Newborn With Bicoronal Synostosis and Syndactyly of Hands and Feet
What's Your Diagnosis?
Sharpen Your Physical Diagnostic Skills
HISTORY
 African American girl born at 36 weeks’ gestation to 24-year-old primigravida via spontaneous vaginal delivery at a community hospital. Apgar scores, 7 at 1 minute and 9 at 5 minutes. Grossly normal placenta, with a 3-vessel cord. On the second day of life, infant required several
African American girl born at 36 weeks’ gestation to 24-year-old primigravida via spontaneous vaginal delivery at a community hospital. Apgar scores, 7 at 1 minute and 9 at 5 minutes. Grossly normal placenta, with a 3-vessel cord. On the second day of life, infant required several
minutes of blow-by oxygen for a desaturation event and subsequent transfer to a level III neonatal ICU for further monitoring.
Pregnancy complicated by a motor vehicle crash early in the first trimester; trauma series radiographs normal. Fetal ultrasonograms at 22 weeks identified a possible chordae tendinae in the left ventricle. Prenatal test results negative for HIV, group B streptococci, syphilis, and rubella.
Mother and infant both blood type O positive. Mother’s medical history unremarkable. No amily history of cranial malformations. No history of consanguinity.
PHYSICAL EXAMINATION
Vital signs normal. Birth weight, 354 g (57th percentile); length, 51.5 cm (80th percentile); head circumference, 31 cm (below the 3rd percentile). Infant noted to have brachycephaly with frontal bossing, midface hypoplasia, downslanting palpebral fissures, proptosis, disconjugate gaze, high-arched intact palate with small uvula, trapezoid-shaped mouth with normal philtrum. Patent nares. Open and large anterior and posterior fontanelles.
Syndactyly of both hands and feet. Limbs otherwise symmetrical and equal in length. Good brachial and femoral pulses. Hips symmetrical, without click. No dimple over the dorsal aspect of the lumbosacral spine. Normal external female genitalia. Skin normal, without rashes or lesions.
Lungs clear to auscultation. Heart rate and rhythm regular, without murmur. Bowel sounds present; abdomen soft, without masses or organomegaly.
WHAT’S YOUR DIAGNOSIS?
ANSWER: APERT SYNDROME
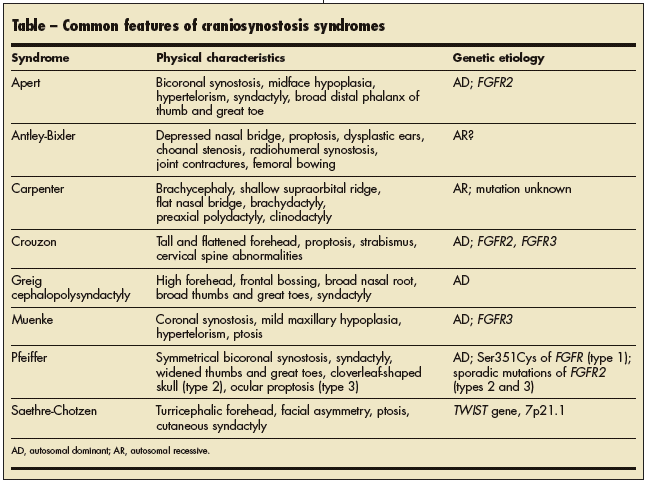
 First described by Wheaton1 in 1894 and later by Apert in 1906,2 Apert syndrome, or acrocephalosyndactyly, is an autosomal dominant malformation syndrome characterized by craniosynostosis, midface hypoplasia,
First described by Wheaton1 in 1894 and later by Apert in 1906,2 Apert syndrome, or acrocephalosyndactyly, is an autosomal dominant malformation syndrome characterized by craniosynostosis, midface hypoplasia,
syndactyly, and various visceral abnormalities. The syndrome presents in 15.5 per million live births.3
Although Apert syndrome has an autosomal dominant inheritance pattern, many cases are de novo mutations,4 and 1 case of germinal mosaicism has been reported.5 The syndrome is typically caused by 2 point mutations (S252W and P253R) and 2 Alu insertions in the fibroblast growth factor receptor 2 gene (FGFR2) located on chromosome 10q26.6 This gene is one of several that regulate growth and differentiation of mesenchymal and neuroectodermal cells and cranial suture fusion.7 The point mutations are paternal in origin.7 Apert, Crouzon, and Pfeiffer syndromes are often associated with advanced paternal age.8

CLINICAL MANIFESTATIONS
The distinctive facial features of Apert syndrome are acrocephaly (or oxycephaly) with bicoronal synostosis, maxillary hypoplasia, and high-arched palate (Figure 1). Cleft palate occurs in one-third of patients.9 The cranial malformations typically cause strabismus and hypertelorism. Patients may have malformations of the corpus callosum and limbic structures with ventriculomegaly that leads to hydrocephalus.10 Hearing impairment can result from persistent middle ear effusion or congenital ossicular chain fixation.11 Airway obstruction secondary to palatal defects is common12; in this case, it may have contributed to the patient’s desaturation event.
Syndactyly of all 4 extremities is characteristic of Apert syndrome (Figure 2). This finding is typically absent in other craniosynostoses. The term “mitten hand” is frequently used to describe the appearance of the fused soft tissues.
