Evidence-Based Cardiovascular Disease Prevention: Challenges to Assessing Risk in Office Practice
ABSTRACT: Cardiovascular (CV) riskreduction regimens require comprehensive assessment, patient education, and followup, which can be difficult and time-consuming in a busy primary care practice. Moreover, compliance among patients at high risk can be poor. The use of evidencebased risk assessment checklists and patient education materials can enhance care and improve compliance; in addition, thorough documentation can ensure full reimbursement for services. The checklists are incorporated in a flow sheet, which is inserted in the front of the patient’s chart. Included are a CV risk reference guide, which documents risk factors; a Framingham global risk assessment scoring chart; a CV checklist that tracks the patient’s history, physical findings, and laboratory results; and a CV risk-reduction treatment plan. Patient education materials consist of checklists, tables, and charts that patients can use to monitor their progress.
Key words: cardiovascular disease, coronary heart disease, coronary artery disease, prevention
___________________________________________________________________________________________
Cardiovascular disease (CVD) affects 64.4 million Americans, or nearly 25% of the US population. Each year more than 1 million deaths are attributed to CVD, and treatment costs for CVD and stroke exceed $368 billion.1 The incidence of CVD is expected to increase as the US population ages and the prevalence of obesity and glucose intolerance rises. About 60 million Americans, or 21% of the population, are now 55 years or older,2 and by 2030, the number of older Americans is expected to reach 70 million.3
Quality-improvement initiatives for patients who have been hospitalized for cardiovascular (CV) events have improved outcomes. In addition, numerous trials have demonstrated the benefit of risk-reduction regimens in secondary prevention.4-11
However, for many patients, waiting to institute risk-reduction regimens until symptoms manifest or an event occurs may be too late; 50% of men and 64% of women who died of sudden cardiac death had no previous symptoms of heart disease.1 Thus, programs that emphasize prevention in the outpatient setting are essential.

Early identification of persons at high risk and modification of risk factors can slow or prevent the progression of CVD and reduce the incidence of serious CV events (Box i).12,13 But risk-reduction regimens require comprehensive assessment, patient education, and follow-up, which can be time-consuming. Several studies have shown that the risk of CV events is difficult to assess accurately in everyday practice.14-18 Furthermore, compliance among high-risk patients who are otherwise healthy can be poor (Box II), and striving to attain stringent goals can be discouraging for those at greatest risk. Finally, because there is no single International Classification of Disease (ICD) diagnostic code for CV risks, careful documentation of patient evaluation and treatment is essential for full reimbursement.
 In this article, I show how evidence-based risk assessment checklists and patient education materials can enhance care, improve compliance, increase efficiency of time spent with patients, and ensure complete reimbursement for services.19,20 The emphasis here is on the use of this strategy for primary and secondary prevention of CV events.
In this article, I show how evidence-based risk assessment checklists and patient education materials can enhance care, improve compliance, increase efficiency of time spent with patients, and ensure complete reimbursement for services.19,20 The emphasis here is on the use of this strategy for primary and secondary prevention of CV events.
RATIONALE FOR AN EVIDENCE-BASED APPROACH TO RISK REDUCTION
The application of evidencebased disease management guidelines improves patient outcomes21,22 and can optimize reimbursement for services. Nonetheless, the use of guidelines is far from universal in clinical practice. A survey of primary care physicians and cardiologists identified the following barriers to implementing ischemic heart disease guidelines:
• Inability of guidelines to accommodate the needs of individual patients.
• Difficulty of accessing guidelines.
• Heavy workloads associated with patients who have multiple disorders.23
Heavy workloads coupled with concern about scrutiny of coding and documentation can result in lost revenue. A study found that 33% of family practice physicians undercoded patient visits.24 At this rate of undercoding, a conservative estimate of annual revenue loss is $57,600 per physician.25 Chart-based checklists that improve documentation and save charting time can facilitate appropriate coding, resulting in increased revenue and decreased workload. This approach can also help you prepare for the chart review section of board recertification examinations.
