Peer Reviewed
Darier Disease Complicated by Herpes Simplex Virus Superinfection
AFFILIATIONS:
Department of Family and Community Medicine, School of Medicine, Texas Tech University Health Science Center, Lubbock, TX
CITATION:
Goldman J, Chapman C, Ray N, Neilson Q, Jones C, Babb FC. Darier disease complicated by herpes simplex virus superinfection. Consultant. 2023;63(8):e6. doi:10.25270/con.2023.08.000003.
Received December 16, 2022. Accepted April 3, 2023. Published August 9, 2023.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Jodi Goldman, BS, Department of Family and Community Medicine, School of Medicine, Texas Tech University Health Science Center, 3601 4th Street, Lubbock, TX 79430 (jodi.goldman@ttuhsc.edu)
A 57-year-old man was admitted to the hospital directly from a dermatology clinic due to suspected herpes simplex virus (HSV)-1 and HSV-2 superinfection.
History. The patient’s medical history was significant for Darier disease (DD) which was diagnosed in 2013 via biopsy, a Staphylococcus aureus and Streptococcus dysgalactiae/canis superinfection in 2014, an HSV-1 and HSV-2 superinfection (not on HSV prophylaxis at the time) in July of 2017, and a fungal superinfection in October of 2017. Prior medications used to control his cutaneous symptoms included doxycycline, isotretinoin, salicylic acid ointment, and antiseptic skin washes, but all were without improvement.
The patient was started on acyclovir 400 mg twice daily for HSV prophylaxis in April 2018 after a small flare of viral disease on his left lower back, which improved. In April of 2019, he was started on 25mg daily of acitretin to control his dermatologic symptoms. His daily doses varied from 10 to 25 mg per day depending on what he could afford, until March 2022 when he stopped taking it due to cost.
Three months before admission, a positive Tzanck smear demonstrated HSV for which he was prescribed a 10-day course of valacyclovir (1 g three times daily). One month before admission, he was started on apremilast to replace acitretin. However, the patient felt that this medication worsened his dermatologic disease. He also stopped taking acyclovir one month prior to admission.
On the day of admission, he had been seen in a dermatology clinic where it was noted that his HSV lesions had acutely worsened to a total body surface area greater than 50% while on valacyclovir. He was referred to the emergency department as the dermatologist felt that intravenous antivirals were required for treatment.
On admission, he presented with hundreds to thousands of red-brown hyperkeratotic papules, large areas of erythema, white-yellow plaques and weeping eczema, and malodorous bullae which were most prominent on his bilateral forearms, groin, and back (Figures 1 and 2). He described intermittent chills but remained afebrile. He had difficulty ambulating because of the pain of his lesions.
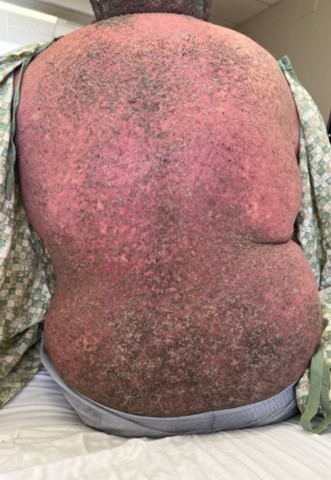
Figure 1. Red-brown hyperkeratotic papules and malodorous bullae of DD on the patient’s back, representative of HSV-1 and HSV-2 superinfection.

Figure 2. White-yellow plaques and malodorous bullae of DD on the patient’s bilateral hands, representative of HSV-1 and HSV-2 superinfection.
Diagnostic testing. Initial laboratory values demonstrated predominantly neutrophilic leukocytosis and elevated HSV-1 and HSV-2 immunoglobin G. With the recent positive Tzanck smear and worsening of disease on valacyclovir, the patient was diagnosed with an HSV-1 and HSV-2 superinfection.
Differential diagnosis. Resistant-HSV superinfection, bacterial superinfection, and fungal superinfection were considered in the differential diagnosis due to the increased risk of cutaneous infections of all etiologies in patients with DD. Although gross examination of the patient's lesions in the context of his medical history made HSV-1 and HSV-2 superinfection the most likely diagnosis, it is essential to investigate all potential etiologies to ensure adequate treatment. These differential diagnoses are ruled out based on bacterial and fungal cultures of the lesions as well as treatment response to empiric therapy.
Treatment and management. The patient was started on intravenous acyclovir 950 mg every 8 hours. On hospital day 4, he was transitioned to oral acyclovir 400 mg every 8 hours due to crusting lesions, improvements in reported pain, and negative blood cultures. With continued improvement of cutaneous symptoms, he was discharged on hospital day 7 to complete a 21-day course of oral acyclovir and to resume acitretin.
Outcome and follow-up. Eight days later, he returned to the emergency department with worsening symptoms, including cutaneous findings similar to the prior admission, fever, chills, fatigue, blurry vision, dysuria, and increased urinary urgency. Upon readmission, laboratory tests demonstrated predominantly neutrophilic leukocytosis and unremarkable urinalysis. The patient was also found to have potential HSV keratitis, as noted by the ophthalmology specialist. The patient reported compliance with acyclovir following initial discharge but noncompliance with acitretin. Given the positive Tzanck smear prior to his previous admission, which served as a rapid and confirmatory test for HSV superinfection, and the recurrence of clinically identical symptoms to his prior admission, the patient’s condition was presumed to be a superinfection possibly due to lack of adherence to the home medication regimen. He was again started on intravenous acyclovir 750 mg every 8 hours.
