Dementia Screening Tools for the Primary Care Physician
At the conclusion of this activity, participants should be able to:
1. Explain the role of neuropsychological testing in screening for dementia in older patients.
2. Describe mental status screening tests used in office testing.
3. Discuss interpretive guidelines for the Mini-Mental State Examination, tests of word list generation, and the Clock Drawing Test.
4. Recognize the sensitivity and specificity of screening tests used for detection of dementia.
________________________________________________________________________________________________________________
INTRODUCTION
The prevalence of dementia increases with age and affects approximately 5- 8% of individuals over age 65, 15-25% of individuals over age 75, and approximately 25-50% of individuals over age 85.1 The most common type of dementia is still thought to be Alzheimer’s disease (AD). However, a review of the recent scientific literature has shown that there can be considerable overlap with respect to the neuropathology associated with any single dementia subtype.2 From a clinical perspective, the diagnostic criteria for some of the dementia syndromes have recently been revised. Therefore, the contribution of a host of other dementing disorders, such as vascular dementia (VaD), Lewy body dementia (LBD), and frontotemporal dementia (FTD), need to be factored into the clinical decision-making process.
In addition to the primary neurodegenerating dementia syndromes, a number of general medical conditions including structural lesions (primary or secondary brain tumor, subdural hematoma, normalpressure hydrocephalus), head trauma, endocrine conditions (hypothyroidism, hypercalcemia, hypoglycemia), nutritional conditions (deficiency of vitamin B12 or folate), infections (HIV, neurosyphilis), renal and hepatic dysfunction, and effects of medications can all be associated with cognitive deficits, and can constitute serious comorbid factors that complicate the clinical expression of a dementia. Not to be forgotten is the tremendous caregiving and financial burden associated with dementia. The effect of alcohol on cognition is controversial and must also be factored into the diagnostic equation.3
Dementia is frequently underdiagnosed in the community. A large cross-sectional study of primary care physicians’ charts found that in 65% of cases of dementia, there was no diagnosis mentioned in the chart.4 Treatments for the dementias in the form of medication designed to slow the course of the illness, and supportive psychological services designed to lessen caregiving burden are available and have been shown to be effective.5 The potential harm of not diagnosing dementia in patients includes missed opportunities for the application of available treatments, participation in research, advance care planning, and support of caregivers. Therefore, both an early diagnosis and a cogent characterization of cognitive strengths and weaknesses will enable patients and families to receive proper medical and psychological care they deserve and make informed decisions.
The U.S. Preventive Services Task Force6 does not endorse routine screening for dementia in asymptomatic older adults. However, it does recommend that clinicians should assess cognitive function whenever cognitive impairment is suspected based on direct observation, patient report, or concerns raised by family members. In this article, the authors review some of the literature and data on four commonly administered neuropsychological tests: the Mini- Mental State Examination (MMSE); tests of letter (letters “FAS”) and category (“animals”) word list generation (WLG); and the Clock Drawing Test (CDT). The overall purpose of this article is to show how primary care physicians can integrate these tests into their clinical practice. Our goals are to show how to interpret the data obtained from these tests to achieve more targeted clinical decision making for patients with suspected dementia.
THE MINI-MENTAL STATE EXAMINATION
The MMSE7 is the most frequently administered neuropsychological screening measure. Although the MMSE was not necessarily intended to be used for the identification of dementia, it is now widely employed as the frontline measure to assess cognitive status in older adults. As originally suggested,7 a score at or below 24 has been the point where cognitive impairment might be present.8 However, subsequent research has indicated that a score at or below 26 provides a better tradeoff between sensitivity (ie, the ability to correctly identify those individuals who are, in fact, cognitively impaired) versus specificity (ie, the ability to correctly identify those individuals who are cognitively intact).9
Since the publication of the MMSE, research has suggested that patients with “cortical” dementia such as AD display differential impairment on tests of memory and language, whereas patients with “subcortical dementia,"10 such as VaD associated with periventricular and deep white matter alteration, produce greater impairment on tests of executive control and motor/visuoconstruction.2,11 However, despite the popularity of the MMSE, relatively few studies have examined its clinical utility beyond the use of the summary score to identify individuals with dementia. van Gorp and colleagues12 administered the MMSE to patients with AD, VaD, and a normal control group and found that the two dementia groups did not differ on the summary MMSE score. However, Brandt, Folstein, and Folstein13 compared patients with AD and Huntington’s disease (HD), a subtype of subcortical dementia, on the MMSE and found that the patients with HD had more difficulty performing the serial subtraction task (a test associated with executive control), while the patients with AD had more difficulty recalling the three words and were more disoriented to the current date.
