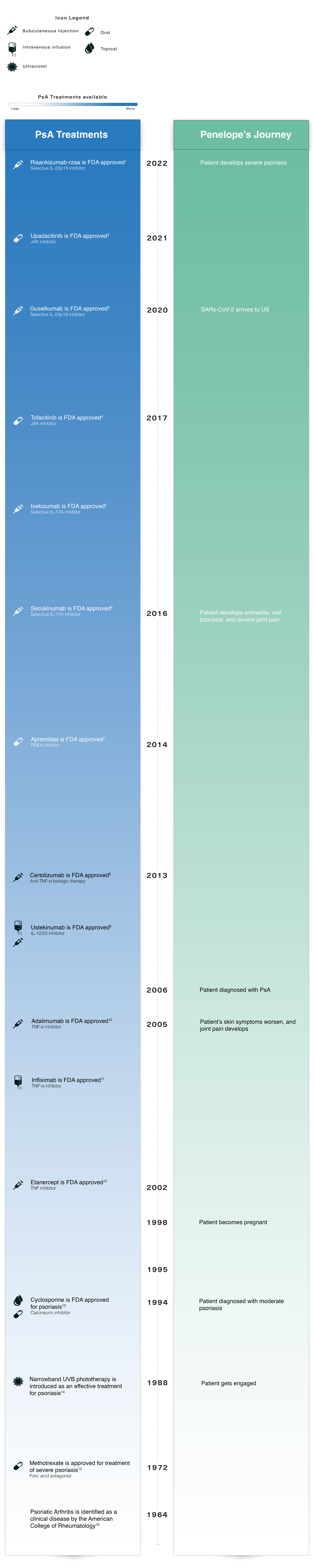
The PsA Treatment Evolution
APRIL 21, 2022
Penelope S. Adams is a patient who has been suffering with symptoms of psoriatic arthritis (PsA) for over 15 years. As she’s battled to manage her dynamic symptoms, the treatment options available to her have evolved. Today, Penelope is at the clinic to discuss her new symptoms, thick, scaley, skin patches on her elbows, and wrist pain making it difficult to pick up her granddaughter. Penelope is looking for a change in her treatment regimen to find relief and get back to her normal activities.
Dive into Penelope's past and discover when PsA became a recognized disease and how treatment options have progressed. Emergence of first-in-class therapies are labeled.
Look below and click on the text to learn more!
- AbbVie. U.S. FDA Approves Second Indication for SKYRIZI (risankizumab-rzaa) to Treat Adults with Active Psoriatic Arthritis. https://news.abbvie.com/news/press-releases/us-fda-approves-second-indication-for-skyrizi-risankizumab-rzaa-to-treat-adults-with-active-psoriatic-arthritis.htm. Published January 21, 2022. Accessed February 10, 2022.
- AbbVie. Rinvoq (upadacitinib) Receives U.S. FDA Approval for Active Psoriatic Arthritis. https://news.abbvie.com/news/press-releases/rinvoq-upadacitinib-receives-us-fda-approval-for-active-psoriatic-arthritis.htm. Published December 14, 2021. Accessed February 10, 2022.
- Johnson and Johnson. Active Psoriatic Arthritis. TREMFYA (guselkumab) Approved by U.S. Food and Drug Administration as the First Selective Interleukin (IL)-23 Inhibitor for Active Psoriatic Arthritis. https://www.jnj.com/tremfya-guselkumab-approved-by-u-s-food-and-drug-administration-as-the-first-selective-interleukin-il-23-inhibitor-for-active-psoriatic-arthritis. Published July 14, 2020. Accessed February 10, 2022.
- Pfizer. Pfizer Announces FDA Approval for Xeljanz (tofacitinib) and Xeljanx XR for the Treatment of Active Psoriatic Arthritis. https://www.pfizer.com/news/press-release/press-release-detailpfizer_announces_fda_approval_of_xeljanz_tofacitinib_and_xeljanz_xr_for_the_ treatment_of_active_psoriatic_arthritis. Published December 14, 2017. Accessed February 10, 2022.
- Lilly. Lilly’s Taltz (ixekizumab) Received U.S. FDA Approval for the Treatment of Active Psoriatic Arthritis. https://investor.lilly.com/news-releases/news-release-details/lillys-taltzr-ixekizumab-receives-us-fda-approval-treatment-0. Published December 1, 2017. Accessed February 10, 2022.
- Novartis. Novartis receives two new FDA approvals for Cosentyx to treat patients with ankylosing spondylitis and psoriatic arthritis in the US. https://www.novartis.com/news/media-releases/novartis-receives-two-new-fda-approvals-cosentyx-treat-patients-ankylosing-spondylitis-and-psoriatic-arthritis-us. Published January 15, 2016. Accessed February 10, 2022.
