Peer Reviewed
A Collection of Dermatologic Conditions
Urticaria Pigmentosa
Authors:
Lisa LaVallee, MD, and Kelly Garcia, MD
Mountain Area Health Education Center, Asheville, North Carolina
Citation:
LaVallee L, Garcia K. Urticaria pigmentosa. Consultant. 2017;57(8):493-494.
An infant girl presented for her 2-month well-child checkup. Her mother reported that the infant had recently developed new skin lesions, the first of which had been a red nodular lesion in her right axilla. Subsequently, the girl had developed other red lesions, some raised and some flat, on her trunk, neck, scalp, and proximal extremities. The lesions did not seem to bother her.
History. The patient had been born at term via uncomplicated vaginal delivery, and she had had no neonatal complications. She was growing well, eating well, and meeting developmental milestones, and she had not been ill. The family had a dog that recently had been treated for fleas, but no one else in the household had any skin lesions.
Physical examination. Examination revealed several heterogeneous lesions. The axillary lesion (Figure 1) was nodular, and multiple maculopapular lesions were present on her neck and trunk (Figure 2). A few of the lesions appeared to have crusting.

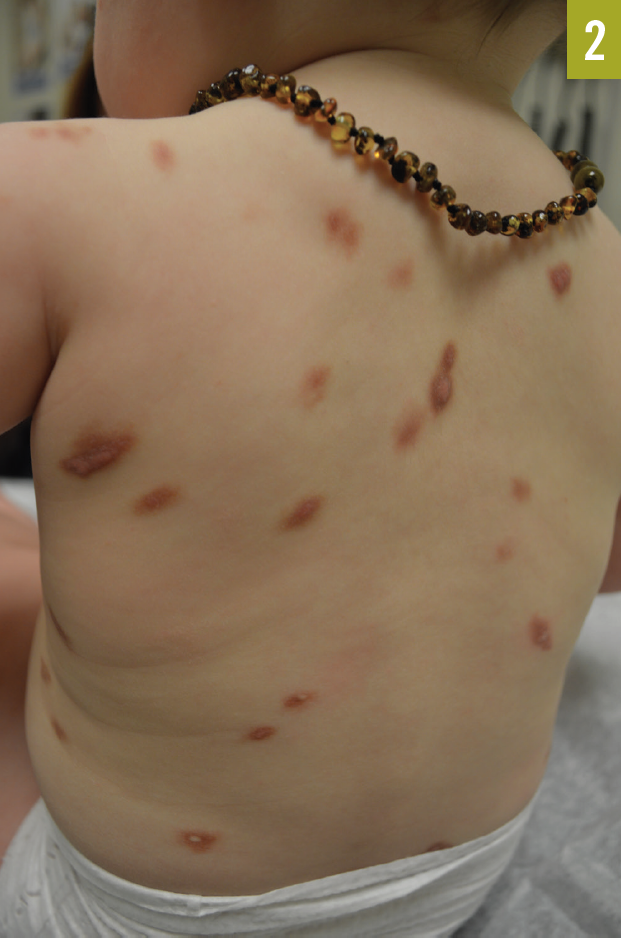
The patient was prescribed an antibiotic ointment for the lesions that appeared to be impetigo. At a 1-month follow-up visit, the patient’s mother reported that the antibiotic ointment had not helped, and that the infant had developed more lesions. The original lesions appeared to be unchanged. The girl was otherwise well and was still developing normally.
The color, location, heterogeneity, and time course of the lesions suggested a diagnosis of urticaria pigmentosa (UP). Results of skin biopsy testing confirmed the diagnosis.
Outcome of the case. The patient was referred to a dermatologist, who in turn referred her to an allergist and a pediatric hematologist. Results of a complete blood cell count, a comprehensive metabolic panel, and a tryptase test ordered by the allergist to screen for systemic involvement were unremarkable, and no further testing was recommended. The patient was prescribed cetirizine as needed for pruritus, as well as an epinephrine auto-injector for emergency use.
At her most recent visit, the patient’s mother reported that the infant had had 2 episodes of flushing without obvious trigger. Both times, her caregiver had treated her with oral diphenhydramine, and her symptoms had resolved within a few minutes. Her family has been reassured that the girl’s lesions are expected to regress before or during adolescence.