Jefferson and colleagues14 studied patients with suspected dementia who were assessed at an outpatient memory clinic. Patients meeting diagnostic criteria for AD15 and possible/probable VaD16 were studied. In addition to the MMSE summary score, between-groups differences were assessed on seven MMSE subscales. These subscales are listed below:
• Temporal Orientation Index—The Temporal Orientation Index (range 0-5) consisted of the five items that assess orientation to time (ie, year, season, date, day, and month).
• Physical Orientation Index—The Physical Orientation Index (range 0-5) consisted of the five items that assess orientation to place (ie, state, county, town, hospital, and floor).
• Total Orientation Index—The Total Orientation Index (range 0-10) was constructed by summing the Temporal and Physical Orientation subscales.
• Language Index—The Language Index (range 0-4) consisted of those items purported to assess language abilities (ie, naming of the watch and pen, repeating the phrase “no ifs, ands, or buts,” and following the written command to “close your eyes”).
• Memory Index—The Memory Index (range 0-3) was comprised of how many of the three words patients recalled after a short delay.
• Attention/Concentration (Working Memory Index)— The Working Memory Index (range 0-8) consisted of the spelling “world” backwards task and carrying out the three-step command. The three-step command was included in the Attention/Concentration Index as opposed to the Language Index because it correlated more strongly with performance on the “world” backwards task (P < 0.090), as compared to the language index tasks (ie, naming, repeating, and following a written command) (P < 0.320).
• Motor Index—The Motor Index (range 0-2) consisted of the two items on the MMSE that require a motor response (ie, copying the polygon and the production of a sentence).
As seen in Table I, the two dementia groups did not differ in terms of the total MMSE score. However, when patients with AD and VaD were compared, the AD group obtained significantly lower scores on the Temporal Orientation Index (Z = -2.21; P < 0.027) and the Memory Index (Z = -2.09; P < 0.037). By contrast, patients with VaD obtained lower scores on the Motor/Constructional Index (Z = -2.58; P < 0.001) and the Attention/Concentration Index (Z = -3.50; P < 0.001).
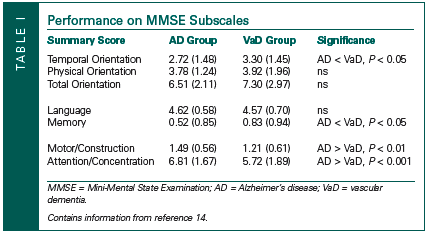
These data suggest that greater specificity regarding the cognitive deficits that may underlie the presentation of a dementing illness may be possible by paying attention to performance on individual MMSE test items. Thus, an analysis where patients fail or lose points on the MMSE can result in the articulation of important clinical issues, perhaps earlier in the course of the evaluation process, and may provide a means for more sophisticated treatment and/or neuropsychological referral questions.
The MMSE is often used in retirement communities and nursing homes to assess for alterations in mental status. The data reported by Jefferson and colleagues14 suggest that while summary scores may not change over time, alterations in the distribution of the errors produced on the MMSE could signal the presence of newly developing medical problems.