- Celgene. Otezla (apremilast) – First Oral Therapy Approved by the U.S. Food and Drug Administration for the Treatment of Adults with Active Psoriatic Arthritis. https://ir.celgene.com/press-releases-archive/press-release-details/2014/OTEZLA-apremilast---First-Oral-Therapy-Approved-by-the-US-Food-and-Drug-Administration-for-the-Treatment-of-Adults-with-Active-Psoriatic-Arthritis/default.aspx. Published March 21, 2014. Accessed February 10, 2022.
- UCB. Cimzia (certolizumab pegol) approved by the U.S. FDA for the treatment of adult patients with active psoriatic arthritis. https://www.ucb.com/stories-media/Press-Releases/article/Cimzia-certolizumab-pegol-approved-by-the-U-S-FDA-for-treatment-of-adult-patients-with-active-psoriatic-arthritis. Published September 30, 2013. Accessed February 10, 2022.
- Johnson and Johnson. Stelara (ustekinumab) receives FDA Approval To Treat Active Psoriatic Arthritis. https://www.jnj.com/media-center/press-releases/stelara-ustekinumab-receives-fda-approval-to-treat-active-psoriatic-arthritis. Published September 23, 2013. Accessed February 10, 2022.
- FDA News. Abbott’s HUMIRA (adalimumab) Approved for Psoriatic Arthritis. https://www.fdanews.com/articles/88749-abbott-s-humira-approved-for-psoriatic-arthritis. Published November 14, 2006. Accessed February 10, 2022.
- Hopkins Arthritis Website. Infliximab Now Approved for Use in Psoriatic Arthritis. https://www.hopkinsarthritis.org/arthritis-news/infliximab-now-approved-for-use-in-psoriatic-arthritis/. Published April 13, 2005. Accessed February 10, 2022.
- Amgen. FDA Expands ENBREL Psoriatic Arthritis Indication—ENDBREL Approved as First and Only Treatment to Improve Physical Function in These Patients. https://www.amgen.com/newsroom/press-releases/2005/06/fda-expands-enbrel-psoriatic-arthritis-indication----enbrel-approved-as-first-and-only-treatment-to-improve-physical-function-in-these-patients. Published June 1, 2005." between the url and "Accessed February 10, 2022.
- World History Project. FDA Approves Novartis’s Neoral. https://worldhistoryproject.org/1995/7/14/fda-approves-novartiss-neoral. Published July 14, 1995. Accessed February 10, 2022.
- Green C, et al. Br J Dermatol. 1988;119(6):691-696. doi:10.1111/j.1365-2133.1988.tb03489.x
- Coates LC et al. Met. 2020 Jun; 96:31-35. doi: 10.3899/jrheum.200124
- Blumberg BS et al. Arthritis Rheum. 1964 Feb; 7:93-7. doi:10.1002/art.1780070113
- Singh JA, Guyatt G, Ogdie A, et al. Special Article: 2018 American College of Rheumatology/National Psoriasis Foundation Guideline for the Treatment of Psoriatic Arthritis. Arthritis Rheumatol. 2019;71(1):5-32. doi:10.1002/art.40726
Risankizumab-rzaa Indications & Important Safety Considerations
INDICATIONS
Risankizumab is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
Risankizumab is indicated for the treatment of active psoriatic arthritis in adults.
IMPORTANT SAFETY CONSIDERATIONS
Risankizumab is contraindicated in patients with a history of serious hypersensitivity reaction to risankizumab or any of the excipients. Serious hypersensitivity reactions, including anaphylaxis, may occur. If a serious hypersensitivity reaction occurs, discontinue risankizumab and initiate appropriate therapy immediately.
Risankizumab may increase the risk of infection. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur. If such an infection develops, discontinue risankizumab until the infection resolves. Evaluate patients for tuberculosis infection prior to initiating treatment with risankizumab.
Avoid use of live vaccines in patients treated with risankizumab.
The most common adverse reactions (≥1%) are upper respiratory infections, headache, fatigue, injection site reactions, and tinea infections.
Upadacitinib Indications, Important Safety Considerations, and Boxed Warning
INDICATIONS
Upadacitinib is a Janus kinase (JAK) inhibitor indicated for the treatment of:
Active psoriatic arthritis (PsA) in adults who have had an inadequate response or intolerance to one or more TNF blockers.
Limitations of Use for RA and PsA: Use of upadacitinib in combination with other JAK inhibitors, biologic DMARDs, or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
Adults and pediatric patients 12 years of age and older with refractory moderate to severe atopic dermatitis whose disease is not adequately controlled with other systemic drug products, including biologics, or when use of those therapies are inadvisable.