From hospital day 2 onward, the patient remained afebrile. By hospital day 3, the patient’s lesions began crusting and his pain was improving, but new lesions continued to appear. The quantity of lesions remained stable by hospital day 8. The patient stayed in the hospital to complete a 14-day course of intravenous acyclovir, with dramatic improvement in skin lesions and pain as well as negative blood cultures.
He was discharged with oral valacyclovir 1 g once daily. For his ocular symptoms, he was discharged with ophthalmic erythromycin 0.5% four times daily and ocular lubricant six times daily. It was suggested to start eyelid scrubs. Upon hospital follow-up, the patient reported no new complaints. His valacyclovir prophylaxis was increased to three times daily, and he was started on a vitamin A supplement 2400 mcg daily to help control the cutaneous symptoms of DD.
Discussion. DD, also known as keratosis follicularis, is a rare autosomal dominant disorder of keratinization. First described in 1889, DD affects 1 in every 30,000 to 100,000 individuals worldwide.1 There are no observed differences in sex.2
DD is caused by a pathogenic variant in the ATP2A2 gene, which encodes the SERCA2 pump. Although the exact mechanism by which disruption of this enzyme, which under normal circumstances regulates intracellular calcium homeostasis, leads to the clinical features of DD is unknown, it has been shown experimentally that dysfunction of SERCA2 disrupts cell-cell adhesion.1
The interruption of cell-cell adhesion is seen histologically as dyskeratosis,1 which manifests clinically during adolescence or early adulthood as reddish-brown skin lesions that are predominantly found in the seborrheic and intertriginous areas of the body. The individual skin lesions vary in size and can present as papules to small plaques, but in some areas of the body, especially areas subject to friction, the lesions can coalesce into larger lesions or become macerated.2
Triggers for the development or worsening of the cutaneous findings include ultraviolet light, heat, humidity, sweating, and friction.1 These skin findings are often accompanied by malodorous discharge, which likely contributes greatly to the psychosocial morbidity of the disease.1
Because of the loss of cell-cell adhesion seen in DD, patients are at increased risk for several infectious complications, including bacterial (S aureus), fungal (Candida sp.), and viral superinfections.1,3,4 These superinfections contribute greatly to the mortality and morbidity of DD. A rare but serious complication is HSV-1 or HSV-2 superinfection, also known as eczema herpeticum (EH) or Kaposi varicelliform eruption. EH is a cutaneous superinfection caused by HSV that most commonly occurs in the context of atopic dermatitis. However, any dermatologic condition that impairs the skin barrier can secondarily be infected with HSV, potentially leading to EH. Historically, HSV-1 is the primary causative agent of EH in the setting of DD, with only a few other reported cases of HSV-2-induced EH.5,6
There are currently no standardized guidelines for the treatment of DD, including its superimposed complications such as EH, as most of the limited evidence for the different treatment options come from case reports and clinical trials with small sample sizes.6 This lack of evidence-based protocol could contribute greatly to the morbidity and mortality of the condition, especially in the case of bacterial, fungal, or viral superinfections that may require longer courses of systemic treatments than superinfections that occur among patients without DD.
With specific regard to EH in DD, an extensive review of the literature demonstrates that there is no consensus on the duration or route of administration of antiviral treatment.1-6 However, all other reported cases of HSV-1 and HSV-2 superinfection in DD showed complete resolution of symptoms following either oral or intravenous inpatient treatment with acyclovir and, in some cases, outpatient maintenance therapy with oral acyclovir.4-5
To our knowledge, this is the first report to describe a case of HSV-1 and HSV-2 superinfection secondary to DD in a patient who initially showed significant improvement with inpatient therapy but ultimately needed hospital readmission despite appropriate management. This case illustrates how the lack of consensus in the current literature for the treatment of EH in the setting of DD could contribute greatly to the morbidity and mortality of the disease.1-6 Further research and a consensus on optimal treatment strategies is still needed. Improved understanding and evidence-based protocols are crucial to effectively manage superinfections in patients with DD.
1. Rogner DF, Lammer J, Zink A, Hamm H. Darier and Hailey‐Hailey disease: update 2021. J Dtsch Dermatol Ges. 2021;19(10):1478-1501. doi:10.1111/ddg.14619
2. Takagi A, Kamijo M, Ikeda S. Darier disease. J Dermatol. 2016;43(3):275-279. doi:10.1111/1346-8138.13230
3. Digby S, Hald M, Arendrup M, Hjort S, Kofoed K. Darier disease complicated by terbinafine-resistant trichophyton rubrum: A case report. Acta Derm Venereol. 2017;97(1):139-140. doi:10.2340/00015555-2455
4. Geroldinger-Simic´ M, Jabkowski J, Klein G. Generalized herpes simplex virus infection in Darier's disease. J Dtsch Dermatol Ges. 2016;14(8):841-842. doi:10.1111/ddg.12899
5. Walker K, Martini A, Philips H, Sharp L, Thomas K, Tarbox M. Darier disease with disseminated herpes simplex virus type 2 infection. Dermatol Online J. 2019;25(4):5. doi:10.5070/d3254043574
6. Hanna N, Lam M, Fleming P, Lynde CW. Therapeutic options for the treatment of Darier’s disease: A comprehensive review of the literature. J Cutan Med Surg. 2022;26(3):280-290. doi:10.1177/12034754211058405