WORD LIST GENERATION
Another popular neuropsychological test is to ask patients to generate examples from either restricted phonemic categories (ie, the letters “F, A, S”) and/or broad semantic categories (animals, fruits, or vegetables). On these tests patients are generally given 60 seconds to generate their output. On WLG tests employing the letters “F, A, and S,” output is generally summed across the three letters. There is now considerable evidence to suggest that performance on letter WLG tests is a skill highly related to the integrity of the left dorsolateral frontal lobe, whereas category WLG is related to the integrity of the left temporal lobe.2,17-19 Normative informative for the “FAS” letter and “animal” category WLG tests can be found in Lezak.9 However, generally speaking, depending on the educational/occupational history, patients can be expected to produce 40 responses on the “FAS” WLG task and 20 responses on the “animals” WLG task. Carew at al17 administered the “FAS” and “animal” WLG tasks to patients with mild AD and VaD. Patients diagnosed with subcortical VaD generated fewer responses on the letter WLG task than patients diagnosed with AD (P < 0.001) (Table II). These data are consistent with research showing that other dementia groups presenting with subcortical neuropathology produce less output on letter WLG tasks as compared to patients with AD.20

Equally as important, and perhaps more interesting, Carew et al17 were also interested in whether patients with dementia were able to use grouping or clustering strategies to help in the generation of their output on the “animal” WLG task. For example, when young and well-elderly participants are given 60 seconds to generate “all the names of animals you can think of,” output tends to be clustered on the basis of well-known categories (ie, “lion, tiger, horse, cow,” etc).21 An example of this is also found in Table II. The number of responses for the two types of patients with dementia is basically the same (ie, both types of patients generated approximately 6-7 responses). However, further inspection of the output generated by the patient with VaD clearly shows that this patient was able to effectively group or cluster their output. For example, the patient with VaD generated two equines, followed by two felines, followed by two canines, followed by two farm-/agricultural-related animals.
It is commonly understood that all tests of WLG rely upon a certain amount of “frontal lobe” capacity. However, when asked to “generate all of the names of animals you can think of,” the task is made more manageable to the extent that one is able to cluster one’s responses into logical categories. Carew and colleagues17 have suggested that clustering strategies on the “animal” WLG provides a cognitive measure of language- related ability. Language-related deficits, along with deficits in memory, are hallmark features of AD. Thus, the authors have suggested that clustering/grouping strategies on the “animal” WLG corroborated by with AD but a subcortical dementia such as VaD associated with subcortical white matter alterations seen on magnetic resonance imaging (MRI) scans.
THE CLOCK DRAWING TEST
There are many variations in how the CDT is administered and scored. The most commonly used version of the CDT is modeled after Goodglass and Kaplan.22 Their version of the CDT contains two conditions: clock drawing to command and clock drawing to copy. Patients are provided with an 8.5 x 11–inch paper (presented lengthwise or landscape). The paper is folded in half. One half of the paper is blank. In the first clock drawing condition (clock drawing to command), patients are asked to “draw the face of a clock, put in all the numbers, and set the hands to read 10 after 11.” Immediately following the command condition, the examiner turns the paper over. The patient sees a model of a clock with hands set for “10 after 11.” In the second, clock drawing to copy condition, the patient is simply asked to copy the model.
Clock drawing has rich history in neurology. Goodglass and Kaplan22 originally employed clock drawing to assess posterior cortical functioning. Their innovation, however, was to include a copy condition and to ask patients to set the hands for “10 after 11.” They demonstrated that specific patterns of impairment in clock drawing in either the command or copy conditions could be seen, depending on the nature and location of the patient’s brain damage.22
Over the last 15 years, there is increasing interest in the CDT as a means to screen for dementia. A complex description of this literature is beyond the scope of this article. However, in previous research, Libon et al23 and Cosentino and colleagues24 have found the CDT to be a very powerful tool to assess executive functioning in dementia. Royall and colleagues25 also provide evidence to suggest that clock drawing provides an excellent measure of the certain types of executive function deficits (ie, frontal lobe) associated with dementia.
Using the Goodglass and Kaplan22 administration instructions for clock drawing, Libon and colleagues23 devised a 10-point scoring system and assessed clock drawing performance in patients with AD and VaD. Several key findings emerged. Patients with VaD make almost twice as many errors as patients with AD. Second, for patients with AD, fewer errors were made in the copy as compared to the command condition (ie, there is improvement from command to copy). By contrast, for patients with VaD, there was no statistically significant reduction in errors from the command and copy conditions. Research comparing patients with AD and VaD has consistently shown that total errors summed across both clock-drawing conditions correlate with poor performance on other executive function tests, including output on the “FAS” WLG test.23,24
INTERPRETATIVE GUIDELINES
Table III offers some broad interpretive guidelines for the neuropsychological tests discussed above. Each test will be reviewed in turn.

