What’s the Explanation for This Girl’s Intractable Rash and Brittle Hair?
A 3-year-old girl presented with an 18-month history of a widespread, pruritic rash that had not responded to numerous topical therapies, including triamcinolone, ketoconazole, nystatin, mupirocin, and tacrolimus. She also had been treated for her numerous soft tissue infections with oral prednisone and several courses of oral antibiotics, including clindamycin, cephalexin, and amoxicillin. The girl’s grandmother reported that there had been occasional improvement with oral antibiotic therapy, but that the rash would never completely resolve, and that exacerbations were frequent.
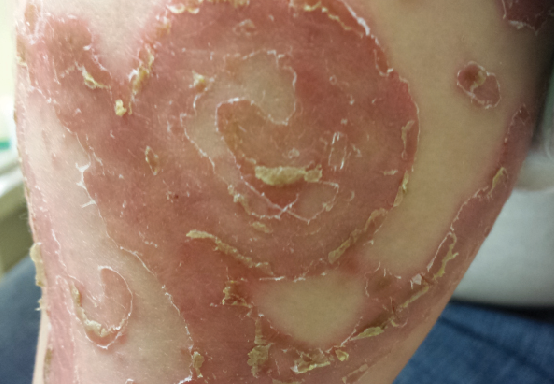
The girl’s past medical history included failure to thrive, patent ductus arteriosus, and an atrial septal defect. The girl had been conceived in rape, and there is an exceedingly high possibility of consanguinity, which may explain the cause of her syndromic features and cardiac abnormalities.
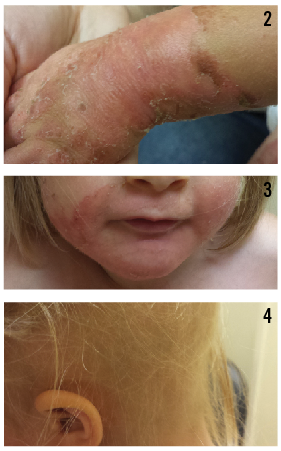 Review of systems revealed a history of diaper rash, infrequent episodes of diarrhea, difficulty swallowing solid food, numerous cutaneous infections, and physical and mental delay.
Review of systems revealed a history of diaper rash, infrequent episodes of diarrhea, difficulty swallowing solid food, numerous cutaneous infections, and physical and mental delay.
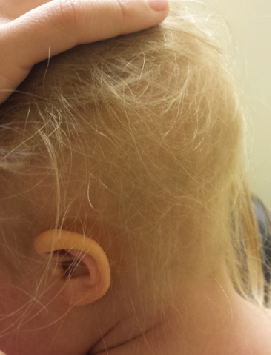
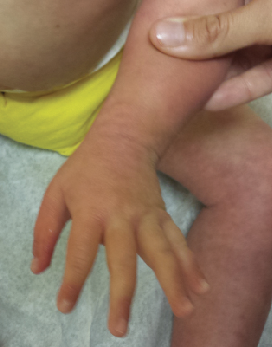
On physical examination, numerous erythematous, annular to serpiginous plaques with peripheral scale, some with a double-edged scale, were noted bilaterally on the girl’s neck, trunk, and upper and lower extremities (Figure 1). Erythematous acral (Figure 2) and perioral (Figure 3) patches with desquamation also were present, along with dry, brittle, sparse hair (Figure 4). No teeth or nail abnormalities were observed, but syndromic features including hypertelorism and epicanthal folds were present. Musculoskeletal examination revealed an inability to stand without support, a resting tremor, and that she was below the fifth percentile for height and weight.
What’s causing these symptoms?
A. Langerhans cell histiocytosis
B. Acrodermatitis enteropathica
C. Leiner disease (erythroderma desquamativum)
D. Glucagonoma
Answer: B, acrodermatitis enteropathica
A 4-mm punch biopsy specimen obtained from a plaque on the girl’s abdomen showed a psoriasiform dermatitis with parakeratosis and a mild perivascular infiltrate of lymphocytes and histiocytes in the dermis, findings consistent with seborrheic dermatitis or psoriasis.