Limitations of Use for AD: Use of upadacitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or with other immunosuppressants.
IMPORTANT SAFETY CONSIDERATIONS AND BOXED WARNING
Serious Infections: Patients treated with upadacitinib are at increased risk for developing serious infections that may lead to hospitalization or death. These infections include tuberculosis (TB), invasive fungal, bacterial, viral, and other infections due to opportunistic pathogens. Most patients who developed these infections were taking concomitant immunosuppressants, such as methotrexate or corticosteroids. Test for latent TB before and during therapy; treat latent TB prior to use. Consider the risks and benefits prior to initiating therapy in patients with chronic or recurrent infection. If a serious infection develops, interrupt upadacitinib until the infection is controlled.
Mortality: In a postmarketing safsameety study in RA patients ≥ 50 years of age with at least one cardiovascular (CV) risk factor comparing another JAK inhibitor to TNF blockers, a higher rate of all-cause mortality, including sudden CV death, was observed with the JAK inhibitor.
Malignancies: Malignancies have been observed in upadacitinib treated patients. In RA patients treated with another JAK inhibitor, a higher rate of lymphomas and lung cancers was observed when compared with TNF blockers. Patients who are current or past smokers are at additional increased risk. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with upadacitinib, particularly in patients with a known malignancy (other than a successfully treated non-melanoma skin cancer), patients who develop a malignancy when on treatment, and patients who are current or past smokers.
Major Adverse Cardiovascular Events (MACE): In RA patients who were ≥ 50 years of age with at least one CV risk factor treated with another JAK inhibitor, a higher rate of MACE (CV death, myocardial infarction, and stroke) was observed compared with TNF blockers. Patients who are current or past smokers are at additional increased risk. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with upadacitinib. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur. Discontinue upadacitinib in patients that have experienced a myocardial infarction or stroke.
Thrombosis: Thrombosis, including deep vein thrombosis, pulmonary embolism, and arterial thrombosis, have occurred in patients treated with JAK inhibitors, including upadacitinib. Many of these adverse events were serious and some resulted in death. In RA patients who were ≥ 50 years of age with at least one CV risk factor treated with another JAK inhibitor, a higher rate of thrombosis was observed when compared with TNF blockers. Avoid upadacitinib in patients at risk. Patients with symptoms of thrombosis should discontinue upadacitinib and be promptly evaluated.
Hypersensitivity Reactions: Upadacitinib is contraindicated in patients with known hypersensitivity to upadacitinib or any of its excipients. Serious hypersensitivity reactions such as anaphylaxis and angioedema were reported in patients receiving upadacitinib in clinical trials. If a clinically significant hypersensitivity reaction occurs, discontinue upadacitinib and institute appropriate therapy.
Other Serious Adverse Reactions: Patients treated with upadacitinib also may be at risk for other serious adverse reactions, including gastrointestinal perforations, neutropenia, lymphopenia, anemia, lipid elevations, liver enzyme elevations, and embryo-fetal toxicity.
Vaccinations: Avoid use of live vaccines during, or immediately prior to, upadacitinib therapy. Prior to initiating upadacitinib, it is recommended that patients be brought up to date with all immunizations, including varicella zoster or prophylactic herpes zoster vaccinations, in agreement with current immunization guidelines.
Common Adverse Reactions in RA and PsA: The most common adverse reactions (≥1%) are upper respiratory tract infections, herpes zoster, herpes simplex, bronchitis, nausea, cough, pyrexia, and acne.
Adalimumab Indications and Important Safety Considerations
HUMIRA is a tumor necrosis factor (TNF) blocker indicated for:
Psoriatic Arthritis (PsA): reducing signs and symptoms, inhibiting the progression of structural damage, and improving physical function in adult patients with active PsA.
Plaque Psoriasis (Ps): treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy, and when other systemic therapies are medically less appropriate.
Serious Infections
Patients treated with HUMIRA are at increased risk for developing serious infections that may lead to hospitalization or death. These infections include active tuberculosis (TB), reactivation of latent TB, invasive fungal infections, and bacterial, viral, and other infections due to opportunistic pathogens. Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
Malignancies
Lymphoma and other malignancies, some fatal, have been reported in patients treated with TNF blockers, including HUMIRA. Post-marketing cases of hepatosplenic T-cell lymphoma (HSTCL), a rare type of T-cell lymphoma, have occurred in adolescent and young adults with inflammatory bowel disease treated with TNF blockers including HUMIRA.