My experience in a family practice that cares for 55,000 patients each year suggests that the use of flow sheets inserted in patient charts can overcome many barriers to implementation of guidelines. A pilot study conducted in my practice assessed 7 quality- of-care indicators—including measurement of hemoglobin A1c (HbA1c), annual retinoscopy, and foot examination—in patients aged 65 or older with type 2 diabetes.19 A flow sheet based on evidence-based medicine and practice guidelines was used for 36 of 109 patients evaluated, resulting in improved compliance with all 7 quality indicators.
In another study in my practice, a program designed for patients with depression evaluated the effects of a disease-management model—including a flow sheet, patient education, and diligent follow-up—on adherence.26 Typically, patient adherence to medical treatment of depression can be as low as 33% at the end of 3 months. Compliance can be even lower when lifestyle or behavioral changes are required.27,28 When a disease-management model was used, 66% of patients enrolled in the program remained on their treatment plan after 9 months of follow-up.26 A similar improvement in compliance with asthma-management quality indicators was shown when flow sheets were used in a study that included 122 asthmatic patients.20
CV RISK-REDUCTION TOOLS
CV risk flow sheet. Thhe flow sheet is a comprehensive tool for evaluation, evidence-based treatment, and monitoring of patients at risk for CVD; an example begins on page 1476 of this issue. The flow sheet is inserted in the front of the patient’s chart to facilitate assessment and monitoring of risk factors. You or a member of your staff can update the flow sheet as needed. The Cardiovascular Risk Reference Guide is formatted for quick documentation of risk factors, such as family history, cigarette smoking, and organ damage.
The second page of the flow sheet is a Framingham global risk assessment scoring chart for primary prevention that has been adapted to include criteria for secondary prevention in high-risk patients. This tool allows you to rapidly calculate and record a patient’s CV risk score. (Numerous Framingham-based risk calculators are also available via the Internet or for downloading on to personal digital assistants or computers. Sheridan and colleagues14 reviewed these products.) Although the Framingham global risk assessment scoring tool may not apply equally well for all gender, racial, and ethnic groups, it nevertheless remains the most widely tested approach to risk assessment.12,13,29-31
The third and fourth pages of the flow sheet are updated at every office visit; they comprise 2 checklists:
• The Cardiovascular Checklist tracks the patient’s history, physical findings, and laboratory test results.
• The Cardiovascular Risk-Reduction Treatment Plan is used to monitor evidence-based treatment decisions.
Patient education tool. This is designed to emphasize the reasons for risk modification and to improve compliance through communication of patient- specific information and goals. Patients can track their progress on the tool’s checklists, tables, and charts. The document describes the patient’s prescribed treatment plan, including the rationale for recommendations, and emphasizes the need for follow-up by providing a calendar on which scheduled appointments can be written. You (or a member of your staff) and your patient can then review progress at each visit.
RISK ASSESSMENT
The American Heart Association (AHA) recommends that all adults aged 20 years or older be screened regularly for CV risk factors.13 Risk assessment, including family history of coronary heart disease (CHD), smoking status, diet, alcohol intake, physical activity, blood pressure, body mass index, waist circumference, and pulse, should be updated at every routine office visit (at least every 2 years). The frequency of measurement of fasting serum lipoprotein and fasting blood glucose is based on the patient’s risk of dyslipidemia or diabetes, respectively; however, these tests should be performed at least every 5 years, or every 2 years if risk factors are present.