A second 4-mm punch biopsy specimen taken from a plaque on the left lower extremity revealed a mixed spongiotic and focal interface dermatitis, findings consistent with a hypersensitivity reaction.

Figure 1. Erythematous, serpiginous plaque with peripheral scale on the right lateral thigh.
A trichogram of hair obtained from the scalp and lateral eyebrows was negative for hair shaft abnormalities. Genetic test results were negative for a SPINK5 gene mutation, associated with Netherton syndrome. Findings of a complete blood count were within normal limits. The girl’s serum zinc concentration was less than 25 µg/dL (reference range, 60-130 µg/dL), and her serum alkaline phosphatase (ALP) level was 44 U/L (reference range, 93-309 U/L). Genetic testing was ordered to confirm SLC39A4 zinc transporter defect, but Medicaid denied coverage, and the test was not performed.
The differential diagnosis included Netherton syndrome, nutritional deficiency, infantile seborrheic dermatitis, infantile psoriasis, Langerhans cell histiocytosis, glucagonoma, and Leiner disease (erythroderma desquamativum). Based on clinical presentation, however, the girl was given a preliminary diagnosis of acrodermatitis enteropathica (AE), a rare, autosomal recessive, inherited form of zinc deficiency. She was started on 30 mg/d of oral elemental zinc, after which the rash rapidly resolved (Figure 5); she began to gain weight, and her motor skills and mentation improved. Her symptoms’ responsiveness to zinc supplementation further supported the diagnosis of AE.
Two months after initiation of therapy, her serum zinc concentration had increased to 67 µg/dL, and her ALP level had risen to 90 U/L. Her resting tremor also had resolved, and she was now able to stand without support. As of 8 months after beginning zinc replacement, the girl’s zinc and ALP levels had been maintained in the normal range, and she had achieved significant weight gain with the ability to swallow solid foods.
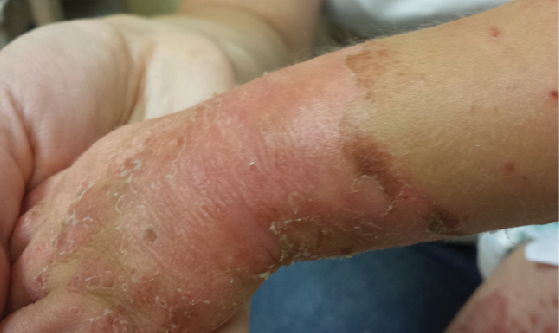
Figure 2. Erythematous patch with desquamation and scale on the right dorsal wrist.
Discussion
Zinc is an essential trace element that plays a vital role in cell proliferation, healing, and tissue repair. Zinc stabilizes cell membranes by reducing free radical production and preventing lipid peroxidation. Zinc also is important in metabolism of proteins, carbohydrates, and vitamins and in growth and development. Proper function of the immune system relies on zinc, and the element has a major role in fertility and conception.1 Zinc deficiency has acquired and inherited forms, both of which present with similar clinical features that vary in severity based on the degree of zinc depletion.
Acrodermatitis enteropathica (AE) is a rare, autosomal recessive, inherited form of zinc deficiency. It is caused by a mutation of the SLC39A4 gene, which encodes a zinc/iron-regulated transporter-like protein (ZIP4) located on chromosome arm 8q24.3.2 This gene is expressed predominately in the duodenum and jejunum, which are the principal sites of zinc absorption.