The Mini-Mental State Examination
There are three important bits of information that can be derived from the MMSE. First, the total summary score can be used to assess general mental status. However, on the basis of our clinical experience, a cut score of 24 is too generous for many patients. We have found that very mild cognitive disorders are often present in patients who consistently present with a borderline MMSE score of 26-24. A MMSE score within this borderline range could signal the presence of a prodromal dementia. Another possibility is that a treatable or occult medical problem may be present and responsible for a subtle compromise in cognition.
In established patients with a dementia, examine the distribution of errors. As discussed above, greater problems on the orientation versus the executive control (ie, spelling “world” backwards, subtracting serial 7’s) test items has been correlated with poor performance on tests of memory.14 Thus, in a patient where a diagnosis of dementia has been established or is suspected, the profile of greater disorientation versus executive/motor may point toward AD. An opposite profile (ie, greater problems on executive/motor test items versus orientation test items) suggests the presence of a dysexecutive syndrome. Executive control deficits could be due to the presence of subcortical neuropathology or a potentially treatable or manageable comorbid medical condition. In an established patient with dementia, a change in the pattern of errors on the MMSE could be caused by a variety of factors. For example, the dementia may now be progressing and/or new medical comorbidities might be present.
Tests of Word List Generation
As discussed above, cut scores on tests of letter and the “animal” WLG task are 40 and 20, respectively, depending on educational and occupational history. In addition to these cut scores, there are two other bits of information that should be examined. First, is there differential impairment on one WLG task versus the other? Poorer performance on the letter WLG task may signal the presence of executive function deficits, whereas poorer performance on the “animal” WLG might suggest a language-related disturbance. A dysexecutive syndrome may be associated with the presence of subcortical neuropathology or some other comorbid medical condition known to affect cognition. Poorer performance on the “animal” WLG task might be due to a compromise in language functioning, in which case it would be reasonable to suspect AD. Second, look for the use of clustering on the “animal” WLG task. Good use of a clustering strategy, even if output is reduced, suggests that certain language- related skills may be relatively intact. In such a situation, a dementia other than AD may be present.
The Clock Drawing Test
A wide variety of parameters could be considered. First, look for obvious signs of a motor/dysexecutive disturbance. This would include the presence of tremor, micrographia, and drawings that are obviously segmented (ie, many strokes are necessary when numbers or other graphic elements are drawn). Another sign of motor/dysexecutive syndrome is the presence of perseveration. Second, examine the type and number of errors that are made. Cosentino and colleagues24 have suggested that total clock drawing errors summed across the command to copy conditions is highly suggestive of subcortical neuropathology, such as subcortical white matter alterations as seen on MRI scans.
CONCLUSION
The purpose of this article was to offer some interpretive guidelines for commonly administered neuropsychological tests that are often used to screen for such cognitive disorders as a dementia. Clearly, the four neuropsychological tests described above do not constitute a comprehensive assessment of mental status. Still, the administration of these tests takes little time. More important, the interpretive guidelines have the advantage of orienting the practitioner to issues related to the sensitivity (whether a cognitive disorder is present), as well as specificity (what type of cognitive disorder may be present) of these tests. We believe that use of these tests in the manner described above will result in a higher quality of care for geriatric patients.
1. Ruitenberg A, Ott A, van Swieten JC, et al. Incidence of dementia: Does gender make a difference? Neurobiol Aging 2001;22: 575-580.
2. Libon DJ, Price CC, Garrett KD, Giovannetti T. From Binswanger’s disease to Leukoaraiosis: What we have learned about subcortical vascular dementia. Clin Neuropsychol 2004;18:83-100.