Discussion. UP, also known as maculopapular cutaneous mastocytosis, is the most common form of cutaneous mastocytosis, a family of rare diseases caused by a proliferation of mast cells within the skin.1 The majority of cases are related to a point mutation in the proto-oncogene kit, which is involved in signaling mast cells to divide. Other recognized forms of cutaneous mastocytosis include diffuse cutaneous mastocytosis, nodular mastocytoma, and plaque-type cutaneous mastocytosis.2 The incidence of these diseases has not been well defined.
UP usually presents within the first 2 years of life but can also present in adulthood. Many children with UP have persistent lesions for years, although the lesions resolve by adolescence in approximately two-thirds of cases.3 Rarely in infantile cases, but more commonly in adult cases, the patient actually has systemic mastocytosis, in which mast cells also proliferate in the bone marrow, liver, spleen, intestines, or other organs. Systemic mastocytosis can manifest with symptoms such as vomiting and diarrhea, bone pain, or even headaches and depression.1 Rare cases have been reported of patients with UP developing mast cell sarcoma or mast cell leukemia.3
The diagnosis of UP is usually based on the history and physical examination findings, although definitive diagnosis is made with punch biopsy.1 Characteristic lesions are tan, brown, or red and are maculopapular. They are most commonly found on the extremities, trunk, and abdomen, although children may also have involvement of the face and scalp. When scratched or rubbed, the skin surrounding the lesions may become erythematous or develop urticaria due to activation of the mast cells in the lesion (the Darier sign).
Patients with UP may have episodes of pruritus, flushing, or nausea triggered by a variety of stimuli, including temperature changes, emotional stress, exercise, alcohol ingestion, and nonsteroidal anti-inflammatory drug use. Symptoms can be controlled with antihistamines or leukotriene-modifying agents. Oral corticosteroids and psoralen–UV-A may be considered for short-term therapy of acute flares. Special consideration should be given to anesthetic choices, and providers should be ready to treat mast cell degranulation and cardiovascular collapse in the postoperative period, since many anesthetic drugs have a theoretical risk of causing mast cell degranulation.4
REFERENCES:
- Akin C, Valent P. Diagnostic criteria and classification of mastocytosis in 2014. Immunol Allergy Clin North Am. 2014;34(2):207-218.
- Tüysüz G, Özdemir N, Apak H, Kutlubay Z, Demirkesen C, Celkan T. Childhood mastocytosis: results of a single center. Turk Pediatri Ars. 2015;50(2):108-113.
- Méni C, Bruneau J, Georgin-Lavialle S, et al. Paediatric mastocytosis: a systematic review of 1747 cases. Br J Dermatol. 2015;172(3):642-651.
- Tew S, Taicher BM. Avoiding cardiovascular collapse: pediatric cutaneous mastocytosis and anesthetic challenges. A A Case Rep. 2015;5(10):179-181.
NEXT: Sago Palm Phytodermatitis
Sago Palm Phytodermatitis
Authors:
MaryJo E. Kramer, BS; Jessica Parsons, MD; and Rajani Katta, MD
The Georgetown University School of Medicine, Washington, DC
Citation:
Kramer ME, Parsons J, Katta R. Sago palm phytodermatitis. Consultant. 2017;57(8):494-495.
A 52-year-old woman presented with itchy red papules on her forearms (Figure 1). She reported that the lesions had developed after contact with her sago palm, while she had been removing seeds from the central part of the plant to protect her dog, given that the seeds are poisonous. Despite her having worn long sleeves and heavy rubber gloves, she had experienced multiple punctures and scratches on the forearms. She reported that complete avoidance of the sharp palm leaves had been impossible, since she needed to reach the center of the plant to remove the seeds.

Treatment with topical hydrocortisone cream, 1%, was initiated. The patient reported that the lesions had improved after 10 days of therapy.