Global risk assessment. The Framingham Global Risk Assessment evaluates the 10-year risk of CHD in persons who have no clinical manifestations of the disease. Generally, this assessment assumes that CHD is defined as angina pectoris, recognized and unrecognized myocardial infarction (MI), coronary insufficiency (unstable angina), and CHD deaths. The scoring system has also been adapted to estimate the risk of “hard” CHD that excludes angina pectoris. Because the Framingham criteria for CHD are broader than those used in most trials, the number of patients identified as “at risk” for CHD using these criteria is likely to be higher than would be expected based on trial data.12
Points are assigned for each of a patient’s defined risk factors and added up to determine a total risk point score. This total point score is then used to determine absolute and relative risks of CHD in tables segmented by gender and age categories. The AHA emphasizes that adults 40 years and older should know their risk scores and what they mean.13
Patients may find the concept of absolute risk, the probability of CHD developing over a specified period, easier to understand than relative risk. However, it is useful to sort absolute risk scores into groups of persons at high, moderately above average, or average risk relative to those at low risk.12 A person of any age who does not smoke or have diabetes and who has the following test results is at low risk for CHD: blood pressure of less than 120 mm Hg systolic and 80 mm Hg diastolic, total cholesterol (TC) between 160 and 199 mg/dL (or low-density lipoprotein [LDL] cholesterol between 100 and 129 mg/dL), and high-density lipoprotein (HDL) cholesterol of at least 45 mg/dL for men and at least 55 mg/dL for women.12 Anyone with a history of CHD (previous MI, stroke, peripheral vascular disease, coronary artery bypass graft, or percutaneous coronary intervention) or diabetes is considered to be at high risk.9,12
The AHA guidelines suggest that the global assessment of 10-year risk of CHD be calculated at least every 5 years for all patients aged 40 years or older.13 However, I have found that by using CV risk flow sheets, this calculation can be done at every physical examination. Moreover, seeing a drop in overall risk after following a treatment plan can be a strong motivation for the patient.
Standard laboratory tests. Key tests for assessing CV risk include an ECG, serum lipid profiles, serum creatinine levels, and a hematocrit. The frequency of testing is determined by risk assessment and by clinical judgment.
Novel risk markers. In a number of studies, markers of inflammation showed promise as predictors of CV risk. Markers of macrophage activation, including C-reactive protein (CRP), intercellular adhesion molecules, neopterin, and matrix metalloproteinase-9 (MMP-9), were strongly correlated with progression of coronary artery disease.32 CRP levels may increase the predictive value of serum lipid values.33 Currently, however, there is no evidence to support the use of these tests in routine clinical practice. Elevated homocysteine, lipoprotein (a), and prothrombotic factors (fibrinogen) also have been shown to correlate with CV risk, but tests for these factors are not recommended for routine use.12,34-37
RISK FACTORS
The primary independent risk factors for CHD are cigarette smoking, elevated blood pressure, elevated serum TC and LDL cholesterol, low serum HDL cholesterol, diabetes mellitus, and advancing age.12 Most evidence from clinical trials, including the Framingham study, suggests that noncoronary CVD (stroke, chronic kidney disease [CKD], and peripheral arterial disease) shares the same primary risk factors.38
Hypertension is a strong predictor of CV events. In select cases, patients who have elevated blood pressure should be evaluated further to identify the cause of hypertension, such as sleep apnea, certain drugs, CKD, primary aldosteronism, renovascular disease, long-term corticosteroid therapy, Cushing syndrome, pheochromocytoma, coarctation of the aorta, and thyroid or parathyroid disease.29
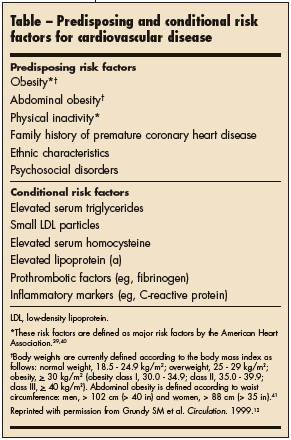 Other factors may further heighten the risk of CVD (Table).39-41 Although these factors may independently increase risk, the extent of their quantitative contribution to risk has not been determined.
Other factors may further heighten the risk of CVD (Table).39-41 Although these factors may independently increase risk, the extent of their quantitative contribution to risk has not been determined.
Among these risks are obesity and physical inactivity.12 Obesity, particularly abdominal obesity, predisposes patients to other risk factors, including diabetes, hypertension, and elevated serum cholesterol. Physical inactivity also increases the incidence of other risk factors.12,40-42
Psychosocial factors, including depression, are often overlooked contributors to CV risk. The association between depression and CVD was evaluated in 4493 Americans aged 65 years or older who were free of CVD at the start of the study.43 After 6 years of follow-up, for every 5-unit increase in the Depression Scale of the Center for Epidemiological Studies, the risk of CHD increased by 15% (P = .006). Among the patients with the highest cumulative mean depression scores, the risk of CHD increased by 40% over 6 years.43 Although treatment of depression is recommended to improve quality of life, no evidence indicates that such treatment reduces CV risk.