Other Serious Adverse Reactions
Patients treated with HUMIRA also may be at risk for other serious adverse reactions, including anaphylaxis, hepatitis B virus reactivation, demyelinating disease, cytopenias, pancytopenia, heart failure, and a lupus-like syndrome.
Common Adverse Reactions
The most common adverse reactions (>10%) are: infections (e.g., upper respiratory, sinusitis), injection site reactions, headache, and rash.
Review accompanying SkyriziTM (risankizumab-rzaa), RinvoqTM (upadacitinib) and Humira® (adalimumab) full Prescribing Information. For additional information, visit www.rxabbvie.com or contact AbbVie medical information at 1-800-633-9110.
The US Medical Affairs department of AbbVie Inc. is the copyright owner of this presentation and has paid Consultant360 to host this content. AbbVie is solely responsible for all written content within this presentation.
© 2022 AbbVie Inc. All rights reserved. RISN-US-00008-MC v1.0 Approved Feb 2022
Risankizumab is approved to treat adult patients with active psoriatic arthritis.
MOA: Selective IL-23p19 inhibitor
FDA Approval: January 2022
Route of Administration: Subcutaneous Drug Class: Interleukin inhibitors, IL-23 inhibitor, Biologics
Notes: Approved for adults with active psoriatic arthritis is administered at Week 0, Week 4, and every 12 weeks thereafter.
Upadacitinib is approved for the treatment of adults with active PsA who had an inadequate response or intolerance to one or more TNF blockers.
MOA: Janus kinase (JAK) inhibitor
FDA Approval: December 2021
Route of Administration: Oral
Drug Class: JAK inhibitors, Targeted Synthetic DMARD
Notes: Second approved indication for upadacitinib following moderately to severely active rheumatoid arthritis in 2019. Learn more here.
Guselkumab is approved for adult patients with active psoriatic arthritis.
MOA: Selective IL-23p19 inhibitor
FDA Approval: July 2020
Route of Administration: Subcutaneous Drug Class: Interleukin inhibitors, IL-23 inhibitor, Biologics
Notes: First treatment approved that selectively inhibits IL-23.
Tofacitinib is approved for the treatment of adults with active PsA who have an inadequate response to, or cannot tolerate, methotrexate or other DMARDs.
MOA: Janus kinase (JAK) inhibitor
FDA Approval: December 2017
Route of Administration: Oral
Drug Class: JAK Inhibitor, Targeted Synthetic DMARD
Notes: First oral JAK inhibitor in the US approved for moderate to severe rheumatoid arthritis, active psoriatic arthritis and ankylosing spondylitis in patients who have an inadequate response to, or cannot tolerate methotrexate or other DMARDs.
Ixekizumab is approved for the treatment of adult patients with active psoriatic arthritis.
MOA: Selective interleukin-17A (IL-17A) inhibitor
FDA Approval: December 2017
Route of Administration: Subcutaneous Drug Class: Interleukin inhibitors, IL-17 inhibitor, Biologics
Notes: First approved in March 2016 for the treatment of adult patients with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
Secukinumab is approved for the treatment of psoriatic arthritis in patients aged 2 years and older.
MOA: Selective interleukin-17A (IL-17A) inhibitor
FDA Approval: January 2016
Route of Administration: Subcutaneous Drug Class: Interleukin inhibitors, IL-17 inhibitor, Biologics
Notes: First IL-17A inhibitor approved for patients with psoriatic arthritis.
Apremilast is approved for the treatment of adults with active psoriatic arthritis.
MOA: Selective phosphodiesterase (PDE) 4 inhibitor
FDA Approval: March 2014
Route of Administration: Oral
Drug Class: PDE4 inhibitor, Oral Small Molecule
Notes: First oral therapy approved for treatment of adults with psoriatic arthritis.
Certolizumab is approved for the treatment of adult patients with active psoriatic arthritis.
MOA: Certolizumab is a PEGylated anti-TNF-α biologic therapy
FDA Approval: September 2013
Route of Administration: Subcutaneous Drug Class: TNF-α inhibitor, Biologics
Notes: Systemic exposure to a breastfed infant is expected to be low because certolizumab pegol is a large molecule and is degraded in the gastrointestinal tract.
Ustekinumab is approved to treat adult patients living with active PsA, alone or in combination with metrotrexate.
MOA: Human IgG1 monoclonal antibody that targets the shared subunit p40 of IL-12 and IL-23
FDA Approval: September 2013
Route of Administration: Intravenous or subcutaneous
Drug Class: Interleukin inhibitors, IL-12/23 inhibitor, Biologics
Notes: First IL-12/23 treatment approved for adult patients living with psoriatic arthritis.