Discussion. While the sago palm (Cycas revoluta) is commonly used as a decorative landscaping plant, it must be approached with caution. The sago palm is actually not a palm but rather is a cycad. Although the toxic effects of ingestion of cycads have been well described in the literature, the dermatologic effects of physical contact with the plant have been rarely reported despite being well known and discussed in the field of horticulture.
Cycads are cold-hardy plants that can grow almost anywhere in the United States despite being native to tropical and subtropical regions. Cycads typically grow to 3 m high and 2 m wide, with sharp, shiny, dark-green leaves and a central cone (Figure 2).1 Cycads are popular ornamental plants used in landscaping, and they can be purchased at most large nurseries across the United States. Despite its common use as a decorative plant, the sago palm is known to contain high levels of toxins, including cycasin and β-methylamino-l-alanine. These toxins are found in all parts of the plant and at highest concentrations in the seeds and roots.2,3 In addition to causing nausea and vomiting, hepatotoxicity, hematochezia, melena, and hemarthrosis, dogs and cats were shown to have a 50% to 75% chance of mortality only 12 hours after ingestion of the cycad fruit, seeds, or young leaves.4

The potential skin effects resulting from plant contact are well known. The skin findings demonstrated in our patient’s case are believed to represent a physical irritant contact dermatitis (ICD) around the puncture points from the sharp cycad plant. Physical irritants, such as sharp thorns, result in mechanical irritant contact dermatitis (MICD) via mechanical trauma or microtrauma to the involved skin. These areas of microtrauma may enhance secondary cutaneous penetration of chemical irritants or infectious agents.5 It is therefore important to monitor response to treatment, since infectious complications have been reported secondary to skin puncture, via lodging of thorny debris carrying fungal toxins or soil bacteria inside the wound. Because some plants contain chemical irritants, MICD may predispose to chemical ICD as well, as in the case of plants that contain oxalate crystals.
Treatment recommendations for ICD due to the sago palm are not well reported in the literature. Topical corticosteroids are often used in the treatment of ICD and were used in this patient’s case. Lipid-rich moisturizers and barrier creams containing ceramides also may be used to treat irritation.6
If exposure to the sago palm is unavoidable, the use of protective equipment, such as gloves and long-sleeved clothing, is important, although this may not fully prevent contact with the skin.
References:
- Northrop RJ, Andreu MG, Friedman MH, McKenzie M, Quintana HV. Cycas revoluta, sago palm. University of Florida/Institute of Food and Agricultural Sciences Extension. Document FOR 254. http://edis.ifas.ufl.edu/fr316. Published May 2010. Reviewed April 2016. Accessed July 19, 2017.
- Albretsen JC, Khan SA, Richardson JA. Cycad palm toxicosis in dogs: 60 cases (1987-1997). J Am Vet Med Assoc. 1998;213(1):99-101.
- Johnson A, Johnson SJ. Garden plants poisonous to people. Primefacts. 2006(359):1-12. https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0008/112796/garden-plants-poisonous-to-people.pdf. Accessed July 19, 2017.
- Ferguson D, Crowe M, McLaughlin L, Gaschen F. Survival and prognostic indicators for cycad intoxication in dogs. J Vet Intern Med. 2011;25(4):831-837.
- Watkins JB III. Toxic effects of plants and animals. In: Klaassen CD, ed. Casarett and Doull’s Toxicology: the Basic Science of Poisons. 8th ed. New York, NY: McGraw-Hill Education; 2013:1131-1168.
- Modi GM, Doherty CB, Katta R, Orengo IF. Irritant contact dermatitis from plants. Dermatitis. 2009;20(2):63-78.
NEXT: Pustular Psoriasis
Pustular Psoriasis
Authors:
John D. Peters, MD, and Jason S. Ballin, DO
United States Navy Medical Corps
Citation:
Peters JD, Ballin JS. Pustular psoriasis. Consultant. 2017;57(8):495-497.
A n 8-year-old girl with a 4-year history of chronic intermittent eruption of a pustular rash on her torso presented for evaluation in the dermatology clinic with an ongoing flare. The current rash had persisted despite the application of topical fluocinonide, topical clobetasol, a selenium sulfide wash, and oral cephalexin.