DIFFERENTIAL DIAGNOSIS
Craniosynostosis is seen in 1 in 2500 births.7 About 10% to 20% of cases ofcraniosynostosis are linkedto a genetic syndrome. Those implicated include Apert, Crouzon, and Pfeiffer syndromes; of these, Apert syndrome is the most common. Nongenetic causes of craniosynostosis include warfarin use during pregnancy and endocrine abnormalities, such as hyperthyroidism.13 In most cases, however, the cause is unknown.14
The genetic syndromes associated with craniosynostosis are characterized by skull base abnormalities, midface hypoplasia, and limb anomalies.14 The Table lists the more distinctive features of these syndromes. For an excellent pictorial review, see Smith’s Recognizable Patterns of Human Malformation.15
Acrocephaly is common in both Apert and Crouzon syndromes. The incidence of cleft palate and other craniofacial deformities is lower in Crouzon syndrome. Patients with Crouzon syndrome generally have a normal IQ15 and normal distal extremities, unlike those with Apert syndrome. One-third of patients with Crouzon syndrome have cervical spine abnormalities, which are rare in patients with Apert syndrome.
Patients with any of the 3 types of Pfeiffer syndrome have broad thumbs and great toes.15 Patients with type 1 typically have normal intelligence and survive to adulthood. Those with type 2 have the distinctive cloverleaf skull syndrome (kleeblattschädel deformity) with severe CNS complications and a poor prognosis. Those with type 3 lack the cloverleaf skull but have CNS defects and a similarly poor prognosis.
DIAGNOSIS
Apert syndrome can be confidently diagnosed based on the distinctive clinical appearance of the anomalies. The presence of the S252W or P253R point mutation in FGFR2 is more common in Apert syndrome than in other craniosynostoses and provides additional confidence in the diagnosis. The S252W mutation is more commonly associated with cleft palate and craniofacial deformity, whereas the P253R mutation is more commonly associated with severe syndactyly.7
Apert syndrome can be diagnosed in utero with ultrasonography; however, it is often missed on prenatal ultrasonographic examination, even in retrospect16—as it was in this case.
MANAGEMENT
Surgical correction of skull malformations is typically a 3-stage process: craniosynostosis release, midface advancement by Le Fort 3 osteotomy, and correction of hypertelorism.17 Correction of distal limb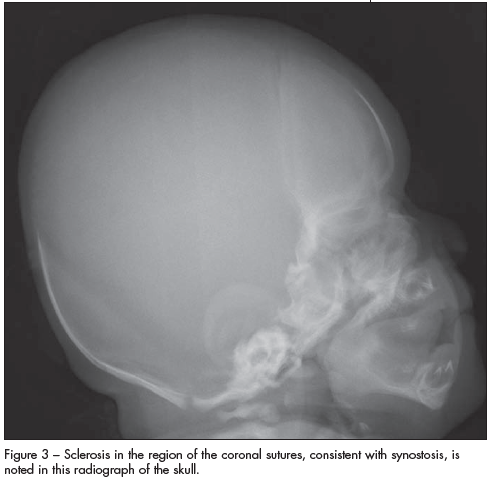 deformities depends on the severity of the underlying malformed bony architecture.
deformities depends on the severity of the underlying malformed bony architecture.
Because hearing impairment, frequent ear infections, and mental deficiencies may develop, children with Apert syndrome require regular follow-up tailored to their individual needs. This may involve several specialists, including an otorhinolaryngologist, plastic surgeon, neurosurgeon, and speech/language pathologist, in addition to the general pediatrician. Follow-up every 6 months for the first few years of life is typical in these patients.
OUTCOME OF THIS CASE
Genetic testing revealed the S252W mutation in FGFR2, consistent with a diagnosis of Apert syndrome. The infant had 46,XX karyotype. Results of blood chemistries and metabolic screening were normal.
A head ultrasonogram revealed normal midline brain structures, including a normal appearing corpus callosum. There was no evidence of intracranial hemorrhage. The cerebellum was unremarkable, and the intracranial internal carotid arteries and middle cerebral arteries were normal. Evaluation of hearing by evoked otoacoustic emissions was
normal bilaterally.
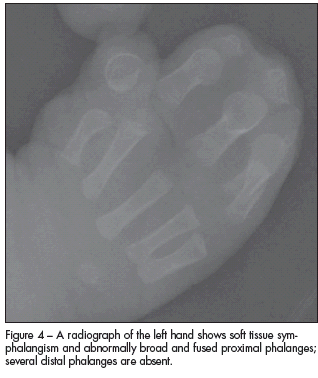 A skeletal survey revealed sclerosis in the region of the coronal sutures that confirmed synostosis (Figure 3). Radiographs of the hands demonstrated soft tissue symphalangism with abnormally broad and fused proximal phalanges; several distal phalanges were absent (Figure 4). Radiographs of the feet demonstrated bifid proximal first metatarsals and some hypoplastic proximal phalanges (Figure 5). The infant had a normal spine, 12 paired ribs, and a normal shoulder girdle and pelvis. The long bones of the upper and lower extremities were symmetrical and normally formed.
A skeletal survey revealed sclerosis in the region of the coronal sutures that confirmed synostosis (Figure 3). Radiographs of the hands demonstrated soft tissue symphalangism with abnormally broad and fused proximal phalanges; several distal phalanges were absent (Figure 4). Radiographs of the feet demonstrated bifid proximal first metatarsals and some hypoplastic proximal phalanges (Figure 5). The infant had a normal spine, 12 paired ribs, and a normal shoulder girdle and pelvis. The long bones of the upper and lower extremities were symmetrical and normally formed.
Findings from an abdominal ultrasonogram and chest radiograph were unremarkable. An echocardiogram revealed a hemodynamically insignificant patent ductus arteriosus and a patent foramen ovale appropriate for age. ECG showed normal sinus rhythm and normal
intervals and voltages for age.
The remainder of the infant’s hospital course was uneventful, and she was discharged at 1 week of life. Surgical correction of her cranial malformations is planned to begin at about 6 months of age. Possible revision of her distal extremities will be evaluated in the future.



