Peer Reviewed
Acute Pancreatitis in Sickle Cell Vaso-occlusive Crisis
Authors:
We’am Hussain, MD
Internal Medicine Resident, Wright State University, Dayton, Ohio
Nneoma Onuorah, MBBS
Internal Medicine Resident, Wright State University, Dayton, Ohio
Immaculate Foy, MD
Internal Medicine Resident, Wright State University, Dayton, Ohio
Marc Raslich, MD
Associate Professor of Internal Medicine and Pediatrics, Wright State University, Dayton, Ohio
Citation:
Hussain W, Onuorah N, Foy I, Raslich M. Acute pancreatitis in sickle cell vaso-occlusive crisis. Consultant. 2019;59(4):127-128.
A 19-year-old woman presented with nausea, vomiting, epigastric abdominal pain with radiation to her back, and bilateral hip, knee, and ankle joint pain for the past 5 hours. Her medical history was significant for homozygous sickle cell disease (SCD), hemoglobin SS, cholecystectomy at age 11 due to cholelithiasis, and multiple hospitalizations for abdominal pain due to diffuse abdominal pain due to vaso-occlusive crisis (VOC). She was taking folic acid and had no history of alcohol use or hypercholesterolemia.
The patient stated that the abdominal pain she was experiencing now was similar to her previous episodes of abdominal pain due to VOC, and that the pain was sharp in quality and had persisted for hours; she did note, however, that the pain was specifically in the epigastric region this time, with radiation to her back, unlike during previous episodes.
Vital signs were within normal limits, and physical examination findings were remarkable for epigastric tenderness radiating to her back. Complete blood count (CBC) results showed the white blood cell count, hemoglobin level, hematocrit level, and platelet count to be within normal limits. Liver function and renal function test results were also normal. The lipase level was found to be elevated at 1468 U/L (reference range, 0-60 U/L).
Given the patient’s history of repeated crisis possibly leading to organ dysfunction, computed tomography (CT) scans of the abdomen were ordered, which showed new fat stranding surrounding the pancreatic tail, representing peripancreatic inflammation (Figure).
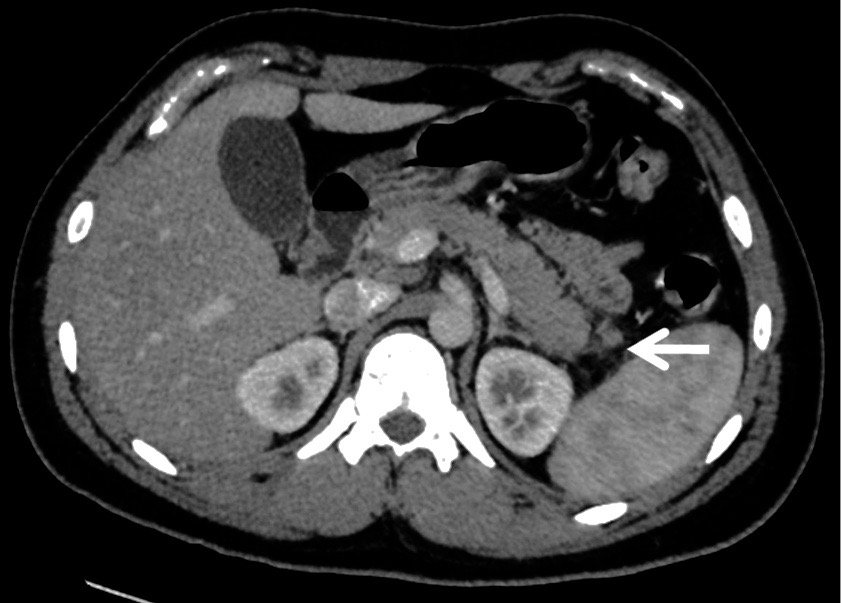
Figure. Abdominal CT scan showing fat stranding surrounding the pancreatic tail.
The symptoms resolved with intravenous fluid hydration and pain medications, and the patient’s diet was advanced from a clear fluid diet to a regular diet by day 5 of admission. She was discharged on day 6. Repeated CT scans and lipase tests were not indicated during admission, since the patient responded appropriately with treatment and no clinical evidence of acute or chronic hemolysis was present. No consultation services were required during the patient’s admission.
The patient failed to follow up at a primary care clinic visit that had been scheduled 1 week after discharge.