Physical examination. The girl’s flanks demonstrated well-demarcated, annular, figurate erythematous plaques with circumferential, peripheral scale and discrete pustules, occupying 15% to 20% of body surface area (Figures 1 and 2). The patient had no associated systemic symptoms.
Diagnostic tests. Laboratory test findings were significant for a neutrophilic leukocytosis (14,200 white blood cells/µL). Histologic review of a punch biopsy specimen demonstrated an epidermal and superficial perivascular neutrophilic infiltrate with overlying serum crust. Results of Grocott methenamine silver and periodic acid–Schiff stains were unremarkable. The results of further evaluation with a basic metabolic panel, serologic testing for hepatitis B, and purified protein derivative skin testing for tuberculosis were normal or negative.

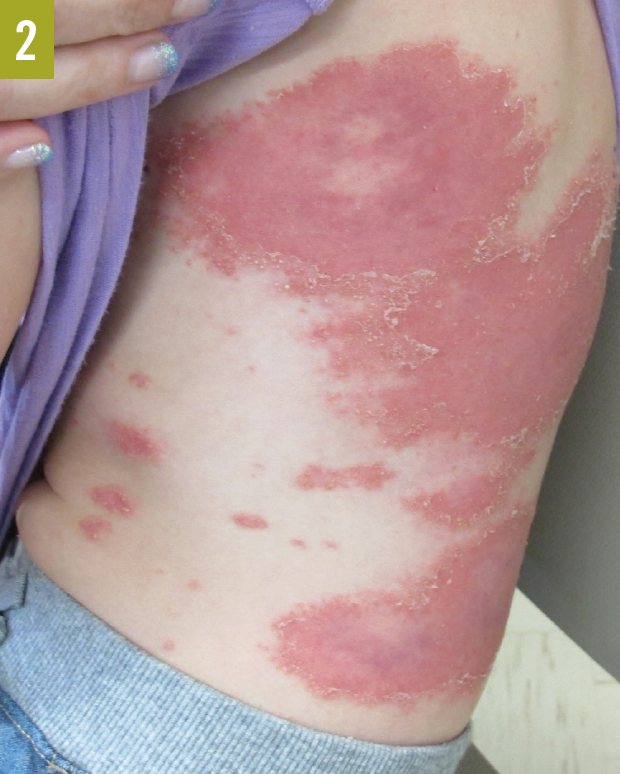
Based on her presentation and laboratory test results, the patient received a diagnosis of pustular psoriasis.
Outcome of the case. A regimen of weekly subcutaneous etanercept injections was initiated, which led to near complete resolution of the plaques over the course of several months without perceivable adverse effects (Figures 3 and 4 show the patient at 6-month follow-up).
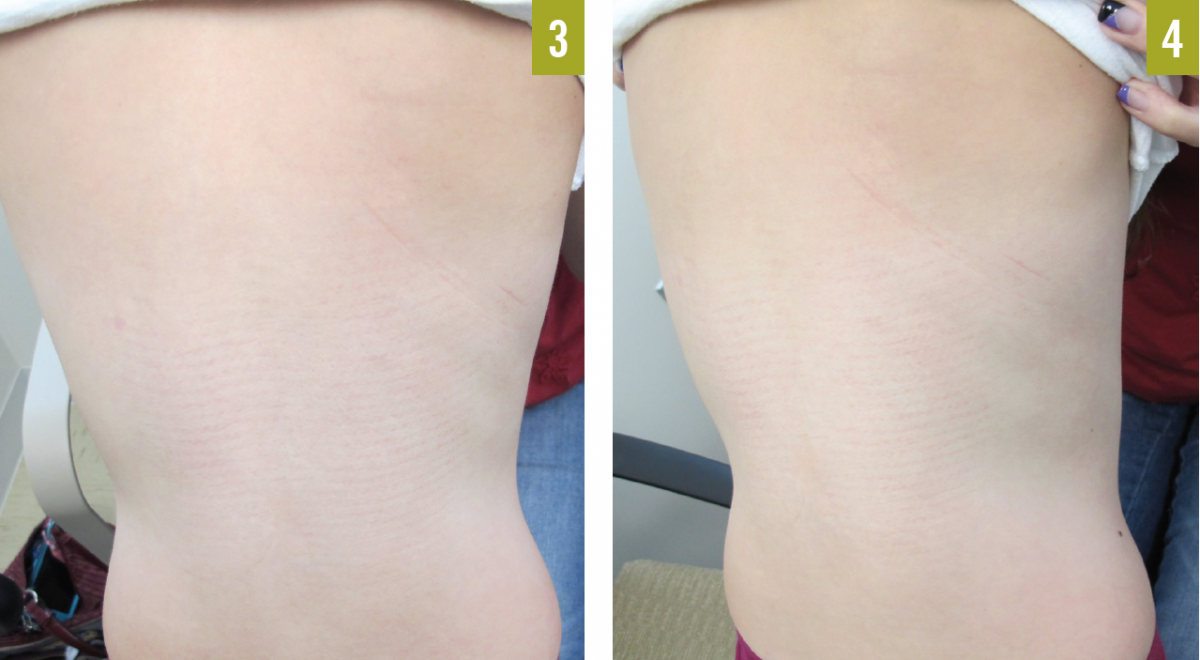
Discussion. Pustular psoriasis is an uncommon variant of psoriasis that can develop independently or in association with preexisting psoriasis. The exact prevalence of generalized pustular psoriasis (GPP) is unknown. It is most often seen in middle-aged adults but can occur in children,1,2 as this patient’s case illustrates.
The development of pustular psoriasis has been linked to upper respiratory tract infection, pregnancy, withdrawal of corticosteroids, hypocalcemia, and the use of various medications, including interferon, lithium, penicillin, cyclosporine, salicylates, and tumor necrosis factor α (TNF-α) inhibitors.1,3-9 It is often observed in patients with a history of psoriasis.1,2
