Hypertension: Treatment in Elders
ABSTRACT: A wealth of evidenced-based data support the value of controlling hypertension in the elderly, and that therapy takes on more importance as the US population ages. Although the optimal blood pressure treatment goal in the elderly has not been determined, a therapeutic target of less than 140/90 mm Hg in persons age 65 years to 79 years and a systolic blood pressure of 140 mm Hg and 145 mm Hg (if tolerated) in persons aged 80 years and older is reasonable. We should be careful, however, to avoid intensive lowering of the blood pressure in elderly persons, especially those with diabetes and coronary artery disease, as this might be poorly tolerated and could increase cardiovascular events (“the J-curve phenomenon”).
____________________________________________________________________________________________________________________________________________________
Hypertension in the elderly is an important issue in geriatric medicine, particularly as the US population ages. This article reviews the prevalence and pathophysiology of hypertension in older adults (aged 60 years and older), discusses the risk of cardiovascular disease in individuals with hypertension, and outlines the effect of antihypertensive treatment in decreasing cardiovascular events. Information on antihypertensive drug therapy, including adverse effects, is also provided.
PREVALENCE AND PATHOPHYSIOLOGY
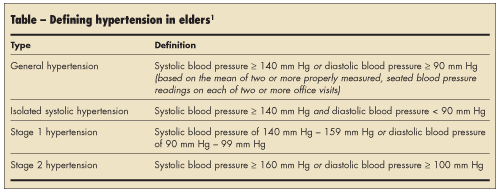
Hypertension in elders is defined as shown in the Table,1 and it occurs in more than two-thirds of persons older than 65 years.2-7 Several biologic factors account for this high prevalence in elderly individuals. An increase in systolic blood pressure in this population is related to an age-associated increase in arterial stiffness due to structural changes within the arterial media (a change in the amount or nature of collagen, interstitial fibrosis, and calcification).8 The increased wall stiffness and tortuosity of the aorta and large arteries that occurs with aging is often reflected by an increased systolic blood pressure and widened pulse pressure. Elderly persons with hypertension are more likely to have increases in left ventricular mass, peripheral resistance, characteristic aortic impedance at rest, and left atrial dimension, as well as reductions in baroreceptor sensitivity, left ventricular early diastolic filling rate, left ventricular early diastolic filling volume, and cardiovascular response to catecholamines.8
The reduction in baroreflex sensitivity that occurs with age and with hypertension leads to an impaired baroreflex-mediated increase in total systemic vascular resistance and to an inability to increase heart rate.9 Therefore, elderly persons with hypertension are more likely to develop orthostatic and postprandial hypotension.10
Hypertension in the elderly also accelerates the age-dependent decline in renal function. Of 143 older persons (mean age, 73 years) with hypertension in an academic nursing home,11 42% (n 5 60) had moderate (33%) or severe (9%) renal insufficiency, with an estimated glomerular filtration rate of less than 60 mL/min/1.73 m2. Renal artery stenosis is also an important cause of secondary hypertension in the elderly.12
HYPERTENSION AND RISK OF CARDIOVASCULAR DISEASE
The higher an individual’s systolic or diastolic blood pressure, the higher his or her risk of cardiovascular morbidity and mortality.13 In elderly persons, increased systolic blood pressure and pulse pressure are stronger risk factors for cardiovascular morbidity and mortality than increased diastolic blood pressure.14,15 An increased pulse pressure found in older persons with isolated systolic hypertension indicates reduced vascular compliance in the large arteries and is an even better risk marker than systolic or diastolic blood pressure.14,15 The Cardiovascular Health Study16 found that a brachial systolic blood pressure higher than 169 mm Hg increased the mortality rate 2.4 times in 5202 older men and women enrolled in the study.
Hypertension in elderly persons is a major risk factor for coronary events, stroke, congestive heart failure (CHF), and peripheral arterial disease.1,17-32 Hypertension is present in approximately 69% of patients with a first myocardial infarction (MI), 77% of patients with a first stroke, 74% of patients with CHF, and 60% of patients with peripheral arterial disease.7,32 Hypertension is also a major risk factor for a dissecting aortic aneurysm, sudden cardiac death, angina pectoris, atrial fibrillation, diabetes mellitus, the metabolic syndrome, chronic kidney disease, thoracic and abdominal aortic aneurysms, left ventricular hypertrophy, vascular dementia, Alzheimer’s disease, and ophthalmologic disorders.22
Older persons are more likely to have hypertension and isolated systolic hypertension, to develop new cardiovascular events, and to have target organ damage and clinical cardiovascular disease than younger individuals. Elderly persons also have the lowest rates of blood pressure control.1,22,33,34 According to a study by Lloyd-Jones and colleagues,34 blood pressure is adequately controlled in 36% of men and 28% of women aged 60 to 79 years and in 38% of men and 23% of women aged 80 years and older.34
Barriers to the treatment of hypertension include a lack of understanding by physicians that frail elderly persons should be treated according to recommended guidelines to reduce cardiovascular morbidity and mortality. Prevalent comorbidities, polypharmacy, an asymptomatic state, side effects from medications, and the high cost of medications contribute to lower blood pressure control rates in elders.22,35
EFFECT OF ANTIHYPERTENSIVE TREATMENT IN DECREASING CARDIOVASCULAR EVENTS
Numerous prospective, double-blind, randomized, placebo-controlled studies have shown that antihypertensive drug therapy decreases the development of new coronary events, stroke, and congestive heart failure (CHF) in older persons.1,22,36-47 If treated appropriately, elderly individuals with hypertension will have a greater absolute reduction in cardiovascular events (eg, major coronary events, stroke, CHF, renal insufficiency) and a greater reduction in dementia than younger persons.48
Antihypertensive drug therapy reduces the incidence of all strokes by 38% in women, by 34% in men, by 36% in older persons (aged 60 years and older), and by 34% in persons older than 80 years.24 The overall data suggest that the reduction of stroke in older persons with hypertension is related more to a decrease in blood pressure than to the type of antihypertensive drugs used.24
In PROGRESS (Perindopril Protection Against Recurrent Stroke Study),49 perindopril plus indapamide reduced stroke-related dementia by 34% and cognitive decline by 45%. In the Syst-Eur (Systolic Hypertension in Europe) trial,50 nitrendipine decreased dementia by 55% at 3.9-year follow-up. A study by Murray and colleagues51 found that antihypertensive drug treatment decreased cognitive impairment by 38% in 1900 older African Americans, whereas the Rotterdam Study52 found that antihypertensive drugs reduced vascular dementia by 70%.