3. Schmidt KS, Gallo JL, Ferri C, et al. The neuropsychological profile of alcohol- related dementia suggests cortical and subcortical pathology. Dement Geriatric Cogn Disord 2005;20:286-291. Epub 2005 Sep 13.
4. Valcour VG, Masaki KH, Curb JD, Blanchette PL. The detection of dementia in the primary care setting. Arch Intern Med 2000;160:2964-2968.
5. Thomas DA, Libon DJ, Ledakis GE. Treating dementia patients with vascular lesions with donepezil: A preliminary analysis. Appl Neuropsychol 2005;12:12-18.
6. U.S. Preventive Service Task Force. Screening for dementia: Recommendations and rationale. June 2003. Available at: www. ahrq.gov/clinic/uspstf/uspsdeme.htm. Accessed December 11, 2006.
7. Folstein MR, Folstein SE, McHugh PR. “Mini-Mental State.” A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975;12:189-198.
8. Cockrell JR, Folstein MF. Mini-Mental State Examination (MMSE). Psychopharmacol Bull 1988;24:689-692.
9. Lezak MD. Neuropsychological Assessment. 3rd ed. New York: Oxford University Press; 1995.
10. Albert ML, Feldman RG, Willis RL. The ‘subcortical dementia’ of progressive supranuclear palsy. J Neurol Neurosurg Psychiatry 1974;37:121-130.
11. Looi JC, Sachdev PS. Differentiation of vascular dementia from AD on neuropsychological tests. Neurology 1999;53:670-678.
12. van Gorp WG, Marcotte TD, Sultzer D, et al. Screening for dementia: Comparison of three commonly used instruments. J Clin Exp Neuropsychol 1999;21:29-38.
13. Brandt J, Folstein SE, Folstein MF. Differential cognitive impairment in Alzheimer’s disease and Huntington’s disease. Ann Neurol 1988;23:555-561.
14. Jefferson AL, Cosentino SA, Ball SK, et al. Errors produced on the Mini-Mental State Examination and neuropsychological test performance among patients with Alzheimer’s disease, ischaemic vascular dementia, and Parkinson’s disease. J Neuropsychiatry Clin Neurosci 2002;14:311-320.
15. McKhann G, Drachman D, Folstein M, et al. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984;34:939-944.
16. Chui HC, Victoroff JI, Margolin D, et al. Criteria for the diagnosis of ischemic vascular dementia proposed by the State of California Alzheimer’s Disease Diagnostic and Treatment Centers. Neurology 1992;42 (3 Pt 1):473-480.
17. Carew TG, Lamar M, Cloud BS, et al. Impairment in category fluency in ischaemic vascular dementia. Neuropsychology 1997;11:400-412.
18. Gourovitch ML, Kirkby BS, Goldberg TE, et al. A comparison of rCBF patterns during letter and semantic fluency. Neuropsychology 2000;14:353-360.
19. Mummery CJ, Patterson K, Hodges JR, Wise RJ. Generating ‘tiger’ as an animal name or a word beginning with T: Differences in brain activation. Proc Biol Sci 1996;263:989-995.
20. Lamar M, Price CC, Davis KL, et al. Capacity to maintain mental set in dementia. Neuropsychologia 2002;40:435-445.
21. Gruenewald PJ, Lockhead GR. The free recall of category examples. Journal of Experimental Psychology: Human Learning and Memory 1980; 6:225-240.
22. Goodglass H, Kaplan E. The Assessment of Aphasia and Related Disorders. 2nd ed. Philadelphia, PA: Lee and Febiger; 1982.
23. Libon DJ, Mattson RE, Glosser G, et al. A nine word dementia version of the California Verbal Learning Test. The Clinical Neuropsychologist 1996;10:237-244.
24. Cosentino S, Jefferson A, Chute DL, et al. Clock drawing errors in dementia: Neuropsychological and neuroanatomical considerations. Cogn Behav Neurol 2004;17:74-83.
25. Royall DR, Cordes JA, Polk M. CLOX: An executive clock drawing task. J Neurol Neurosurg Psychiatry 1998;64:588-594.