The incidence of AE is estimated at 1 per 500,000 children per year, with no predilection for race or sex.2
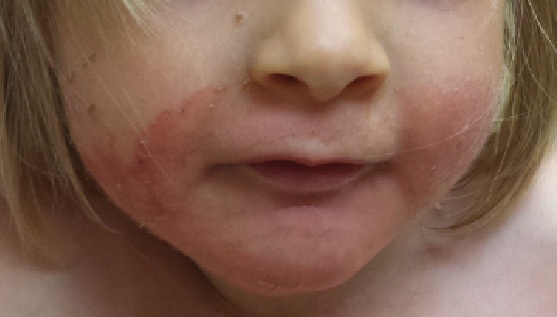
Figure 3. Erythematous perioral patch with desquamation and peripheral scale.
AE presents during infancy, with the classic clinical triad of acral dermatitis, alopecia, and diarrhea occurring in approximately 20% of patients.1 The cutaneous manifestations of AE typically occur within 1 to 2 weeks after a child has been weaned from breast milk to formula or cereal, since the latter have lower zinc bioavailability than does breast milk.
AE also can present in exclusively breastfed infants, most often those born prematurely.1 Preterm infants or debilitated children who are on prolonged therapy with a parenteral nutrition formulation that is deficient in zinc also are at risk.3,4
The initial expression of the disease includes erosive perioral dermatitis with angular cheilitis, with progression to involve the face, acral surfaces, and diaper region. Palmar erythema, sometimes with annular scale, also may be present.
Diarrhea is the predominant extracutaneous feature of AE; it also is the most variable, and it can be intermittent or totally absent. Decreased zinc absorption resulting from diarrhea may further aggravate the condition, leading to a vicious cycle.5 Other extracutaneous features include growth retardation, anemia, hypogonadism, delayed puberty, and neuropsychological disturbances such as altered mental status and anorexia.5
Persons with AE frequently have compromised immune function that leads to persistent cutaneous infections, including conjunctivitis, blepharitis, candidiasis, and paronychia. These features may be evident even in the absence of the classic cutaneous manifestations.5 Photosensitivity also is a frequent association of AE.
The laboratory test finding of a low plasma zinc level is diagnostic for AE, with many patients having a plasma concentration of less than 50 µg/dL.6 Serum ALP is a zinc-dependent enzyme that may serve as surrogate diagnostic marker of zinc deficiency.
For cases in which results of laboratory zinc testing results are ambiguous, histopathology testing may be of diagnostic help, but characteristic histopathologic findings are not diagnostic for AE and are shared by other nutritional deficiencies.
Histopathology test findings in AE include necrolysis with subsequent confluent necrosis of keratinocytes in the upper spinous layer. The affected keratinocytes often have pyknotic nuclei. Confluent parakeratosis with associated hypogranulosis, and dermal edema may be present, along with subcorneal vesicle formation in the pale areas. Associated neutrophilic crust is a variable finding. Scattered dyskeratotic keratinocytes often are present within all levels of the epidermis.
Late lesions can mimic psoriasis,7 as was the case with our patient, whose initial biopsy results were consistent with a diagnosis of psoriasis.
Figure 4. Diffuse alopecia with dry, brittle, sparse hair. No teeth or nail abnormalities were observed.
Treatment of the hereditary form of AE is lifelong elemental zinc supplementation, initiated at 3 mg/kg/d.2 The dosage then is adjusted, since requirements vary from patient to patient. The duration of therapy for acquired forms of AE varies with etiology and degree of zinc depletion.2 Rapid resolution of diarrhea typically occurs within 24 hours, and marked improvement of skin lesions generally is noted just days after initiation of zinc replacement.