Based on the data available at the time, Aronow53 proposed in an editorial that unless the results of HYVET (Hypertension in the Very Elderly Trial)47 showed that antihypertensive drug therapy was not beneficial in patients aged 80 years and older, this group should receive antihypertensive drug treatment. Goodwin54 disagreed with this approach, and his response to Aronow’s editorial was accompanied by several commentaries, some of which supported the treatment of very elderly hypertensive patients and some of which did not.
In HYVET, 3845 individuals aged 80 years and older (mean age, 83.6 years) with a sustained systolic blood pressure of 160 mm Hg or higher were randomized to receive either the diuretic indapamide (sustained release, 1.5 mg) or matching placebo.47 Perindopril 2 mg or 4 mg, or matching placebo, was added if needed to achieve the target blood pressure of 150/80 mm Hg. The median follow-up was 1.8 years. Antihypertensive drug treatment reduced the incidence of the primary end point of fatal or nonfatal stroke by 30% (P 5 .06). It reduced fatal stroke by 39% (P 5 .05), all-cause mortality by 21% (P 5 .02), death from cardiovascular causes by 23% (P 5 .06), and heart failure by 64% (P < .001). The significant 21% reduction in all-cause mortality by antihypertensive drug treatment was unexpected. The benefits of antihypertensive drug treatment began to be apparent during the first year of follow-up.
The prevalence of baseline cardiovascular disease was only 12% in the patients in HYVET.47 In a cohort of patients (mean age, 80 years) with hypertension seen in a university geriatrics practice, 70% had baseline cardiovascular disease, target organ damage, or diabetes mellitus.5 An elderly population such as this one, with a high prevalence of cardiovascular disease, would be expected to have a greater absolute reduction in cardiovascular events resulting from antihypertensive drug therapy. Although the results of HYVET clearly indicate that hypertensive patients aged 80 years and older should be treated with antihypertensive drug therapy, the study does not provide data on target blood pressure, demonstrating further research in this area is required.22,55
Although the optimal blood pressure treatment goal has not been determined, a therapeutic target of less than 140/90 mm Hg in persons younger than 80 years and a systolic blood pressure of between 140 mm Hg and 145 mm Hg, if tolerated, is reasonable in persons aged 80 years and older.22 We should also be careful to avoid intensive lowering of blood pressure, especially in elderly persons with diabetes and coronary artery disease (CAD), as this might be poorly tolerated and could increase cardiovascular events (“the J-curve phenomenon”). However, until additional data become available from randomized controlled trials, such as SPRINT (Systolic Blood Pressure Intervention Trial), that compare various blood pressure targets in the elderly and in younger persons, existing epidemiologic and clinical trial data suggest that a diagnostic and therapeutic threshold for hypertension of 140/90 mm Hg is reasonable in adults younger than 65 years and between 65 years and 79 years, and a threshold systolic blood pressure of 150 mm Hg in adults aged 80 years and older.22
Continued on next page
USE OF ANTIHYPERTENSIVE DRUG THERAPY IN OLDER PERSONS
A meta-analysis of 147 randomized trials that together included 464,000 persons with hypertension showed that, except for the extra protective effect of beta blockers given after MI and a minor additional effect of calcium channel blockers in preventing stroke, use of beta blockers, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), diuretics, and calcium channel blockers cause a similar decrease in coronary events and stroke for a given decrease in blood pressure.56,57 The proportionate decrease in cardiovascular events was the same or similar regardless of pretreatment blood pressure levels and the presence or absence of cardiovascular events.56,57 Diuretics, ACE inhibitors, ARBs, calcium channel blockers, or beta blockers may be used as initial therapy in the treatment of primary hypertension in both older and younger persons. Atenolol should not be used to treat younger or older persons with hypertension because this drug is not more effective than placebo in reducing cardiovascular events and mortality.58-60 Carvedilol, nebivolol, and bisoprolol are preferred over other beta blockers because of their more favorable hemodynamic and metabolic actions.60
Centrally acting agents, such as clonidine, reserpine, and guanethidine, should not be used as monotherapy in elderly persons because they have been associated with a high incidence of significant side effects, including sedation, depression, and constipation. Most older persons with hypertension require two or more antihypertensive drugs to control their blood pressure.1,22 If the blood pressure is more than 20/10 mm Hg above the goal blood pressure, drug therapy should be initiated with two antihypertensive agents.1,22
The initial antihypertensive drug should be given at the lowest dose and gradually increased to the maximum dose. If the antihypertensive response to the initial drug is inadequate after reaching the full dose of drug, a second drug from another class should be given if the person is tolerating the initial drug. If there is no therapeutic response or if there are significant adverse effects, a drug from another class should be substituted. If the antihypertensive response is inadequate after reaching the full dose of two classes of drugs, a third drug from another class should be added.