Nonmodifiable risk factors—such as age, ethnicity, family history, and diabetes—may affect the intensity of treatment for hypertension, dyslipidemia, and other factors. The absolute risk of a CV event increases substantially with advancing age, probably as a result of cumulative damage caused by the progression of atherosclerosis over time. The 10-year risk of CHD for an otherwise low-risk patient almost doubles between the ages of 45 years and 55 years based on age alone. The Framingham risk assessment score calculation weights age accordingly and thus can suggest more aggressive therapy for an elderly person than is warranted.12 Relative-risk estimates allow you to compare risk among patients of similar ages.12 However, the AHA emphasizes the importance of clinical judgment in determining who will benefit from aggressive therapy.13,44
The level of absolute risk associated with a given risk factor may differ among ethnic groups. The Framingham Risk Assessment Score is based on a population of mostly European origin. Be aware that patients from other ethnic groups will not be accurately assessed for absolute risk using this model. However, relativerisk estimates are thought to be reliable regardless of ethnicity.12
Diabetes confers the same level of risk as a history of a serious CV event.45 Therefore, patients with diabetes in whom CVD develops are at much greater risk for poor outcomes than are patients with similar risk profiles who do not have diabetes. Consider increasing the risk category to high for these patients.
Patients at risk for diabetes require careful follow-up.12 For patients who have diabetes, use fasting blood glucose and HbA1c values routinely to monitor glycemic control.
Although a family history of premature CHD is not modifiable, some of the familial tendency to CVD may reflect dietary and other lifestyle habits that are amenable to change. The latest National Cholesterol Education Program (NCEP) guidelines rank family history of CHD as an independent risk factor that modifies the intensity of LDL-lowering therapy.46 However, the degree of independence from other factors has not been clearly established.12
STRATEGIES TO REDUCE CV RISK
Appropriate lifestyle changes are recommended for all patients. If therapeutic lifestyle changes do not achieve risk-reduction goals, pharmacotherapy may be started.
Antiplatelet agents and anticoagulants. Aspirin therapy is recommended for both primary and secondary prevention of CV events. In primary prevention, dosages of 75 to 160 mg/d are recommended for patients with a 10-year risk of CHD of 10% or higher.13 For patients with coronary or other vascular disease, dosages of 75 to 325 mg/d are recommended. Clopidogrel and warfarin are options for patients who cannot tolerate aspirin. Consider warfarin dosed to an international normalized ratio of 2.0 to 3.0 in patients who have sustained an MI and in those who cannot take aspirin or clopidogrel.44
Antihypertensive agents. Lowering blood pressure with antihypertensive drugs reduces the complicationsof hypertension.29,47-53
Thiazide diuretics. Based on clinical data, including those from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT),51 the JNC 7 recommends thiazide diuretics as first-line pharmacologic intervention for elevated blood pressure.29 However, diuretics are rarely used as monotherapy in practice because most patients require multiple agents to reach target blood pressure levels.
Angiotensin-converting enzyme (ACE) inhibitors. These are the only antihypertensive agents with evidence-based indications for heart failure, MI, CVD, diabetes, CKD, and recurrent stroke prevention.29 Differences in efficacy among these agents are becoming more evident as clinical data accrue. The Heart Outcomes Prevention Evaluation (HOPE) trial demonstrated that ramipril added to standard treatment significantly reduced the relative risk of CV death, MI, or stroke in patients who were at high CV risk but were normotensive at study entry.54,55 Based on the results of the HOPE study, the AHA recommends that all patients who have sustained an MI be treated with an ACE inhibitor indefinitely. Long-term ACE inhibitor therapy is suggested for all other patients with coronary or other vascular disease unless it is contraindicated.44
However, data from studies with other ACE inhibitors have not substantiated that the results of the HOPE study reflect a class effect.52,56,57 Furthermore, an analysis of mortality among elderly patients who received an ACE inhibitor after an MI revealed that lower mortality rates were associated with ramipril compared with other ACE inhibitors.58 These findings are consistent with earlier results showing that shortterm therapy with ramipril after MI resulted in lower rates of in-hospital mortality and adverse nonfatal coronary and cerebrovascular events.59
ß-Blockers. The pharmacokinetics of -blockers differ widely, particularly in their relative affinities for ß1 -, ß2 -, and alpha-adrenergic receptor subtypes.60 The clinical relevance of these differences is not completely understood. Although many -blockers reduce mortality after MI, some data suggest that the nonselective agent carvedilol may provide a greater survival benefit in patients with heart failure. Not all ß -blockers are associated with survival, and differences in efficacy may be associated with different patient subtypes.60
Lipid-lowering agents. The AHA recommends statins alone or in combination with resins or fibrates to reach target lipid levels. For patients at higher risk, niacin, fibrates, and statins may be considered to raise HDL cholesterol levels.13,44
The NCEP Adult Treatment Panel III (ATP III) guidelines published in 2001 have recently been updated to include results from 5 clinical trials.46 These trial results support the ATP III target LDL cholesterol level of less than 100 mg/dL for patients at high risk (those with CHD or a CHD risk equivalent whose 10-year risk is greater than 20%). The results also support the inclusion of patients with diabetes mellitus in the high-risk category. In addition, based on results comparing intensive and moderate lipid lowering with statins, the ATP III update states that a target LDL cholesterol level of less than 70 mg/dL is a reasonable goal for patients at high risk for CHD.
