Peer Reviewed
What Is This Asymptomatic, Slow-Growing Lesion on a 40-Year-Old Man’s Back?
Authors:
Alexander K. C. Leung, MD, and Benjamin Barankin, MD
Citation:
Leung AKC, Barankin B. What is this asymptomatic, slow-growing lesion on a 40-year-old man’s back? Consultant. 2017;57(10):611-613.
A 40-year-old man presented with a slow-growing, asymptomatic lesion on the right upper back, which had been present for approximately 4 years. The patient was otherwise in good health. There was no history of trauma to the affected area. He denied any recent weight loss. There was no personal or family history of malignancy.
Physical examination revealed a firm, erythematous to violaceous plaque on the right upper back. The lesion was not warm or tender. It was attached to the skin but not to the underlying structure. There was a dark, benign-looking melanocytic nevus on the left upper back. There was no axillary lymphadenopathy. The rest of the examination findings were unremarkable.
A punch biopsy of the lesion showed fascicles of spindle cells arranged in a storiform pattern in the dermis with infiltration of the subcutaneous fat in a lacelike pattern. Atypia and mitotic activity were absent. Immunohistochemical staining showed that the spindle cells were positive for CD34 and vimentin and negative for factor XIIIa, S100, and CD68.
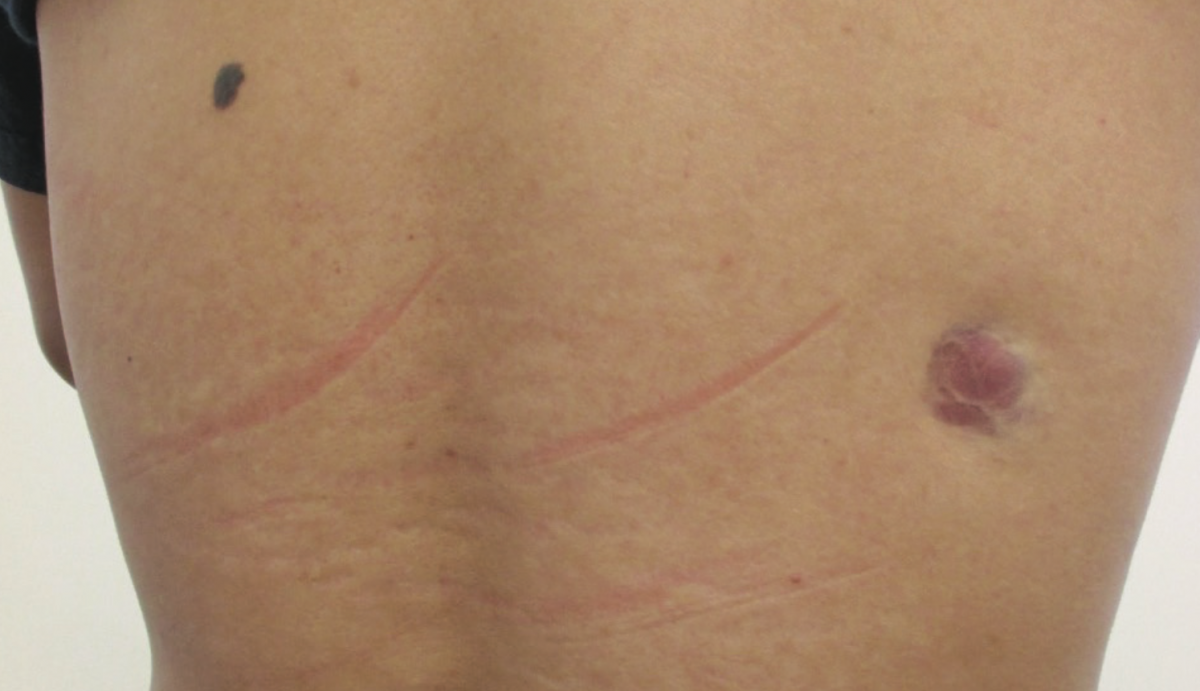
Answer on next page
Answer: Dermatofibrosarcoma protuberans
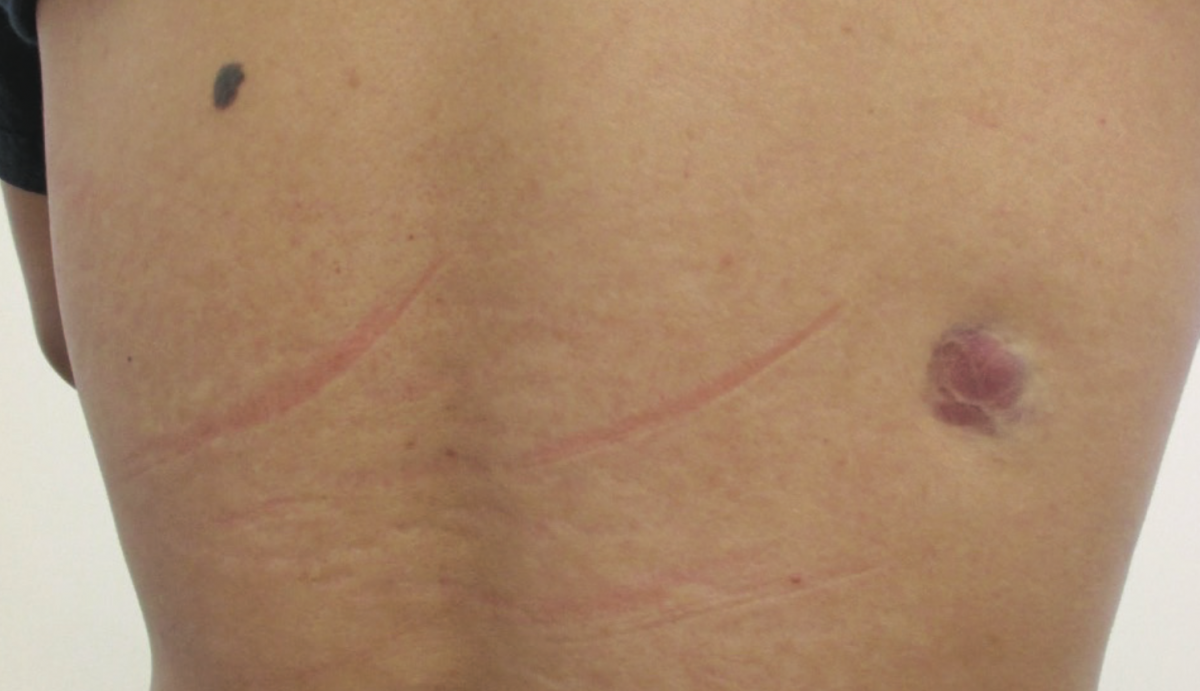
Dermatofibrosarcoma protuberans is the most common dermal sarcoma and accounts for approximately 18% of all cutaneous soft-tissue sarcomas.1-3 The disease is characterized clinically by a slow-growing, insidious, locally aggressive cutaneous tumor with a high rate of local recurrence but a low risk of metastasis, and histologically by infiltrative storiform (cartwheel pattern) fascicles of uniform bland spindle cells with diffuse CD4 expression.3-5
The condition was first described by Taylor in 1890.6 The term dermatofibrosarcoma protuberans was coined by Hoffmann in 1925.7
EPIDEMIOLOGY
The estimated incidence of dermatofibrosarcoma protuberans is between 0.8 and 5 cases per million individuals per year.2,8,9 There is a slight male predominance in the adult population and a slight female predominance in the pediatric age group.2,4,10,11 Typically, patients present between 20 and 50 years of age.2,8,12,13 The condition, however, has been described in all age groups, including a number of congenital cases.4,14 Approximately 6% of cases occur in the pediatric age group.3,13,15 The condition is more common among the black population than among the white population.4,8,10-12 Trauma has been implicated as a possible etiologic factor, but solid evidence is lacking.8,10,15
PATHOGENESIS
More than 90% of cases exhibit reciprocal chromosomal translocation t(17;22)(q22;q13) and supernumerary ring chromosomes composed of interspersed sequences from 17q22 and 22q12.3,10,16 This translocation results in the fusion of the collagen type I α 1 chain gene (COL1A1) on chromosome 17 with the platelet-derived growth factor B gene (PDGFB) on chromosome 22.4 The fusion protein COL1A1-PDGFB activates PDGF receptor, leading to its autocrine activation and subsequent tumor formation.3,4,16 In approximately 8% of cases, genes located on chromosomes 2, 5, 7, 8, 11, 12, 13, 14, 16, 18, and 21 have been shown to be responsible for the tumor formation.1,15
