated slit-like and stellate ulcerations covered by eschar in the right temporal area. The presentation resembles the only other such case published in the literature.12

Figure 5. Acral calciphylaxis with an eschar-covered ulceration in the lateral aspect of right anterior foot.
Discussion. This case illustrates the current controversies in calciphylaxis diagnosis and treatment, emphasizing the gaps in knowledge regarding its pathophysiology. Calciphylaxis, also known as calcific uremic arteriopathy (CUA), is a rare ischemic vasculopathy seen mainly (although not exclusively) in patients with ESRD who are on HD. It presents with exquisitely painful erythema and induration in adipose-rich areas of the thighs, buttocks and abdomen that progresses to violaceous retiform purpura, cutaneous necrosis, and ulceration. Histologically, it is a small-vessel disease with calcification and thrombosis of dermal and subcutaneous arterioles and capillaries, and extravascular calcification. The condition heralds a poor prognosis in terms of survival, and no therapies have a proven effect on mortality.1
Among patients on HD, risk factors for calciphylaxis include diabetes mellitus, obesity, female gender, hypercoagulable states, and higher levels of serum calcium, phosphorus, and parathyroid hormone. Abnormalities in the calcium-phosphate product are not always present, and only a fraction of calciphylaxis patients demonstrate these alterations at presentation.2,3 Recent studies estimate the prevalence of the disease as 5.7 cases per 10,000 chronic HD patients.4 The associated morbidity and mortality is significant, with an estimated 1-year survival rate of only 45.8%5 and with mortality rates 2.5 to 3 times higher for calciphylaxis patients compared with the average in HD patients.4
Matrix Gla protein (MGP) is a potent inhibitor of vascular calcification originally described in animal models.6 The molecule activation depends on the phosphorylation and carboxylation of its amino acids. The latter process is highly dependent on the availability of vitamin K,7 the concentration of which is usually decreased in patients with chronic kidney disease.8 A significant correlation between the inactive form of MGP (dp-ucMGP) and a vascular calcification score was found in a cohort of HD patients; those treated with warfarin had significantly higher dp-ucMGP concentrations than untreated patients.9 A recent study showed increased lower-extremity calcification in patients receiving warfarin,10 and although there is a theoretical effect of supplementing vitamin K (30 mg weekly) to treat calciphylaxis, the safety and efficacy in humans is still unknown. An ongoing clinical trial is currently evaluating this issue.11 In patients needing anticoagulation, therapeutic alternatives are low-molecular-weight heparin with factor Xa monitoring (as used in our patient), continuous intravenous heparin infusion, aspirin, and apixaban.
Most cases of calciphylaxis present on the lower extremities below the knees. There are no data evaluating the frequency of acral distribution, but several case reports have focused on digital involvement. An exceedingly rare presentation is facial calciphylaxis, with only 1 prior study published in the literature to the best of our knowledge.12 In this case, as in our patient’s case, the patient presented with extensive widespread calcifications and died soon after the diagnosis as a result of gastrointestinal tract bleeding and sepsis. The slit-like and stellate appearance of the facial ulcers is similar in both cases. In our patient’s case, a possible explanation for the extensive lesions in atypical areas was the low vitamin K level as demonstrated by the patient’s borderline-elevated international normalized ratio (INR) throughout hospitalization. Of note, our patient received several courses of antibiotics that also might have depleted her vitamin K stores. Low levels of protein C and protein S have been documented in patients with calciphylaxis,13 but at the same time, reports have shown that lesions can heal despite persistently abnormal protein C and protein S levels.14
Calciphylaxis is both a clinical and histopathologic diagnosis. The advantage of skin biopsies is their ability to rule out alternative diagnosis, such as peripheral arterial disease, cholesterol embolization, vasculitis, and antiphospholipid antibody syndrome. However, given the need for deep samples, biopsies are usually low-yield and lack specificity. For the same reason, the benefits of a biopsy must be weighed against the risk of poor wound healing, formation of new lesions, and superimposed infection.
The largest calciphylaxis histopathology case series showed that the presence of both subcutaneous thrombotic occlusion and vascular calcification were the most significant findings associated with this condition, and only 18% of the samples showed evidence of classic features of arteriolar calcification.15 Detection of microcalcifications, often around eccrine glands, usually requires combining special calcium stains such as von Kossa and Alizarin red. Biopsies can be considered in atypical cases such as nonuremic calciphylaxis, those lacking classic cutaneous findings, immunosuppressed patients, or case in which a vasculitis is suspected. In cases where biopsy is not an option, clinical suspicion must support the diagnosis, since no imaging modality has been systematically evaluated. Finally, as in our patient’s case, evaluation by a dermatopathologist with experience in diagnosing calciphylaxis is key, since subtle features can be missed by less-experienced pathologists.