Although the ATP III falls short of changing the recommended target level for patients with moderately high risk (2 or more risk factors and a 10-year risk of 10% to 20%) from LDL cholesterol levels of less than 130 mg/dL, reducing the target to less than 100 mg/dL is reasonable based on the data, particularly that of the Anglo-Scandinavian Cardiac Outcomes Trial Lipid-Lowering Arm.61
When LDL-lowering drug therapy is used in high-risk or moderately high-risk patients, the intensity of therapy should be sufficient to achieve a minimum of a 30% to 40% reduction in LDL cholesterol levels.62 Although statins are generally well tolerated, the incidence of rhabdomyolysis is dose-dependent, and the benefit of high-dose therapy needs to be weighed against the risk of this adverse event.
Differences in efficacy and safety have been reported among statins. At fixed or allowable dosages, rosuvastatin, atorvastatin, and simvastatin reduce LDL cholesterol to a significantly greater degree than do other statins.63 Although statins are generally safe and well tolerated, clinically important differences in safety have been reported for specific agents within this class.64,65

REFERENCES:
1. American Heart Association. Heart Disease and Stroke Statistics—2004 Update. Dallas: American Heart Association; 2003.
2. US Census Bureau. Table 1. Population by age, sex, race and Hispanic origin: March 2002. Available at: http://www.census.gov/population/www/socdemo/age/ppl-167.html. Accessed December 14, 2004.
3. Federal Interagency Forum on Aging. Older Americans 2000: key indicators of well-being. Available at: http://www.agingstats.gov/chartbook2000/population.html. Accessed December 14, 2004.
 4. Williams MA, Fleg JL, Ades PA, et al. Secondary prevention of coronary heart disease in the elderly (with emphasis on patients = 75 years of age). An American Heart Association Scientific Statement from the Council on Clinical Cardiology Subcommittee on Exercise, Cardiac Rehabilitation, and Prevention. Circulation. 2002;105:1735-1743.
4. Williams MA, Fleg JL, Ades PA, et al. Secondary prevention of coronary heart disease in the elderly (with emphasis on patients = 75 years of age). An American Heart Association Scientific Statement from the Council on Clinical Cardiology Subcommittee on Exercise, Cardiac Rehabilitation, and Prevention. Circulation. 2002;105:1735-1743.
5. Hunt SA, Baker DW, Chin MH, et al. ACC/AHA Guidelines for the Evaluation and Management of Chronic Heart Failure in the Adult: Executive Summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). Circulation. 2001;104:2996-3007.
6. Julius S, Alderman MH, Beevers G, et al. Cardiovascular risk reduction in hypertensive black patients with left ventricular hypertrophy. The LIFE study. J Am Coll Cardiol. 2004;43:1047-1055.
7. Koennecke HC. Secondary prevention of stroke. A practical guide to drug treatment. CNS Drugs. 2004;18:221-241.
8. Gluckman TJ, Baranowski B, Ashen MD, et al. A practical and evidence-based approach to cardiovascular disease risk reduction. Arch Intern Med. 2004;164:1490-1500.