Pustular psoriasis is characterized by an acute, subacute, or chronic eruption of sterile pustules associated with erythema and inflammation. This patient’s case represents the annular variant (also called subacute GPP), characterized by pustules on the periphery of expanding erythematous plaques with central healing, most often localized to the trunk. The acute variant (also called von Zumbusch psoriasis) is characterized by the abrupt onset of numerous pustules on a background of widespread erythema with associated systemic symptoms such as fever, malaise, headache, nausea, arthralgia, and skin tenderness. The exanthematous type, acute generalized exanthematous pustulosis (AGEP), is precipitated by medications or infection and is usually not accompanied by systemic symptoms. The localized variant involves eruption of pustules on the palms, soles, fingertips, and/or nail beds.1,2
Laboratory analysis commonly demonstrates leukocytosis of up to 30,000 cells/µL, absolute lymphocytopenia, an elevated erythrocyte sedimentation rate, hypocalcemia, transaminitis, hypoalbuminemia, and/or an elevated antistreptolysin antibody level. Cultures of the pustules will usually not yield significant growth of pathogenic organisms unless superinfection is present.1 The diagnosis is solidified with skin biopsy findings, which commonly demonstrate classic psoriasiform changes, including parakeratosis and elongation of the rete ridges, along with an epidermal neutrophil infiltrate, upper dermal perivascular lymphocyte infiltrate, and intraspinous aggregates of neutrophils known as spongiform pustules of Kogoj.10
The differential diagnosis mainly includes dermatitis with secondary infection, subcorneal pustular dermatosis (Sneddon-Wilkinson disease), immunoglobulin A pemphigus, tinea corporis, and AGEP.11 Distinguishing GPP from AGEP can be challenging, since the clinical presentations are nearly identical. However, AGEP is more likely to be associated with antibiotic use.12 Biopsy findings that favor AGEP include a lack of significant psoriasiform changes, the presence of a mixed neutrophil-rich infiltrate, eosinophils and necrotic keratinocytes in the dermis, and lack of tortuous blood vessels.10
Patients with acute GPP may require hospitalization to ensure adequate supportive care. Causative drugs must be identified and discontinued. First-line systemic agents include acitretin, methotrexate, infliximab, and cyclosporine.13 Topical therapies are used as adjuvants to systemic therapy. Systemic glucocorticoids can induce rapid improvement in GPP but have been identified as a potential precipitant.
