Peer Reviewed
Erythema Multiforme Major Following COVID-19 Infection
AFFILIATIONS:
1Resident, Internal Medicine Department, Iowa Medical Education Collaborative, Des Moines, Iowa
2Internist, MercyOne Des Moines Medical Center, Des Moines, Iowa
CITATION:
Yang J, Sanders T, Voelsvhow E, Guervara-Hernandex M. Erythema multiforme major following COVID-19 infection. Consultant. 2023;63(5):e5. doi:10.25270/con.2023.03.000001
Received March 29, 2022. Accepted June 4, 2022. Published online March 2, 2023.
DISCLOSURES:
The authors report no relevant financial relationships.
DISCLAIMER:
The authors report that informed patient consent was obtained for publication of the images used herein.
CORRESPONDENCE:
Johnathone Yang, DO, Iowa Medical Education Collaborative, 900 65th Street, Windsor Height, Iowa, 50324 (johnathone.yang@commonspirit.org)
Introduction. A young man presented to the hospital with an atypical cutaneous complication of COVID-19: a disseminated targetoid and pruritic rash.
Key words: COVID-19 infection, SARS-CoV-2, erythema multiforme major, post–COVID-19 cutaneous complication
Patient history. A 26-year-old man with a history of type 1 diabetes mellitus and recent diagnosis of COVID-19 was admitted to the hospital for a disseminated targetoid rash, which had been diagnosed 14 days earlier. The rash originated on his lips and eyes before spreading to his entire body, including the palms of the hands, soles of his feet, trunk, and genital region. In addition to the rash, he had crusting to his bilateral eyelids with purulent fluid, frontal headache, subjective fevers, chills, and odynophagia. He denied any photophobia, cough, nausea, vomiting, diarrhea, dysuria, or urethral discharge. There was no history of recent travel or camping, use of new hygiene products, changes to his clothes or food, presence of pets at home, prior diagnosis of sexually transmitted infections (despite having a prior female partner with herpes simplex virus [HSV]), or sexual intercourse with men. His recent COVID-19 diagnosis was made via a home testing kit 14 days prior to admission. This home test was prompted by 3 days of loss of taste and smell. He was not given any treatment as he did not have any respiratory symptoms.
Physical examination. His initial vital signs were as follows: temperature 102.6°F; blood pressure 129/86 mm Hg; heart rate 117 beats/min; respiratory rate 16 breaths/min; and oxygen saturation level 96% on room air. Physical examination was remarkable for nuchal rigidity, white plaques on the tongue, and numerous pruritic and tender macular rashes with central darkening noted diffusely in the scalp, four extremities, chest, torso, back, and scrotum (Figures 1-3). Thick, yellow crust to his bilateral upper eyelids and conjunctival injections bilaterally were also present.
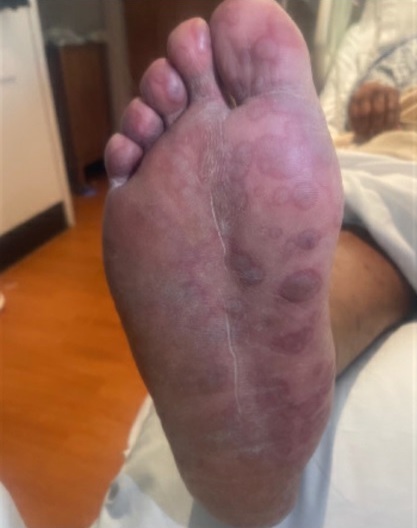
Figure 1. Discrete macular rash with central darkening located on the plantar aspect of the patient's foot.

Figure 2. Diffuse macular rash with central darkening located on palmar aspect of the patient's left hand.
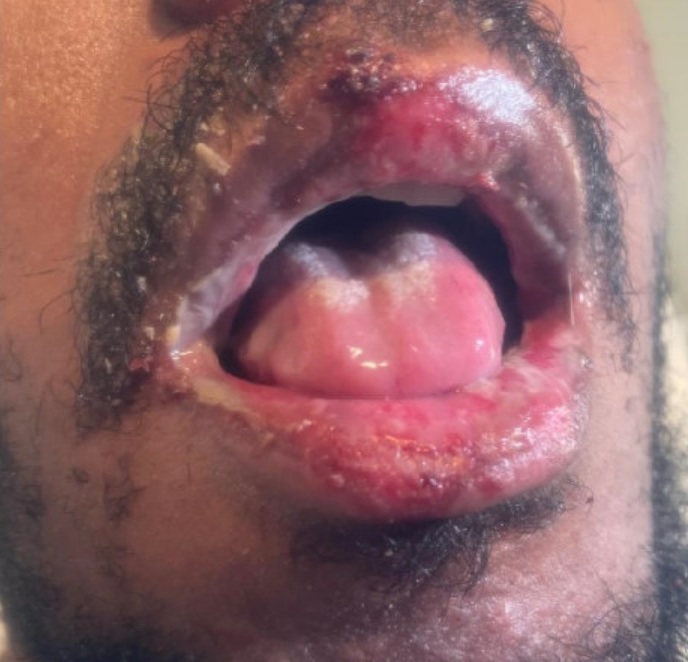
Figure 3. Mucocutaneous lesions noted on the patient's lips.
Laboratory and diagnostic testing. Initial laboratory tests were significant for elevated inflammatory markers of C-reactive protein, 14.4 mg/dL (reference 0-0.9 mg/dL) and erythrocyte sedimentation rate 61 mm/hr (reference 0-15 mm/hr). A nasopharyngeal COVID-19 polymerase chain reaction test was positive, confirming the positive result of the home test performed by the patient 14 days earlier. Of note, his white blood cell count was normal at 6.3 × 103/uL (reference, 4.5-11 × 103/uL) along with a hemoglobin A1C level of 6.8%. A computed tomography of the head was negative for any intracranial pathology.