9. Ketola E, Sipilä R, Mäkelä M. Effectiveness of individual lifestyle interventions in reducing cardiovascular disease and risk factors. Ann Med. 2000; 32:239-251.
10. Critchley J, Capewell S. Smoking cessation for the secondary prevention of coronary heart disease [review]. Cochrane Database Syst Rev. 2003(4): CD003041.
11. Hennekens CH, Sacks FM, Tonkin A, et al. Additive benefits of pravastatin and aspirin to decrease risks of cardiovascular disease. Randomized and observational comparisons of secondary prevention trials and their meta-analyses. Arch Intern Med. 2004;164:40-44.
12. Grundy SM, Pasternak R, Greenland P, et al. Assessment of cardiovascular risk by use of multiple- risk-factor assessment equations. A statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation. 1999;100:1481-1492.
13. Pearson TA, Blair SN, Daniels SR, et al. AHA guidelines for primary prevention of cardiovascular disease and stroke: 2002 update. Consensus Panel guide to comprehensive risk reduction for adult patients without coronary or other atherosclerotic vascular diseases. Circulation. 2002;106: 388-391.
14. Sheridan S, Pignone M, Mulrow C. Framingham-based tools to calculate the global risk of coronary heart disease. A systematic review of tools for clinicians. J Gen Intern Med. 2003;18:1039-1052.
15. Montgomery AA, Fahey T, Mac Kintosh C, et al. Estimation of cardiovascular risk in hypertensive patients in primary care. Br J Gen Pract. 2000;50: 127-128.
16. Grover SA, Lowensteyn I, Esrey KL, et al. Do doctors accurately assess coronary risk in their patients? Preliminary results of the coronary health assessment study. BMJ. 1995;310:975-978.
17. Friedmann PD, Brett AS, Mayo-Smith MF. Differences in generalists’ and cardiologists’ perceptions of cardiovascular risk and the outcomes of preventive therapy in cardiovascular disease. Ann Intern Med. 1996;124:414-421.
18. Meland E, Laerum E, Lehmann EH. Assessment of coronary heart disease risk, II: a clinical multicentre study of general practitioners’ risk assessment. Fam Pract. 1994;11:122-126.
19. Ruoff G, Gray LS. Using a flow sheet to improve performance in treatment of elderly patients with type 2 diabetes. Fam Med. 1999;31:331-336.
20. Ruoff G. Effects of flow sheet implementation on physician performance in the management of asthmatic patients. Fam Med. 2002;34:514-517.
21. Ofman JJ, Badamgarav E, Henning JM, et al. Does disease management improve clinical and economic outcomes in patients with chronic diseases? A systematic review. Am J Med. 2004;117:182-192.
22. Mehta RH, Montoye CK, Faul J, et al, on behalf of the American College of Cardiology Guidelines Applied in Practice Steering Committee. Enhancing quality of care for acute myocardial infarction: shifting the focus of improvement from key indicators to process of care and tool use. The American College of Cardiology Acute Myocardial Infarction Guidelines Applied in Practice Project in Michigan: Flint and Saginaw Expansion. J Am Coll Cardiol. 2004;43:2166-2173.
23. Powell-Cope GM, Luther S, Neugaard B, et al. Provider-perceived barriers and facilitators for ischaemic heart disease (IHD) guideline adherence. J Eval Clin Pract. 2004;10:227-239.
24. King MS, Sharp L, Lipsky MS. Accuracy of CPT evaluation and management coding by family physicians. J Am Board Fam Pract. 2001;14:184-192.
25. Hill E. How to get all the 99214s you deserve. Fam Pract Manag. 2003;10(9):31-36.
26. Ruoff G. A method that dramatically improves patient adherence to depression treatment. J Fam Pract. 2005;54:846-852.
27. Bull SA, Hu XH, Hunkeler EM, et al. Discontinuation of use and switching of antidepressants: influence of patient-physician communication. JAMA. 2002;288:1403-1409.
28. Haynes RB, MacDonald HP, Garg AX. Helping patients follow prescribed treatment: clinical applications. JAMA. 2002;288:2880-2883.