Adalimumab is approved for reducing signs and symptoms, inhibiting the progression of structural damage, and improving physical function in adult patients with active PsA.
MOA: Recombinant human IgG1 monoclonal antibody specific for human TNF-α
FDA Approval: October 2005
Route of Administration: Subcutaneous Drug Class: TNF-α inhibitor, Biologics
Notes: Adalimumab is typically given at 2-week intervalsIn treatment-naive patients with active PsA, a TNF blocker is recommended over an oral small molecule such as methotrexate as a first-line option.17
Infliximab is approved for reducing signs and symptoms of active PsA, inhibiting the progression of structural damage, and improving physical function in adult patients.
MOA: Chimeric IgG1 monoclonal antibody which binds to and neutralizes soluble and membrane bound TNF-α
FDA Approval: May 2005
Route of Administration: Intravenous Drug Class: TNF-α inhibitor, Biologics
Notes: Infliximab is given typically at six- to eight-week intervals. In treatment-naive patients with active PsA, a TNF blocker is recommended over an oral small molecule such as methotrexate as a first-line option.17
Etanercept is indicated for reducing signs and symptoms, inhibiting the progression of structural damage of active arthritis, and improving physical function in patients with PsA. Etanercept can be used with or without methotrexate.
MOA: Etanercept is a TNF p75 soluble receptor construct
FDA Approval: January 2002
Route of Administration: Subcutaneous Drug Class: TNF Blocker, Biologics
Notes: For adult patients step dosing is recommended. Twice daily injections for the first 3 months and once a day thereafter. In treatment-naive patients with active PsA, a TNF blocker is recommended over an oral small molecule such as methotrexate as a first-line option.17
Cyclosporine is approved for treating plaque psoriasis.
MOA: Calcineurin inhibitor acting as an immunosuppressant.
FDA Approval: July 1995
Route of Administration: Oral, Topical Drug Class: Calcineurin inhibitors, immunomodulators
Notes: The discovery of cyclosporine marked a turning point in the history of psoriasis treatment and the direction of future translational research. A deeper understanding of the immune system would lead to more effective disease control.
Narrowband UVB phototherapy introduced as an effective treatment for psoriasis.
MOA: Alters antigen-presenting cell activity lowering peripheral NK-cell activity, lymphocyte proliferation, and cytokine production.
FDA Approval: N/A
Route of Administration: Topical
Drug Class: Phototherapy
Methotrexate is approved for treatment of severe psoriasis.
MOA: Folic acid antagonist
FDA Approval: March 1972
Route of Administration: Oral
Drug Class: Conventional synthetic disease-modifying antirheumatic drug (csDMARD)
Notes: Has not been approved for PsA but is recommended for use in some PsA patients.17
Text here
February 2022
Penelope is having an inadequate response to her current treatment. Her skin condition is worsening with thick, scaley patches on her elbows and wrist pain making it difficult to pick up her granddaughter.
Her doctor discusses current PsA treatment options to determine if her medication should be switched.
March 2020
Penelope worries about the strength of her immune system and risk of contracting COVID-19. She discusses whether she should discontinue her medication with her doctor.
June 2016
Penelope has worsening psoriatic arthritis symptoms, including the development of enthesitis in the hands, nail psoriasis, and severe joint pain. Management with NSAIDs and over the counter medication is no longer feasible.
Her doctor discusses current PsA treatment options to determine if her medication should be switched.
December 2006
Penelope is noticing she needs to stretch when she wakes up to be able to improve the stiffness in her knees, ankles, and knuckles. She sees a rheumatologist. A full diagnostic battery is conducted, including X-rays of the limbs and blood work, specifically testing for Rheumatoid Factor (RF) and C-reactive protein
(CRP).
Her doctor discusses current PsA treatment options to determine if her medication should be switched.
March 2005
Penelope’s psoriasis has worsened. She has more visible plaques and she worries about needing to cover her arms in the approaching summer. A few of her fingers are also swollen, even though it is late winter. The dermatologist is concerned and refers her to a rheumatologist.
May 1998
During Penelope’s third trimester, she developed heel pain and found it difficult to walk the short distance from her parking spot to work. She goes to see a podiatrist and is diagnosed with plantar fasciitis.
April 1994
Motivated to improve her skin condition, Penelope gets a referral from her primary care physician to see a dermatologist. Penelope is diagnosed with moderate psoriasis.
Her doctor discusses current psoriasis treatment options.
January 1988
Penelope’s anxiety sets in while dress shopping when she notices her skin rash has gotten worse. The rash can now be seen on her elbows, wrists, and hands. She contemplates getting gloves to match her dress.