HISTOPATHOLOGY
Histopathologic examination of the lesion shows prominent storiform fascicles of spindle cells with elongated nuclei, with even chromatin in the dermis often infiltrating the subcutaneous fat in the form of tentacle-like projections through the septa and fat lobules, resulting in a lacelike or honeycomb appearance.1-5 There is often thinning of the overlying epidermis and a narrow tumor-free zone (grenz zone). Atypia and mitotic activity are usually minimal or absent.3,15,17
The spindle cells stain positively for CD34, hyaluronate, and vimentin and negatively for factor XIIIa, S100, desmin, CD68, and smooth muscle actin.10,12,15
CLINICAL MANIFESTATIONS
Clinically, dermatofibrosarcoma protuberans starts as an asymptomatic papule or nonindurated patch. The tumor grows very slowly and may go unrecognized for years. Typically, by the time it presents, dermatofibrosarcoma protuberans is an asymptomatic indurated plaque or firm nodule.3 The color varies from erythematous, pink, violaceous, brownish, bluish, or gray, to flesh-colored.1,2,5,10 Telangiectasia may be present on the surface or at the periphery of the tumor.18 The tumor is usually adherent to the overlying skin and not to the underlying structure.4,10,14 Fixation to the underlying structure may occur late in the course of the disease.5,18
The lesion usually measures from 1 to 5 cm at the time of the diagnosis.13,15 If neglected, the tumor may enlarge and protrude through the skin, hence the name protuberans. Sites of predilection include the trunk (40% to 50%), proximal extremities (30% to 40%), and head and neck (10% to 15%).3 Atypical sites include the breast, vulva, pubic area, and scalp.17 The most common sites in children are the back and the extremities.10,15
DIAGNOSIS
The diagnosis is based on the history (asymptomatic, very slow growing cutaneous tumor) and physical examination findings (indurated plaque or firm nodule attached to the skin, typical distribution). Dermoscopy may aid in the diagnosis. Dermoscopic features include delicate pigmented network, unfocused linear-irregular vessels, structureless light-brown areas, structureless hypopigmented or depigmented areas, shiny white streaks, and pink background coloration.18,19 A punch or incisional biopsy for histopathologic examination and direct immunohistochemical staining is generally recommended to confirm the diagnosis.15
DIFFERENTIAL DIAGNOSIS
The differential diagnosis includes hypertrophic scar, keloid, epidermal inclusion cyst, dermatofibroma, dermatomyofibroma, hemangioma, neurofibroma, schwannoma, lipoma, leiomyoma, desmoid tumor, morphea, melanoma, pilomatrixoma, granuloma annulare, nodular fasciitis, sarcoidosis, basal cell carcinoma, fibrosarcoma, and Kaposi sarcoma.2-4,12,13
COMPLICATIONS
The tumor may undergo fibrosarcomatous or pleomorphic sarcomatous transformation, which is associated with rapid growth of the tumor and a more aggressive course.3,5,16 Metastasis is uncommon, occurring in approximately 5% of cases, and often follows multiple local recurrences.3,9,12,15 The lungs are the most frequent site of distant metastasis.3,15 Metastasis to regional lymph nodes occur in less than 1% of cases.15 Other possible complications include ulceration and bleeding.15