29. National Heart, Lung, and Blood Institute, National Institutes of Health, US Department of Health and Human Services. The Seventh Report of the Joint National Committee. Available at: http://www.nhlbi.nih.gov/guidelines/hypertension/. Accessed October 28, 2004.
30. Hung J, for the Medical Issues Committee of the National Heart Foundation of Australia. Aspirin for cardiovascular disease prevention. Med J Aust. 2003;179:147-152.
31. Hayden M, Pignone M, Phillips C, Mulrow C. Aspirin for the primary prevention of cardiovascular events: a summary of the evidence for the US Preventive Services Task Force. Ann Intern Med. 2002;136:161-172.
32. Zouridakis E, Avanzas P, Arroyo-Espliguero R, et al. Markers of inflammation and rapid coronary artery disease progression in patients with stable angina pectoris. Circulation. 2004;110:1747-1753.
33. Bassuk SS, Rifai N, Ridker PM. High-sensitivity C-reactive protein: clinical importance. Curr Probl Cardiol. 2004;29:439-493.
34. Malinow MR, Bostom AG, Krauss RM. Homocyst(e)ine, diet, and cardiovascular diseases. A statement for healthcare professionals from the Nutrition Committee, American Heart Association. Circulation. 1999;99:178-182.
35. Montalescot G, Collet JP, Choussat R, Thomas D. Fibrinogen as a risk factor for coronary heart disease. Eur Heart J. 1998;19(suppl H):H11-H17.
36. Bostom AG, Cupples LA, Jenner JL, et al. Elevated plasma lipoprotein (a) and coronary heart disease in men aged 55 years and younger: a prospective study. JAMA. 1996;276:544-548.
37. Fruchart JC, Nierman MC, Stroes ESG, et al. New risk factors for atherosclerosis and patient risk assessment. Circulation. 2004;109(suppl III): III15-III19.
38. Smith SC, Milani RV, Arnett DK, et al. Atherosclerotic Vascular Group Disease Conference. Writing Group II: risk factors. Circulation. 2004;109: 2613-2616.
39. Eckel RH. Obesity and heart disease. A statement for the healthcare professionals from the Nutrition Committee, American Heart Association. Circulation. 1997;96:3248-3250.
40. Fletcher GF, Balady G, Blair SN, et al. Statement on exercise: benefits and recommendations for physical activity programs for all Americans. A statement for healthcare professionals by the Committee on Exercise and Cardiac Rehabilitation of the Council on Clinical Cardiology, American Heart Association. Circulation. 1996;94:857-862.
41. NHLBI Obesity Education Initiative Expert Panel. Clinical Guidelines on Identification, Evaluation, and Treatment of Overweight and Obesity in Adults: The Evidence Report. Bethesda, Md: National Institutes of Health, National Heart, Lung, and Blood Institute; 1998. NIH No. 98-4083.
42. US Department of Health and Human Services. Physical Activity and Health. A Report of the Surgeon General. Executive Summary. Atlanta: Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion; 1996.
43. Ariyo AA, Haan M, Tangen CM, et al, for the Cardiovascular Health Study Collaborative Research Group. Depressive symptoms and risks of coronary heart disease and mortality in elderly Americans. Circulation. 2000;102:1773-1779.
44. Smith SC Jr, Blair SN, Bonow RO, et al. AHA/ACC guidelines for preventing heart attack and death in patients with atherosclerotic cardiovascular disease: 2001 update: a statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation. 2001;104:1577-1579.
45. Haffner SM, Lehto S, Rönnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339:229-234.
46. National Cholesterol Education Program. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) Executive Summary. 2001. Available at: http://rover.nhlbi.nih.gov/guidelines/cholesterol/atp_iii.htm. Accessed October 28, 2004.
47. Black HR, Elliott WJ, Grandits G, et al. Principal results of the controlled onset verapamil investigation of cardiovascular end points (CONVINCE) trial. JAMA. 2003;289:2073-2082.
48. Wing LMH, Reid CM, Ryan P, et al. A comparison of outcomes with angiotensin-convertingenzyme inhibitors and diuretics for hypertension in the elderly. N Engl J Med. 2003;348:583-592.
49. Neal B, MacMahon S, Chapman N, et al. Effects of ACE inhibitors, calcium antagonists, and other blood-pressure-lowering drugs: results of prospectively designed overviews of randomised trials. Lancet. 2000;355:1955-1964.