Elderly persons with hypertension have a very high prevalence of associated medical conditions and concerns, such as increasing obesity, smoking, excessive alcohol intake, and insulin resistance; thus, selection of antihypertensive agent should consider these factors.1,22 Before adding new antihypertensive drugs, the physician should evaluate the possible reasons for inadequate response to antihypertensive drug therapy, including associated conditions, nonadherence to therapy, volume overload, and drug interactions (ie, use of nonsteroidal anti-inflammatory drugs [NSAIDs], caffeine, antidepressants, nasal decongestants, sympathomimetics, and other agents).1,22 Causes of secondary hypertension should be identified and treated in accordance with current guidelines.1,12,22
ADVERSE EFFECTS OF ANTIHYPERTENSIVE DRUG THERAPY
All antihypertensive drugs may predispose elderly persons to develop symptomatic orthostatic hypotension and postprandial hypotension, for the pathophysiological reasons explained earlier, and this may result in falls or syncope.10 In these cases, the dose of antihypertensive drug may need to be decreased or another agent given, and measurements of blood pressure in the upright position, especially after eating, are indicated.
Diuretics may cause volume depletion. Vasodilators such as ACE inhibitors, ARBs, calcium channel blockers, nitrates, hydralazine, and prazosin may cause a reduction in systemic vascular resistance and venous dilation.
Compared with amlodipine, ramipril significantly decreased progression of renal disease in 1094 African Americans with hypertensive nephrosclerosis.61 If an elder cannot tolerate an ACE inhibitor because of cough, angioneurotic edema, rash, or altered taste sensation, an ARB should be given.62 Compared with ramipril alone, the addition of telmisartan to ramipril in patients (mean age, 67 years) with vascular disease or high-risk diabetes mellitus did not reduce cardiovascular events but increased hypotensive symptoms (4.8% vs 1.7%), syncope (0.3% vs 0.2%), and renal dysfunction (1.1% vs 0.7%).63
To avoid hyperkalemia, potassium-sparing diuretics should not be given to elderly persons receiving ACE inhibitors or ARBs. Risk factors for renal insufficiency in elderly persons receiving these agents include renal artery stenosis (usually bilateral); polycystic renal disease; decreased absolute or effective arterial blood volume; use of NSAIDs, cyclosporine, or tacrolimus; and sepsis.64,65 However, reversible renal failure may occur in elderly persons who are treated with ACE inhibitors or ARBs and are dehydrated or salt-depleted. ACE inhibitors or ARBs can cause an azotemic response when there is an absolute reduction in intravascular volume due to aggressive diuresis, poor oral intake, gastroenteritis, or an effective reduction in intravascular volume related to severe CHF.66
Beta blockers depress the sinus node and the atrioventricular (AV) node and are contraindicated in patients with severe sinus bradycardia, sinoatrial disease, and marked first-, second-, and third-degree AV block.67 Beta blockers should also not be given to patients with bronchial asthma or to those with lung disease and severe bronchospasm.67 Beta blockers may cause depression or confusion in elderly individuals.
Short-acting dihydropyridine calcium channel blockers, such as short-acting nifedipine, have the potential to increase cardiovascular events and should be avoided.68 Verapamil and diltiazem depress the sinus node and the AV node and are contraindicated in patients with severe sinus bradycardia, sinoatrial disease, and marked first-, second-, and third-degree AV block.69
Diltiazem and verapamil are contraindicated in treating postinfarction patients who have an abnormal left ventricular ejection fraction (LVEF) because these agents will increase the risk of coronary events, mortality, and CHF.70-72 Calcium channel blockers, such as nifedipine, diltiazem, and verapamil, exacerbate CHF in patients who also have abnormal LVEF.73
In the ALLHAT (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial), the doxazosin arm, which included 9067 patients, was prematurely stopped at a median of 3.3 years.74 Compared with the diuretic chlorthalidone, the alpha blocker doxazosin significantly increased CHF by 204%, stroke by 19%, angina pectoris by 16%, coronary revascularization by 15%, and combined cardiovascular disease (coronary heart disease death, nonfatal MI, stroke, angina pectoris, coronary revascularization, CHF, or peripheral arterial disease) by 25%.