A recent review comparing the efficacy and safety of biologic agents in the treatment of pediatric GPP concluded that etanercept should be considered before infliximab and adalimumab as a first-line biologic agent due its comparatively favorable efficacy and safety profile.14 However, the eruption of lesions of pustular psoriasis has been associated with TNF-α inhibitors.15,16
Chief features of the disease are its tendency to recur and its persistence. The course of GPP is often associated with periods of quiescence and recurrence over the course of years. However, with therapy, flares may be avoided.1
References:
- Choon SE, Lai NM, Mohammad NA, Nanu NM, Tey KE, Chew SF. Clinical profile, morbidity, and outcome of adult-onset generalized pustular psoriasis: analysis of 102 cases seen in a tertiary hospital in Johor, Malaysia. Int J Dermatol. 2014;53(6):676-684.
- Borges-Costa J, Silva R, Gonçalves L, Filipe P, Soares de Almeida L, Marques Gomes M. Clinical and laboratory features in acute generalized pustular psoriasis: a retrospective study of 34 patients. Am J Clin Dermatol. 2011;12(4):271-276.
- Georgala S, Koumantaki E, Rallis E, Papadavid E. Generalized pustular psoriasis developing during withdrawal of short-term cyclosporin therapy. Br J Dermatol. 2000;142(5):1057-1058.
- Sugiura K, Shoda Y, Akiyama M. Generalized pustular psoriasis triggered by amoxicillin in monozygotic twins with compound heterozygous IL36RN mutations: comment on the article by Navarini et al. J Invest Dermatol. 2014;134(2):578-579.
- Wu M-C, Lee J-Y. Generalized flare of pustular psoriasis induced by PEGylated interferon-α2b therapy for chronic hepatitis C. Australas J Dermatol. 2012;53(4):e69-e72.
- Shmidt E, Wetter DA, Ferguson SB, Pittelkow MR. Psoriasis and palmoplantar pustulosis associated with tumor necrosis factor-α inhibitors: the Mayo Clinic experience, 1998 to 2010. J Am Acad Dermatol. 2012;67(5):e179-e185.
- Oumeish OY, Parish JL. Impetigo herpetiformis. Clin Dermatol. 2006;24(2):101-104.
- Lowe NJ, Ridgway HB. Generalized pustular psoriasis precipitated by lithium carbonate. Arch Dermatol. 1978;114(12):1788-1789.
- Katz M. Seidenbaum M, Weinrauch L. Penicillin-induced generalized pustular psoriasis. J Am Acad Dermatol. 1987;17(5 pt 2):918-920.
- Kardaun SH, Kuiper H, Fidler V, Jonkman MF. The histopathological spectrum of acute generalized exanthematous pustulosis (AGEP) and its differentiation from generalized pustular psoriasis. J Cutan Pathol. 2010;37(12):1220-1229.
- Kalb RE. Pustular psoriasis: pathogenesis, clinical manifestations, and diagnosis. UpToDate. http://www.uptodate.com/contents/pustular-psoriasis-pathogenesis-clinical-manifestations-and-diagnosis. Updated December 30, 2016. Accessed July 19, 2017.
- Duckworth L, Maheshwari MB, Thomson MA. A diagnostic challenge: acute generalized exanthematous pustulosis or pustular psoriasis due to terbinafine. Clin Exp Dermatol. 2012;37(1):24-27.
- Robinson A, Van Voorhees AS, Hsu S, et al. Treatment of pustular psoriasis: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2012;67(2):279-288.
- Collamer AN, Battafarano DF. Psoriatic skin lesions induced by tumor necrosis factor antagonist therapy: clinical features and possible immunopathogenesis. Semin Arthritis Rheum. 2010;40(3):233-240.
- Saikaly SK, Mattes M. Biologics and pediatric generalized pustular psoriasis: an emerging therapeutic trend. Cureus. 2016;8(6):e652.
- Ephrem G, Jour G, Smith BL. Successful treatment of von Zumbusch generalized pustular psoriasis with cyclosporine after eruption post etanercept injection. J Med Liban. 2011;59(3):168-169.