50. Dahlöf B, Devereux RB, Kjeldsen SE, et al. Cardiovascular morbidity and mortality in the losartan intervention for endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet. 2002;359:995-1003.
51. The ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA. 2002;288: 2981-2997.
52. PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6105 individuals with previous stroke or transient ischaemic attack [published corrections appear in Lancet. 2001;358:1556 and Lancet. 2002;359:2120]. Lancet. 2001;358:1033-1041.
53. Psaty BM, Smith NL, Siscovick DS, et al. Health outcomes associated with antihypertensive therapies used as first-line agents: a systematic review and meta-analysis. JAMA. 1997;277:739-745.
54. The Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting- enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N Engl J Med. 2000;342: 145-153.
55. Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000; 355:253-259.
56. Pitt B, O’Neill B, Feldman R, et al. The quinapril ischemic event trial: evaluation of chronic ACE inhibitor therapy in patients with ischemic heart disease and preserved left ventricular function. Am J Cardiol. 2001;87:1058-1063.
57. Prevention of events with angiotensin converting enzyme inhibition (PEACE) trial investigators. Angiotensin-converting-enzyme inhibition in stable coronary artery disease. N Engl J Med. 2004;351: 2058-2068.
58. Pilote L, Abrahamowicz M, Rodrigues E, et al. Mortality rates in elderly patients who take different angiotensin-converting enzyme inhibitors after acute myocardial infarction: a class effect? Ann Intern Med. 2004;141:102-112.
59. Wienbergen H, Schiele R, Gitt AK, et al; MITRA PLUS Study Group. Impact of ramipril versus other angiotensin-converting enzyme inhibitors on outcome of unselected patients with ST-elevation acute myocardial infarction. Am J Cardiol. 2002;90: 1045-1049. 60. Reiter MJ. Cardiovascular drug class specificity: beta-blockers. Prog Cardiovasc Dis. 2004;47:11-33.
61. Sever PS, Dahlöf B, Poulter NR, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lowerthan- average cholesterol concentrations, in the Anglo-Scandinavian cardiac outcomes trial—lipid lowering arm (ASCOT-LLA): a multicentre randomised controlled trial. Lancet. 2003;361:1149-1158.
62. Grundy SM, Cleeman JI, Merz CNB, et al, for the Coordinating Committee of the National Cholesterol Education Program. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation. 2004;110:227-239.
63. Chong PH. Lack of therapeutic interchangeability of HMG-CoA reductase inhibitors. Ann Pharmacother. 2002;36:1907-1917.
64. Pasternak RC, Smith SC Jr, Bairey-Merz CN, et al; American College of Cardiology; American Heart Association; National Heart, Lung and Blood Institute. ACC/AHA/NHLBI clinical advisory on the use and safety of statins. Circulation. 2002;106:1024-1028.
65. Staffa JA, Chang J, Green L. Cerivastatin and reports of fatal rhabdomyolysis. N Engl J Med. 2002; 346:539-540.
66. Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med. 1999;340:115-126.
67. Landmesser U, Hornig B, Drexler H. Endothelial function. A critical determinant in atherosclerosis? Circulation. 2004;109(suppl II):II-27–II-33.
68. Kavey RE, Daniels SR, Lauer RM, et al. American Heart Association guidelines for primary prevention of atherosclerotic cardiovascular disease beginning in childhood. Circulation. 2003;107: 1562-1566.
69. Schroeder K, Fahey T, Ebrahim S. Interventions for improving adherence to treatment in patients with high blood pressure in ambulatory settings. Cochrane Database Syst Rev. 2004(2):CD004804.
70. Murray MD, Young JM, Morrow DG, et al. Methodology of an ongoing, randomized, controlled trial to improve drug use for elderly patients with chronic heart failure. Am J Geriatr Pharmacother. 2004;2:53-65.
71. Shuttleworth A. Improving drug concordance in patients with chronic conditions. Nursing Times. 2004;100:28-29.
72. Butler J, Khadim G, Belue R, et al. Tolerability to -blocker therapy among heart failure patients in clinical practice. J Card Fail. 2003;9:203-209.