At 40-month mean follow-up in an observational study of elderly patients (mean age, 80 years) with a previous MI and hypertension, patients treated with alpha blockers had a significant increase in new coronary events of 1.69 times compared with beta blockers, of 1.5 times when compared with ACE inhibitors, and of 1.35 times when compared with diuretics.75 Alpha blockers cause a high incidence of orthostatic hypotension, especially in patients receiving diuretics or other vasodilator drugs.75
Centrally acting drugs should not be used as monotherapy in elders because they cause a high rate of sedation, precipitate or exacerbate depression, and cause constipation.75 Direct vasodilators may cause headache, fluid retention, tachycardia, and may aggravate angina pectoris. Hydralazine caused a lupus-like syndrome in 6.7% of 281 patients treated with that agent for 3 years.76 Minoxidil may cause hirsutism and pericardial effusion.77
USE OF ANTIHYPERTENSIVE DRUGS IN PERSONS WITH ASSOCIATED MEDICAL CONDITIONS
Elderly persons with a previous MI should be treated with beta blockers and ACE inhibitors.1,22,72,78-82 At 40-month follow-up in an observational prospective study of 1212 elderly men and women with a previous MI and hypertension who were treated with beta blockers, ACE inhibitors, diuretics, calcium channel blockers, or alpha blockers, the incidence of new coronary events in those treated with one antihypertensive drug was the lowest in those who received beta blockers or ACE inhibitors.72 In elderly persons treated with two antihypertensive drugs, the incidence of new coronary events was the lowest in those treated with beta blockers plus ACE inhibitors.72
Beta blockers should be used to treat older persons with complex ventricular arrhythmias and abnormal or normal LVEF, and for those with CHF and abnormal or normal LVEF.83-87 Beta blockers should also be used to treat hypertensive elders who have angina pectoris, myocardial ischemia, supraventricular tachyarrhythmias (eg, atrial fibrillation with a rapid ventricular rate), hyperthyroidism, preoperative hypertension, migraine, or essential tremor.1,88-92
In addition to beta blockers, elderly persons with CHF should be treated with diuretics and ACE inhibitors.93 ACE inhibitors or ARBs should be administered to elderly persons who have diabetes mellitus, chronic renal insufficiency, or proteinuria.1,22,61,62,79 In the seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure, diuretics and ACE inhibitors are recommended to prevent recurrent stroke in elderly persons with hypertension.1,48 Thiazide diuretics should be used to treat elderly persons with osteoporosis.1
It is also very important to treat other cardiovascular risk factors in elderly persons with hypertension to reduce cardiovascular events and mortality. Patients must stop smoking, dyslipidemia must be treated, and diabetes mellitus must be controlled.11,94-100 More aggressive control of blood pressure among persons at high risk for CAD—those with diabetes mellitus, chronic kidney disease, coronary artery risk equivalent, or a 10-year Framingham risk score of 10% or higher with maintenance of the blood pressure at less than 130/80 mm Hg or less than 120/80 mm Hg in patients with left ventricular dysfunction, as noted by the AHA Task Force67 scientific statement in 2007—was based on expert medical opinion at that time, not on prospective, randomized, and adequately controlled trial data.101
The PROVE IT-TIMI (Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction) 22 trial enrolled 4162 patients with an acute coronary syndrome (acute MI with or without ST-segment elevation or high-risk unstable angina pectoris).102 The lowest rates of cardiovascular events occurred with a systolic blood pressure of approximately 130 mm Hg to 140 mm Hg and a diastolic blood pressure of approximately 80 mm Hg to 90 mm Hg, with a nadir of 136/85 mm Hg.
An observational subgroup analysis was performed in 6400 of the 22,576 participants enrolled in INVEST (International Verapamil SR-Trandolapril Study).103 The participants had diabetes mellitus and CAD. Patients were categorized as having tight control of blood pressure if they could maintain a systolic blood pressure of less than 130 mm Hg and a diastolic blood pressure of less than 85 mm Hg; usual control if they could maintain a systolic blood pressure between 130 mm Hg and 139 mm Hg; and uncontrolled if their systolic blood pressure was 140 mm Hg or higher. During 16,893 patient-years of follow-up, a cardiovascular event rate of 12.6% occurred in patients with usual control of blood pressure versus 19.8% in patients with uncontrolled hypertension (P < .001). The incidence of cardiovascular events was 12.6% in patients with usual control of blood pressure versus 12.7% in patients with tight control (P 5 NS). The all-cause mortality rate was 11.0% with tight control of blood pressure versus 10.2% with usual control (P 5 .06). When extended follow-up was included, the all-cause mortality rate was 22.8% with tight control of blood pressure versus 21.8% with usual control (P 5 .04).
The ACCORD (Action to Control Cardiovascular Risk in Diabetes) blood pressure trial randomized 4733 patients with type 2 diabetes mellitus to intensive blood pressure control with a target systolic blood pressure of less than 120 mm Hg or to standard blood pressure control with a target systolic blood pressure of less than 140 mm Hg.104 The primary composite outcome was nonfatal MI, nonfatal stroke, or death from cardiovascular causes. The mean follow-up was 4.7 years. After 1 year, the mean systolic blood pressure was 119.3 mm Hg in the intensive blood pressure control group versus 133.5 mm Hg in the standard blood pressure control group. The annual rate of the primary outcome was 1.87% in the intensive blood pressure control group versus 2.09% in the standard blood pressure control group (P 5 NS). The annual rate of death from any cause was 1.28% in the intensive blood pressure control group versus 1.19% in the standard blood pressure control group (P 5 NS). The annual rate of stroke, a prespecified secondary outcome, was 0.32% in the intensive blood pressure control group versus 0.53% in the standard blood pressure control group (P 5 .01). Serious adverse events attributed to antihypertensive treatment occurred in 3.3% of the intensive blood pressure control group versus 1.3% of the standard blood pressure control group (P < .001).104
We studied the impact of baseline systolic blood pressure on outcomes in 7785 patients with mild-to-moderate chronic CHF in the Digitalis Investigation Group trial.105 During 5 years of follow-up, a baseline systolic blood pressure of 120 mm Hg or lower was associated with a 15% increase in cardiovascular mortality (P 5 .032), a 30% increase in heart failure mortality (P 5 .006), a 13% increase in cardiovascular hospitalization (P 5 .008), a 10% increase in all-cause hospitalization (P 5 .017), and a 21% increase in heart failure hospitalization (P 5 .002).105
CONCLUSION
A wealth of evidenced-based data support the value of controlling hypertension in the elderly, and that therapy takes on more importance as the US population ages. Although the optimal blood pressure treatment goal in the elderly has not been determined, a therapeutic target of less than 140/90 mm Hg in persons age 65 years to 79 years and a systolic blood pressure of 140 mm Hg and 145 mm Hg (if tolerated) in persons aged 80 years and older is reasonable.22,101,106 We should be careful, however, to avoid intensive lowering of the blood pressure in elderly persons, especially those with diabetes and CAD, as this might be poorly tolerated and could increase cardiovascular events (“the J-curve phenomenon”). n
Editor’s note: This article originally appeared in Clinical Geriatrics, November 2011, volume 19, pages 39 to 46.
1. Chobanian AV, Bakris GL, Black HR, et al; National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; National High Blood Pressure Education Program Coordinating Committee. The seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report [published correction appears in JAMA. 2003;290(2):197]. JAMA. 2003;289(19):2560-2572.