Treatment and management. Given the initial concerns for a possible infectious etiology causing the disseminated rash, the infectious disease team was consulted, who advised starting empiric intravenous acyclovir 10 mg/kg every 8 hours, intravenous vancomycin 15 mg/kg twice daily, intravenous ceftriaxone 2 g twice daily, and intravenous fluconazole 200 mg daily.
A lumbar puncture was attempted several times but due to an elevated international normalized ratio secondary to newly diagnosed factor VII deficiency, it was unable to be performed. An in-depth infectious panel returned unmarkable findings; it included a respiratory biofire polymerase chain reaction test as well as testing for human immunodeficiency virus, syphilis, HSV 1 and 2, gonorrhea and chlamydia, mycoplasma pneumoniae, enteroviruses, varicella, Epstein-Barr virus, and cytomegalovirus. Furthermore, all cultures collected, including those from the blood, rash, eyelid purulent fluid, and sputum, were negative. The patient's headache and meningeal signs eventually resolved, so lumbar puncture was no longer pursued. His odynophagia and white plaque on his tongue also resolved, so an esophagogastroduodenoscopy was not performed by the gastroenterologist. Given the negative infectious workup, an active infectious etiology was ruled out and so empiric vancomycin, ceftriaxone, acyclovir, and fluconazole were discontinued by the infectious disease team. At this point, ophthalmology and dermatology teams were consulted for a possible skin biopsy to help shed light on this case, but due to patient refusal a biopsy was not performed. After an extensive review of the mucocutaneous lesions that appeared targetoid in nature, the patient's condition was diagnosed as erythema multiforme major secondary to recent COVID-19 infection.
Discussion. Erythema multiforme is an autoimmune reaction resulting in targetoid lesions on the skin. In the major form of this reaction, people with the condition develop ulcers on the mucosal surfaces, such as eyes and lips. Infection causes 90% of cases, with HSV and mycoplasma pneumoniae being the leading causes. Less than 10% of cases stem from adverse drug reactions, such as nonsteroidal anti-inflammatory drugs (NSAIDs), antiepileptics, and antibiotics (eg, sulfonamides).1,2
Skin biopsy demonstrating necrotic keratinocytes, lymphocytic exocytosis, and basal cell vacuolar degeneration can help in the diagnosis of this condition but sometimes cannot be performed, as in our patient.2 Although the HSV test was negative in our patient, it was presumed that he had dormant HSV infection given his prior relationship with an HSV-positive female partner. He did not exhibit any vesicular lesions consistent with HSV reactivation, so this was unlikely to be the etiology for the erythema multiforme. Our patient did have a recent history of using NSAIDs, but they were used to alleviate pain in the days after the onset of the rash so it was unlikely to be the etiology as well.
More case reports are surfacing linking recent COVID-19 infection with dermatologic complications, such as erythema multiforme major. The pathophysiology linking the two conditions is still not fully understood but is postulated to be a lymphocyte-mediated hypersensitivity reaction to COVID-19 inducing cell necrosis and lymphoid infiltration.4 The average period between onset of COVID-19 symptoms and appearance of mucocutaneous lesions has been reported to be 16 to 24 days,3 which is consistent with our case. Current literature suggests that erythema multiforme major associated with COVID-19 can be divided into three categories2,4: virus-related juvenile type, virus-related older type, and drug-induced type. The virus-related juvenile type commonly affects people younger than 30 years of age, with the rash involving the extremities initially. The virus-related older type affects people aged 55 years of age or older, with the rash originating in the trunks initially. The drug-induced type has been shown to be associated with hydroxychloroquine use, with the onset of rash occurring 3 to 10 days after initiation of the drug.4
Patient outcome. Our patient was started on prednisone 40 mg daily with a 10 mg taper every 7 days along with HSV suppression with valaciclovir 500 mg twice daily, both to be taken orally. The decision to include valaciclovir for HSV suppression was made due to his history of unprotected sexual intercourse with an HSV-positive female partner. With this regimen, his rash and pain completely resolved. After 9 days in the hospital, he was discharged home with recommended outpatient follow-up with his primary care physician, dermatologist, and ophthalmologist. Unfortunately, he was lost to follow-up, so his health care providers were unable to monitor for any recurrence of the targetoid rash while on the steroid taper.
Conclusion. As cases of COVID-19 increase worldwide, the possible long-term effects and complications of the virus remains of high interest. This case adds to the growing body of evidence that erythema multiforme major may be a dermatologic complication of COVID-19. More research is needed to elucidate whether this is an effect of the virus itself or from the exaggerated immune response directed at the skin and mucosal surfaces.
1. Shah SN, Chuahan GR, Manjunatha BS, Dagrus K. Drug-induced erythema multiforme: two case series with review of literature. J Clin Diagn Res. 2014;8(9):ZH01-ZH04. https://doi.org/10.7860/JCDR/2014/10173.4761
2. Tahir D, Souliman M; De La Rosa AM, Al-Jobory O, Naguib T. Erythema multiforme-like presentation in an asymptomatic COVID-19 patient. Cureus. 2021;13(12):e201814. https://doi.org/10.7759/cureus.20814
3. Jimenez-Cauhe J, Ortega-Quijano D, Carretero-Barrio I, et al. Erythema multiforme-like eruptions in patients with COVID-19 infection: clinical and histological findings. Clin Exp Dermatol. 2020;45(7):892-895. https://doi.org/10.1111/ced.14281
4. Bennardo L, Nisticò S, Dastoli S, et al. Erythema multiforme and COVID-19: What do we know? Medicina (Kaunas). 2021;57(8):828. https://doi.org/10.3390/medicina57080828