DISCUSSION
SCD is a genetic autosomal recessive disorder resulting from a mutation of hemoglobin S and characterized by VOC and hemolytic anemia. Vaso-occlusion is caused by sickle-shaped red blood cells (RBCs) that obstruct microvessels, ultimately resulting in ischemia, pain, and necrosis.1 SCD affects approximately 100,000 Americans, occurring in approximately 1 in 365 black or African American births and 1 in 16,300 Hispanic births.2 Approximately 1 in 13 black or African American children are born with sickle cell trait (SCT).2
Pain is the most commonly reported symptom of SCD and often is disabling.3 Adult respondents in the Pain in Sickle Cell Epidemiology Study (PiSCES) reported SCD pain on 54.5% of the 31,017 days surveyed. In fact, 29.3% of respondents had pain on greater than 95% of the days surveyed.4,5
Acute abdominal pain is a common presentation of sickle cell VOC, which could involve any of several abdominal organs and results from capillary engorgement, sickling of RBCs, hypercoagulability, and stasis in the vasa vasorum of larger vessels.6 Approximately 10% of patients with SCD hospitalized for VOC present with acute abdominal pain.7 Common etiologies include acute splenic sequestration, splenic infarction, ischemic bowel, biliary colic, acute cholecystitis, opioid-induced constipation, renal papillary necrosis, hepatic crisis, urinary tract infection, hepatitis, and liver abscesses.8
However, acute ischemic pancreatitis is a very rare complication of VOC.9,10 In a small prospective study in 2011, Akingbola and colleagues showed that 70% of cases of acute abdomen in VOC involved the epigastric region, with presentation similar to that of acute pancreatitis.11 When pancreatitis occurs, it may be a result of biliary obstruction or microvessel occlusion causing ischemia.10
Despite acute abdominal pain being a relatively common occurrence in patients with VOC, life-threatening complications can occur if the diagnosis of microvessel occlusion-induced pancreatitis is missed. Moreover, pancreatitis due to vaso-occlusion is clinically indistinguishable from other etiologies of acute abdominal pain from sickle cell VOC, and the initial diagnosis is based on clinical suspicion. Our patient met the criteria for acute pancreatitis with a serum lipase level elevated at greater than 3 times the upper limit, in association with the presence of epigastric pain radiating to the back and abdominal CT scans showing fat stranding surrounding the pancreatic tail.
Treatment is similar to any other presentation of acute pancreatitis, which is initially conservative with intravenous fluids, pain control, and electrolyte repletion. The clinical significance of early diagnosis of VOC-induced acute pancreatitis relies on the indication for exchange blood transfusion (EBT) in worsening cases of acute pancreatitis characterized by multiorgan failure.10,12,13 Although it was not required in this patient’s case, EBT helps dilution of hemoglobin S and reduction in sickling, with a hemoglobin S goal of less than 20% to 30%.14
EBT offers better control of blood volume and viscosity while decreasing the risk of transfusion-related hemochromatosis. EBT is most readily accomplished through automated red-cell exchange (erythrocytapheresis).15 However, the drawbacks of EBT include relatively high cost, limited availability, and often a requirement for large central venous catheters.15
Routine blood transfusion may also be used for the care for SCD, although less-preferred to EBT due to potential complications of iron overload, transfusion reactions, infections, acute lung injury, pain crisis, stroke, immunomodulation, anaphylaxis, and alloimmunization.16,17
- Bunn HF. Pathogenesis and treatment of sickle cell disease. N Engl J Med. 1997;337(11):762-769.
- Data & statistics on sickle cell disease. Centers for Disease Control and Prevention. https://www.cdc.gov/ncbddd/sicklecell/data.html. Reviewed August 9, 2017. Accessed February 19, 2019.
- Hassell KL. Population estimates of sickle cell disease in the U.S. Am J Prev Med. 2010;38(4 suppl):S512-S521.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease—rates and risk factors. N Engl J Med. 1991;325(1):11-16.
- Smith WR, Penberthy LT, Bovbjerg VE, et al. Daily assessment of pain in adults with sickle cell disease. Ann Intern Med. 2008;148(2):94-101.
- Dhiman RK, Yusif RA, Nabar UJ, Albaqali A. Gastrointestinal: ischemic enteritis and sickle cell disease. J Gastroenterol Hepatol. 2004;19(11):1318.
- Ebert EC, Nagar M, Hagspiel KD. Gastrointestinal and hepatic complications of sickle cell disease. Clin Gastroenterol Hepatol. 2010;8(6):483-489.
- Meshikhes A-WN. Gastroenterological manifestations of sickle cell disease. Saudi J Gastroenterol. 1997;3(1):29-33.
- Hasan B, Asif T, Braun C, Bahaj W, Dosokey E, Pauly RR. Pancreatitis in the setting of vaso-occlusive sickle cell crisis: a rare encounter. Cureus. 2017;9(4):e1193.
- Ahmed S, Siddiqui AK, Siddiqui RK, Kimpo M, Russo L, Mattana J. Acute pancreatitis during sickle cell vaso-occlusive painful crisis. Am J Hematol. 2003;73(3):190-193.
- Akingbola TS, Kolude B, Aneni EC, et al. Abdominal pain in adult sickle cell disease patients: a Nigerian experience. Ann Ib Postgrad Med. 2011;9(2):100-104.
- Kumar A, Posner G, Marsh F, Bellvue R, Dosik H. Acute pancreatitis in sickle cell crisis. J Nat Med Assoc. 1989;81(1):91-92.
- US Department of Health and Human Services, National Institutes of Health, National Heart, Lung, and Blood Institute. Evidence-Based Management of Sickle Cell Disease: Expert Panel Report, 2014. https://www.nhlbi.nih.gov/sites/default/files/media/docs/sickle-cell-disease-report%20020816_0.pdf. Accessed February 19, 2019.
- Lombardo T, Rosso R, La Ferla A, et al. Acute chest syndrome: the role of erythro-exchange in patients with sickle cell disease in Sicily. Transfus Apher Sci. 2003;29(1):39-44.
- Danielson CFM. The role of red blood cell exchange transfusion in the treatment and prevention of complications of sickle cell disease. Ther Apher. 2002;6(1):24-31.
- Wanko SO, Telen MJ. Transfusion management in sickle cell disease. Hematol Oncol Clin North Am. 2005;19(5):803-826.
- Vichinsky EP, Luban NLC, Wright E, et al; Stroke Prevention Trail in Sickle Cell Anemia. Prospective RBC phenotype matching in a stroke-prevention trial in sickle cell anemia: a multicenter transfusion trial. Transfusion. 2001;41(9):1086-1092.