2.The sixth report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure [published correction appears in Arch Intern Med. 1998;158(6):573]. Arch Intern Med. 1997;157(21):2413-2446.
3. Aronow WS, Ahn C, Gutstein H. Prevalence and incidence of cardiovascular disease in 1160 older men and 2464 older women in a long-term health care facility. J Gerontol A Biol Sci Med Sci. 2002;57(1):M45-M46.
4. Mendelson G, Ness J, Aronow WS. Drug treatment of hypertension in older persons in an academic hospital-based geriatrics practice. J Am Geriatr Soc. 1999;47(5):597-599.
5. Koka M, Joseph J, Aronow WS. Adequacy of control of hypertension in an academic nursing home. J Am Med Dir Assoc. 2007;8(8):538-540.
6. Joseph J, Koka M, Aronow WS. Prevalence of a hemoglobin A1c less than 7.0%, of a blood pressure less than 130/80 mm Hg, and of a serum low-density lipoprotein cholesterol less than 100 mg/dL in older patients with diabetes mellitus in an academic nursing home. J Am Med Dir Assoc. 2008;9(1):51-54.
7. Lloyd-Jones D, Adams R, Carnethon M, et al; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2009 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee [published correction appears inCirculation. 2009;119(3):e182]. Circulation. 2009;119(3):e21-e181.
8. Lakatta EG. Mechanisms of hypertension in the elderly. J Am Geriatr Soc. 1989;37(8):780-790.
9. Gribbin B, Pickering GT, Sleight P, Peto R. Effect of age and high blood pressure on baroreflex sensitivity in man. Circ Res. 1971;29(4):424-431.
10. Aronow WS. Dizziness and syncope. In: Hazzard WR, Blass JP, Ettinger WH Jr, Halter JB, Ouslander JG, eds. Principles of Geriatric Medicine and Gerontology. 4th ed. New York, NY: McGraw-Hill; 1998:1519-1534.
11. Joseph J, Koka M, Aronow WS. Prevalence of moderate and severe renal insufficiency in older persons with hypertension, diabetes mellitus, coronary artery disease, peripheral arterial disease, ischemic stroke, or congestive heart failure in an academic nursing home. J Am Med Dir Assoc. 2008;9(4):257-259.
12. Chiong JR, Aronow WS, Khan IA, et al. Secondary hypertension: current diagnosis and treatment. Int J Cardiol. 2008;124(1):6-21.
13. National High Blood Pressure Education Program Working Group report on hypertension in the elderly. Hypertension. 1994;23(3):275-285.
14. Madhavan S, Ooi WL, Cohen H, Alderman MH. Relation of pulse pressure and blood pressure reduction to the incidence of myocardial infarction. Hypertension. 1994;23(3):395-401.
15. Rigaud AS, Forette B. Hypertension in older adults. J Gerontol A Biol Sci Med Sci. 2001;56(4):M217-M225.
16. Fried LP, Kronmal RA, Newman AB, et al. Risk factors for 5-year mortality in older adults: the cardiovascular health study. JAMA. 1998;279(8):585-592.
17. Aronow WS, Ahn C, Kronzon I, Koenigsberg M. Congestive heart failure, coronary events and atherothrombotic brain infarction in elderly blacks and whites with systemic hypertension and with and without echocardiographic and electrocardiographic evidence of left ventricular hypertrophy. Am J Cardiol. 1991;67(4):295-299.
18. Aronow WS, Ahn C. Risk factors for new coronary events in a large cohort of very elderly patients with and without coronary artery disease. Am J Cardiol. 1996;77(10):864-866.
19. Vokonas PS, Kannel WB. Epidemiology of coronary heart disease in the elderly. In Aronow WS, Fleg JL, Rich MW, eds. Cardiovascular Disease in the Elderly. 4th ed. New York, NY: Informa Healthcare; 2008:215-241.
20. Franklin SS, Larson MG, Khan SA, et al. Does the relation of blood pressure to coronary heart disease risk change with aging? The Framingham Heart Study. Circulation. 2001;103(9):1245-1249.
21. Psaty BM, Furberg CD, Kuller LH, et al. Association between blood pressure level and the risk of myocardial infarction, stroke, and total mortality: the Cardiovascular Health Study. Arch Intern Med. 2001;161(9):1183-1192.
22. Aronow WS, Fleg JL, Pepine CJ, et al. ACCF/AHA 2011 expert consensus document on hypertension in the elderly: a report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus. Documents developed in collaboration with the American Academy of Neurology, American Geriatrics Society, American Society for Preventative Cardiology, American Society of Hypertension, American Society of Nephrology, Association of Black Cardiologists, and European Society of Hypertension. J Am Coll Cardiol. 2011;57(20):2037-2114.
23. Aronow WS, Ahn C, Gutstein H. Risk factors for new atherothrombotic brain infarction in 664 older men and 1488 older women. Am J Cardiol. 1996;77(15):1381-1383.
24. Aronow WS, Frishman WH. Treatment of hypertension and prevention of ischemic stroke. Curr Cardiol Rep. 2004;6(2):124-129.
25. Wolf PA. Cerebrovascular disease in the elderly. In: Tresch DD, Aronow WS, eds. Cardiovascular Disease in the Elderly Patient. New York, NY: Marcel Dekker; 1994:125-147.
26. Aronow WS, Ahn C, Kronzon I. Comparison of incidences of congestive heart failure in older African-Americans, Hispanics, and whites. Am J Cardiol. 1999;84(5):611-612, A9.
27. Levy D, Larson MG, Vasan RS, Kannel WB, Ho KK. The progression from hypertension to congestive heart failure. JAMA. 1996;275(20):1557-1562.