The dermatologic findings of WISN are often indistinguishable from calciphylaxis. However, the onset of WISN is rapid, usually within weeks of starting warfarin. It improves with administration of vitamin K and heparin, and clinical progression halts when warfarin is discontinued. Calciphylaxis typically develops months to years after warfarin initiation and generally does not improve with warfarin discontinuation alone. Consideration of calciphylaxis in the differential diagnosis has important therapeutic implications, since some of the medications used to treat autoimmune conditions can worsen calciphylaxis.16
A laboratory panel designed to rule out calciphylaxis mimickers such as vasculitis and acquired and inherited hypercoagulable states should include tests for prothrombin G20210A mutation, factor V Leiden, partial thromboplastin time, prothrombin time/INR, D-dimer, fibrinogen, lactate dehydrogenase, antineutrophil cytoplasmic antibodies, antiphospholipid antibodies, protein C, protein S, antithrombin III, cryoglobulins, cryofibrinogens, and both serum and urine electrophoresis.
Calciphylaxis treatment often requires a multidisciplinary approach with the use of different treatment modalities. The optimal treatment for calciphylaxis is still unknown, and the available therapies are based on studies with a low quality of evidence. In particular, the use intravenous sodium thiosulfate in HD patients is supported by numerous case reports, with questionable mortality benefit. No randomized controlled trial has ever examined the safety and efficacy of sodium thiosulfate in this setting. The mechanism of action is unknown, the doses used are empiric, and the optimal duration of therapy has not been determined. Additionally, it is an expensive drug in North America, with monthly costs of approximately US$10,000.17 Local wound care and pain management with opioids remain the cornerstone of therapy. The rationale for removing necrotic tissue is to prevent infection and to facilitate wound healing. In 1 retrospective nonmatched study, patient candidates for surgical debridement showed a 1-year survival rate of 62% compared with 27% for those without debridement.5 However, this study was not standardized to indications for debridement as well as patient comorbidities.
References:
- Brandenburg VM, Evenepoel P, Floege J, et al; ERA-EDTA Working Group on CKD-MBD and EUCALNET. Lack of evidence does not justify neglect: how can we address unmet medical needs in calciphylaxis? Nephrol Dial Transplant. 2016;31(8):1211-1219.
- Nigwekar SU, Zhao S, Wenger J, et al. A nationally representative study of calcific uremic arteriolopathy risk factors. J Am Soc Nephrol. 2016;27(11):3421-3429.
- Block GA. Prevalence and clinical consequences of elevated Ca x P product in hemodialysis patients. Clin Nephrol. 2000;54(4):318-324.
- Nigwekar SU, Solid CA, Ankers E, et al. Quantifying a rare disease in administrative data: the example of calciphylaxis. J Gen Intern Med. 2014;29(suppl 3):724-731.
- Weenig RH, Sewell LD, Davis MDP, McCarthy JT, Pittelkow MR. Calciphylaxis: natural history, risk factor analysis, and outcome. J Am Acad Dermatol. 2007;56(4):569-579.
- Luo G, Ducy P, McKee MD, et al. Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature. 1997;386(6620):78-81.
- Schurgers LJ, Cranenburg ECM, Vermeer C. Matrix Gla-protein: the calcification inhibitor in need of vitamin K. Thromb Haemost. 2008;100(4):593-603.
- Cranenburg ECM, Schurgers LJ, Uiterwijk HH, et al. Vitamin K intake and status are low in hemodialysis patients. Kidney Int. 2012;82(5):605-610.
- Delanaye P, Krzesinski J-M, Warling X, et al. Dephosphorylated-uncarboxylated matrix Gla protein concentration is predictive of vitamin K status and is correlated with vascular calcification in a cohort of hemodialysis patients. BMC Nephrol. 2014;15:145.
- Han KH, O'Neill WC. Increased peripheral arterial calcification in patients receiving warfarin. J Am Heart Assoc. 2016;5(1):e002665.
- Evaluation of vitamin K supplementation for calcific uremic arteriolopathy (VitK-CUA). ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/NCT02278692. Updated March 21, 2018. Accessed July 9, 2018.
- Mathur RV, Shortland JR, El Nahas AM. Calciphylaxis with facial involvement. Nephrol Dial Transplant. 2001;16(11):2256-2257.
- Rostaing L, el Feki S, Delisle M-B, et al. Calciphylaxis in a chronic hemodialysis patient with protein S deficiency. Am J Nephrol. 1995;15(6):524-527.
- Mehta RL, Scott G, Sloand JA, Francis CW. Skin necrosis associated with acquired protein C deficiency in patients with renal failure and calciphylaxis. Am J Med. 1990;88(3):252-257.
- Mochel MC, Arakaki RY, Wang G, Kroshinsky D, Hoang MP. Cutaneous calciphylaxis: a retrospective histopathologic evaluation. Am J Dermatopathol. 2013;35(5):582-586.
- Lee JL, Naguwa SM, Cheema G, Gershwin ME. Recognizing calcific uremic arteriolopathy in autoimmune disease: an emerging mimicker of vasculitis. Autoimmun Rev. 2008;7(8):638-643.
- AlBugami MM, Wilson JA, Clarke JR, Soroka SD. Oral sodium thiosulfate as maintenance therapy for calcific uremic arteriolopathy: a case series. Am J Nephrol. 2013;37(2):104-109.