Zinc deficiency is a rare condition, and other diagnoses should be considered if a patient fails to respond to zinc replacement within a few days and his or her ALP level remains stable. Serum zinc and ALP levels should be monitored every 3 to 6 months and the zinc dosage adjusted accordingly.8,9
Care should be taken to avoid excessive supplementation of zinc, which can deplete copper levels in the blood and, in extreme cases, cause multisystem organ failure. Adverse effects of zinc replacement can include but are not limited to nausea, vomiting, and gastric hemorrhage.6
The differential diagnosis of AE should include diaper dermatitis, candidiasis, infantile seborrheic dermatitis, infantile psoriasis, cystic fibrosis, Langerhans cell histiocytosis, glucagonoma, other metabolic acidopathies, and Leiner disease.1
 The Take-Home Message
The Take-Home Message
AE is a rare disease, but it is critically important to recognize the cutaneous manifestations of zinc deficiency, the long-term sequelae of which are unnecessary and largely preventable. Because results of measurements of serum zinc levels can be misleading and not consistent with actual levels of the element in patients with AE, is it important to base the diagnosis and treatment on the patient’s clinical presentation and therapeutic response to zinc supplementation.
Patients whose zinc deficiency recurs after the cessation of zinc supplementation therapy should undergo testing for the SLC39A4 gene mutation.
It is also important to note that recent shortages of injectable zinc used as a component of parenteral nutrition formulations have led to increased reports of AE-like eruptions.10
Figure 5. Near complete resolution of skin eruption after initiation of zinc therapy at 2-week follow-up visit.
Michael Centilli, DO, is the chief dermatology resident at Botsford Hospital in Farmington Hills, Michigan.
Michael Legacy is a third-year medical student at Michigan State University College of Osteopathic Medicine in East Lansing.
Michelle Legacy, DO, is a dermatologist at the Skin and Vein Center in Fenton, Michigan.
Eric Seiger, DO, is a dermatologist and cosmetic surgeon at the Skin and Vein Center in Fenton, Michigan.
Kirk Barber, MD, FRCPC—Series Editor, is a consultant dermatologist at Alberta Children’s Hospital and clinical associate professor of medicine and community health sciences at the University of Calgary in Alberta.
References
1. Canpolat F, Canpolat FE, Eskioglu F. Acrodermatitis enteropathica in a full-term exclusively breast-fed infant. Eur J Dermatol. 2008;18(2):192-193.
2. Park C-H, Lee MJ, Kim H-J, Lee G, Park J-W, Cinn Y-W. Congenital zinc deficiency from mutations of the SLC39A4 gene as the genetic background of acrodermatitis enteropathica. J Korean Med Sci. 2010;25(12):1818-1820.
3. Barbarot S, Chantier E, Kuster A, et al. Symptomatic acquired zinc deficiency in at-risk premature infants: high dose preventive supplementation is necessary. Pediatr Dermatol. 2010;27(4):380-383.
4. Jeejeebhoy K. Zinc: an essential trace element for parenteral nutrition. Gastroenterology. 2009;137(5 suppl):S7-S12.
5. Schmuth M, Fritsch PO. Cutaneous changes in nutritional diseases. In: Krutmann J, Humbert P, eds. Nutrition for Healthy Skin: Strategies for Clinical and Cosmetic Practice. Berlin, Germany: Springer; 2011:3-13.
6. Maverakis E, Fung MA, Lynch PJ, et al. Acrodermatitis enteropathica and an overview of zinc metabolism. J Am Acad Dermatol. 2007;56(1):116-124.
7. Jensen SL, McCuaig C, Zembowicz A, Hurt MA. Bullous lesions in acrodermatitis enteropathica delaying diagnosis of zinc deficiency: a report of two cases and review of the literature. J Cutan Pathol. 2008;35(suppl 1):1-13.
8. Van Wouwe JP. Clinical and laboratory diagnosis of acrodermatitis enteropathica. Eur J Pediatr. 1989;149(1):2-8.
9. Arlette JP. Zinc and the skin. Pediatr Clin North Am. 1983;30(3):583-596.
10. Ruktanonchai D, Lowe M, Norton SA, et al; Centers for Disease Control and Prevention (CDC). Zinc deficiency-associated dermatitis in infants during a nationwide shortage of injectable zinc—Washington, DC, and Houston, Texas, 2012–2013. MMWR Morb Mortal Wkly Rep. 2014;63(2):35-37.