28. Stokes J 3rd, Kannel WB, Wolf PA, Cupples LA, D’Agostino RB. The relative importance of selected risk factors for various manifestations of cardiovascular disease among men and women from 35 to 64 years old: 30 years of follow-up in the Framingham Study. Circulation. 1987;75(6 Pt 2):V65-V73.
29. Aronow WS, Sales FF, Etienne F, Lee NH. Prevalence of peripheral arterial disease and its correlation with risk factors for peripheral arterial
disease in elderly patients in a long-term health care facility. Am J Cardiol. 1988;62(9):644-646.
30. Ness J, Aronow WS, Ahn C. Risk factors for peripheral arterial disease in an academic hospital-based geriatrics practice. J Am Geriatr Soc. 2000;48(3):312-314.
31. Ness J, Aronow WS, Newkirk E, McDanel D. Prevalence of symptomatic peripheral arterial disease, modifiable risk factors, and appropriate use of drugs in the treatment of peripheral arterial disease in older persons seen in a university general medicine clinic. J Gerontol A Biol Sci Med Sci. 2005;
60(2):255-257.
32. Meyer P, Ekundayo OJ, Adamopoulos C, et al. A propensity-matched study of the association of peripheral arterial disease with cardiovascular outcomes in community-dwelling older adults. Am J Cardiol. 2009;103(6):839-844.
33. Hyman DJ, Pavlik VN. Characteristics of patients with uncontrolled hypertension in the United States [published correction appears in N Engl J Med. 2002;346(7):544]. N Engl J Med. 2001;345(7):479-486.
34. Lloyd-Jones DM, Evans JC, Levy D. Hypertension in adults across the age spectrum: current outcomes and control in the community. JAMA. 2005;294(4):466-472.
35. Gandelman G, Aronow WS, Varma R. Prevalence of adequate blood pressure control in self-pay or Medicare patients versus Medicaid or private insurance patients with systemic hypertension followed in a university cardiology or general medicine clinic. Am J Cardiol. 2004;94(6):815-816.
36. The Australian therapeutic trial in mild hypertension. Report by the management committee. Lancet. 1980;1(8181):1261-1267.
37. Medical Research Council Working Party. MRC trial of mild hypertension: principal results. Br Med J (Clin Res Ed). 1985;291(6488):97-104.
38. Medical Research Council Working Party. Medical Research Council Trial of treatment of hypertension in older adults: principal results. BMJ. 1992;304(6824):405-412.
39. Amery A, Birkenhäger W, Brixko P, et al. Morbidity and mortality results from the European Working Party on High Blood Pressure in the Elderly Trial. Lancet. 1985;1(8442):1349-1354.
40. Coope J, Warrender TS: Randomised trial of the treatment of hypertension in elderly patients in primary care. Br Med J (Clin Res Ed). 1986;293
(6555):1145-1151.
41. Dahlöf B, Lindholm LH, Hansson L, Scherstén B, Ekbom T, Wester PO. Morbidity and mortality in the Swedish Trial in Old Patients With Hypertension (STOP-Hypertension). Lancet. 1991;338(8778):1281-1285.
42. SHEP Cooperative Research Group. Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). JAMA. 1991;265(24):3255-3264.
43. Kostis JB, Davis BR, Cutler J, et al; SHEP Cooperative Research Group. Prevention of heart failure by antihypertensive drug treatment in older persons with isolated systolic hypertension. JAMA. 1997;278(3):212-216.
44. Perry HM Jr, Davis BR, Price TR, et al. Effect of treating isolated systolic hypertension on the risk of developing various types and subtypes of stroke. The Systolic Hypertension in the Elderly Program (SHEP). JAMA. 2000;284(4):465-471.
45. Staessen JA, Fagard R, Thijs L, et al; Systolic Hypertension in Europe (Syst-Eur) Trial Investigators. Randomised double-blind comparison of placebo and active treatment for older patients with isolated systolic hypertension. Lancet. 1997;350(9080):757-764.
46. Wang JG, Staessen JA, Gong L, Liu L; Systolic Hypertension in China (Syst-China) Collaborative Group. Chinese trial on isolated systolic hypertension in the elderly. Arch Intern Med. 2000;160(2):211-220.
47. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358(18):1887-1898.
48. Aronow WS, Frishman WH. Effect of antihypertensive drug treatment on cognitive function. Clinical Geriatrics. 2006;14(11):25-28.
49. PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack [published correction appears in Lancet. 2011;358(9292):1556]. Lancet. 2001;358(9287):1033-1041.
50. Forette F, Seux ML, Staessen JA, et al; Systolic Hypertension in Europe Investigators. The prevention of dementia with antihypertensive treatment: new evidence from the Systolic Hypertension in Europe (Syst-Eur) study [published correction appears in Arch Intern Med. 2003;163(2):241]. Arch Intern Med. 2002;162(18):2046-2052.
51. Murray MD, Lane KA, Gao S, et al. Preservation of cognitive function with antihypertensive medications. A longitudinal analysis of a community-based sample of African Americans. Arch Intern Med. 2002;162(18):2090-2096.
52. in’t Veld BA, Ruitenberg A, Hofman A, Stricker BH, Breteler MM. Antihypertensive drugs and incidence of dementia: the Rotterdam study. Neurobiol Aging. 2001;22(3):407-412.
53. Aronow WS. What is the appropriate treatment of hypertension in elders? J Gerontol A Biol Sci Med Sci. 2002;57(8):M483-M486.
54. Goodwin JS. Embracing complexity: a consideration of hypertension in the very old. J Gerontol A Biol Sci Med Sci. 2003;58(7):M653-M658.
55. Aronow WS. Older age should not be a barrier to the treatment of hypertension. Nat Clin Pract Cardiovasc Med. 2008;5(9):514-515.
56. Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665.
57. Mancia G, Laurent S, Agabiti-Rosei E, et al. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension Task Force document. Blood Press. 2009;18(6):308-347.
58. Aronow WS. Might losartan reduce sudden cardiac death in diabetic patients with hypertension? Lancet. 2003;362(9384):591-592.
59. Carlberg B, Samuelson O, Lindholm LH. Atenolol in hypertension: is it a wise choice? Lancet. 2004;364(9446):1684-1689.
60. Aronow WS. Current role of beta-blockers in the treatment of hypertension. Expert Opin Pharmacother. 2010;11(16):2599-2607.
61. Agodoa LY, Appel L, Bakris GL, et al; African American Study of Kidney Disease and Hyperten-
sion (AASK) Study Group. Effect of ramipril versus amlodipine on renal outcomes in hypertensive nephrosclerosis: a randomized controlled trial. JAMA. 2001;285(21):2719-2728.
62. Brenner BM, Cooper ME, de Zeeuw D, et al; RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345(12):861-869.
63. Yusuf S, Teo KK, Pogue J, et al; ONTARGET Investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358(15):1547-1559.
64. Hricick DE, Browning PJ, Kopelman R, Goorno WE, Madias NE, Dzau VJ. Captopril-induced functional renal insufficiency in patients with bilateral renal artery stenosis or renal artery stenosis in a solitary kidney. N Engl J Med. 1983;308(7):373-376.
65. Palmer BF. Renal dysfunction complicating the treatment of hypertension. N Engl J Med. 2002;347(16):1256-1261.
66. Toto RD, Mitchell HC, Lee HC, Milam C, Pettinger WA. Reversible renal insufficiency due to angiotensin-converting enzyme inhibitors in hypertensive nephrosclerosis. Ann Intern Med. 1991;115(7):513-519.
67. Rosendorff C, Black HR, Cannon CP, et al; American Heart Association Council for High Blood Pressure Research; American Heart Association Council on Clinical Cardiology; American Heart Association Council on Epidemiology and Prevention. Treatment of hypertension in the prevention and management of ischemic heart disease: a scientific statement from the American Heart Association Council for High Blood Pressure Research and the Councils on Clinical Cardiology and Epidemiology and Prevention [published correction appears in Circulation. 2007;116(5):e121]. Circulation. 2007;115(12):2761-2788.
68. Pahor M, Guralnik JM, Corti C, et al. Long-term survival and use of antihypertensive medications in older persons. J Am Geriatr Soc. 1995;43(11):1191-1197.
69. Aronow WS. Verapamil as an antiarrhythmic agent. In: Gould LA, ed. Drug Treatment of Cardiac Arrhythmias. Mount Kisco, NY: Futura Publishing Company Inc; 1983:325-341.
70. The Multicenter Diltiazem Postinfarction Trial Research Group. The effect of diltiazem on mortality and reinfarction after myocardial infarction. N Engl J Med. 1988;319(7):385-392.
71. Goldstein RE, Boccuzzi SJ, Cruess D, Nattel S; Adverse Experience Committee; Multicenter Diltiazem Postinfarction Research Group. Diltiazem increases late-onset congestive heart failure in postinfarction patients with early reduction in ejection fraction. Circulation. 1991;83(1):52-60.
72. Aronow WS, Ahn C. Incidence of new coronary events in older persons with prior myocardial infarction and systemic hypertension treated with beta blockers, angiotensin-converting enzyme inhibitors, diuretics, calcium antagonists, and alpha blockers. Am J Cardiol. 2002;89(10):1207-1209.
73. Elkayam U, Amin J, Mehra A, Vasquez J, Weber L, Rahimtoola SH. A prospective, randomized, double-blind, crossover study to compare the efficacy and safety of chronic nifedipine therapy with that of isosorbide dinitrate and their combination in the treatment of chronic congestive heart failure. Circulation. 1990;82(6):1954-1961.
74. ALLHAT Collaborative Research Group. Major cardiovascular events in hypertensive patients randomized to doxazosin vs chlorthalidone: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) [published correction appears in JAMA. 2002;288(23):2976]. JAMA. 2000;283(15):1967-1975.
75. Frishman WH, Aronow WS, Cheng-Lai A. Cardiovascular drug therapy in the elderly. In: Aronow WS, Fleg JL, Rich MW, eds. Cardiovascular Disease in the Elderly. 4th ed. New York, NY: Informa Healthcare; 2008:99-135.
76. Cameron HA, Ramsay LE. The lupus syndrome induced by hydralazine: a common complication with low-dose treatment. Br Med J (Clin Res Ed).1984;289(6442):410-412.
77. Krehlik JM, Hindson DA, Crowley JJ Jr, Knight LL. Minoxidil-associated pericarditis and fatal cardiac tamponade. West J Med. 1986;143(4):527-529.
78. Ryan TJ, Antman EM, Brooks NH, et al. 1999 update: ACC/AHA guidelines for the management of patients with acute myocardial infarction: executive summary and recommendations: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on Management of Acute Myocardial Infarction). Circulation. 1999;100(9):1016-1030.
79. Yusuf S, Sleight P, Pogue J, Bosch J, Davies R, Dagenais G; Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting-enzyme inhibitor, ramipril on cardiovascular events in high-risk patients [published correction appears in N Engl J Med. 2000;342(18):1376 and 2000;342(10):748]. N Engl J Med. 2000;342(3):145-153.
80. Aronow WS, Ahn C, Kronzon I. Effect of beta blockers alone, of angiotensin-converting enzyme inhibitors alone, and of beta blockers plus angiotensin-converting enzyme inhibitors on new coronary events and on congestive heart failure in older persons with healed myocardial infarcts and asymptomatic left ventricular systolic dysfunction. Am J Cardiol. 2001;88(11):1298-1300.
81. Aronow WS, Ahn C. Effect of beta blockers on incidence of new coronary events in older persons with prior myocardial infarction and diabetes mellitus. Am J Cardiol. 2001;87(11):780-781, A8.
82. Aronow WS, Ahn C. Effect of beta blockers on incidence of new coronary events in older persons with prior myocardial infarction and symptomatic peripheral arterial disease. Am J Cardiol. 2001;87(11):1284-1286.
83. Kennedy HL, Brooks MM, Barker AH, et al. Beta-blocker therapy in the Cardiac Arrhythmia Suppression Trial. Am J Cardiol. 1994;74(3):674-680.
84. Aronow WS, Ahn C, Mercando AD, Epstein S, Kronzon I. Effect of propranolol versus no antiarrhythmic drug on sudden cardiac death, total cardiac death, and total death in patients ≥62 years of age with heart disease, complex ventricular arrhythmias, and left ventricular ejection fraction ≥40%. Am J Cardiol. 1994;74(3):267-270.
85. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet. 1999;353(9169):2001-2007.
86. Flather MD, Shibata MC, Coats AJ, et al; SENIORS Investigators. Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS). Eur Heart J. 2005;26(3):215-225.
87. Aronow WS, Ahn C, Kronzon I. Effect of propranolol versus no propranolol on total mortality plus nonfatal myocardial infarction in older patients with prior myocardial infarction, congestive heart failure, and left ventricular ejection fraction ≥40% treated with diuretics plus angiotensin-converting enzyme inhibitors. Am J Cardiol. 1997;80(2):207-209.
88. Aronow WS, Frishman WH. Angina in the elderly. In: Aronow WS, Fleg JL, Rich MW, eds. Cardiovascular Disease in the Elderly. 4th ed. New York, NY: Informa Healthcare; 2008:269-292.
89. Aronow WS, Ahn C, Mercando AD, Epstein S, Kronzon I. Decrease of mortality by propranolol in patients with heart disease and complex ventricular arrhythmias is more an anti-ischemic than an antiarrhythmic effect. Am J Cardiol. 1994;74(6):613-615.
90. Aronow WS. Treatment of atrial fibrillation. Part 1. Cardiol Rev. 2008;16(4):181-188.
91. Aronow WS. Treatment of atrial fibrillation and atrial flutter. Part II. Cardiol Rev. 2008;16(5):230-239.
92. Aronow WS. The heart and thyroid disease. In: Gambert SR, ed. Clinics in Geriatric Medicine. Thyroid Disease. Philadelphia, PA: W.B. Saunders; 1995:219-229.
93. Hunt SA, Baker DW, Chin MH, et al; American College of Cardiology; American Heart Association. ACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). J Am Coll Cardiol. 2001;38(7):2101-2113.
94. Smith SC Jr, Allen J, Blair SN, et al; American Heart Association, American College of Cardiology; National Heart, Lung, and Blood Institute. ACC/AHA guidelines for secondary prevention for patients with coronary and other atherosclerotic vascular disease: 2006 update: endorsed by the National Heart, Lung, and Blood Institute [published correction appears in Circulation. 2006;113(22):e847]. Circulation. 2006;113(19):2363-2372.
95. Grundy SM, Cleeman JI, Merz CNB, et al; National Heart, Lung, and Blood Institute; American College of Cardiology Foundation; American Heart Association. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines [published correction appears in Circulation. 2004;110(6):763]. Circulation. 2004;110(2):227-239.
96. Fleg JL, Aronow WS, Frishman WH. Cardiovascular drug therapy in the elderly: benefits and challenges. Nat Rev Cardiol. 2011;8(1):13-28.
97. American Diabetes Association. Standards of medical care for patients with diabetes mellitus [published correction appears in Diabetes Care. 2003;
26(3):972]. Diabetes Care. 2003;26(suppl 1):S33-S50.
98. Stratton IM, Adler AI, Neil HA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321(7258):405-412.
99. Ravipati G, Aronow WS, Ahn C, Sujata K, Saulle LN, Weiss MB. Association of hemoglobin A1c level with the severity of coronary artery
disease in patients with diabetes mellitus. Am J Cardiol. 2006;97(7):968-969.
100. Aronow WS, Ahn C, Weiss MB, Babu S. Relation of hemoglobin A1c levels to severity of peripheral arterial disease in patients with diabetes mellitus. Am J Cardiol. 2007;99(10):1468-1469.
101. Aronow WS. Hypertension guidelines. Hypertension. 2011;58(3):347-348.
102. Bangalore S, Qin J, Sloan S, Murphy SA, Cannon CP; PROVE-IT-TIMI 22 Trial Investigators. What is the optimal blood pressure in patients after acute coronary syndromes? Relationship of blood pressure and cardiovascular events in the Pravastatin or Atorvastatin Evaluation and Infection Therapy-Thrombolysis in Myocardial Infarction (PROVE IT-TIMI) 22 trial. Circulation. 2010;122(21):2142-2151.
103. Cooper-DeHoff RM, Gong Y, Handberg EM, et al. Tight blood pressure control and cardiovascular outcomes among hypertensive patients with
diabetes and coronary artery disease. JAMA. 2010;304(1):61-68.
104. Cushman WC, Evans GW, Byington RP, et al; ACCORD Study Group. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362(17):1575-1585.
105. Banach M, Bhatia V, Feller MA, et al. Relation of baseline systolic blood pressure and long-term outcomes in ambulatory patients with chronic mild to moderate heart failure. Am J Cardiol. 2011;107(8):1208-1214.
106. Aronow WS, Banach M. Ten most important things to learn from the ACCF/AHA 2011 Expert Consensus Document on Hypertension in the
Elderly. Blood Pressure. In press.