LABORATORY STUDIES
Chest radiography should be considered if pulmonary metastasis is suspected.4 When necessary, magnetic resonance imaging can be used to define the extent of tissue infiltration and depth of involvement if the tumor is large or recurrent.12,15 Focal appendage-like tapered elongation of the tumor at the lesion/skin interface may be noted.20 This radiological “claw” sign is classic.20
Molecular testing such as reverse transcription polymerase chain reaction and fluorescence in situ hybridization for the presence of COL1A1-PDGFB fusion gene are not routinely indicated but should be considered to confirm a difficult diagnosis and prior to the initiation of imatinib therapy.15,16 In general, tumors lacking the t(17,22) translocation do not respond to imatinib therapy.3
MANAGEMENT
Because the tumor has a propensity for local recurrence, Mohs micrographic surgery and wide local excision are the treatment of choice for a localized tumor, since the tumor often exhibits extensive infiltration beyond gross margins.3,9,12,21 Conservative resection with undefined surgical margins has been associated with a recurrence rate ranging from 26% to 60%,13 whereas the recurrence rate after Mohs micrographic surgery is approximately 1%.8 Radiotherapy can be used as primary treatment of an inoperable tumor or as an adjuvant therapy after positive surgical margins are found after resection of the tumor and further surgery is not feasible.9,10 Imatinib, a tyrosine kinase inhibitor, can be used for an unresectable locally advanced, recurrent, or metastatic tumor.4,15,16 The recommended oral dose is 400 to 800 mg/day.4
Because of the propensity for local recurrences, most of which occur within 3 years of surgery, patients should be examined every 6 months during this period, and annually for the rest of their life.10 This patient underwent wide excision of the lesion and has been free of recurrence for 3 years.
PROGNOSIS
The tumor is locally aggressive. Incomplete removal results in a high recurrence rate. The overall prognosis depends on the staging of the disease. With localized disease where the tumor can be excised with Mohs micrographic surgery, the 5-year survival rate is close to 100%, while the 15-year survival rate is approximately 98%.9,22 With metastasis, the mean survival is approximately 2 years.15 In general, older age at presentation, male sex, and large tumor size are associated with an overall poorer survival.23 n
Alexander K. C. Leung, MD, is clinical professor of pediatrics at the University of Calgary and a pediatric consultant at the Alberta Children’s Hospital in Calgary, Alberta, Canada.
Benjamin Barankin, MD, is a dermatologist and the medical director and founder of the Toronto Dermatology Centre in Toronto, Ontario, Canada.
REFERENCES:
- Kornik RI, Muchard LK, Teng JM. Dermatofibrosarcoma protuberans in children: an update on the diagnosis and treatment. Pediatr Dermatol. 2012;29(6):707-713.
- Posso-De Los Rios CJ, Lara-Corrales I, Ho N. Dermatofibrosarcoma protuberans in pediatric patients: a report of 17 cases. J Cutan Med Surg. 2014;18(3):180-185.
- Thway K, Noujaim J, Jones RL, Fisher C. Dermatofibrosarcoma protuberans: pathology, genetics, and potential therapeutic strategies. Ann Diagn Pathol. 2016;25:64-71.
- Bogucki B, Neuhaus I, Hurst EA. Dermatofibrosarcoma protuberans: a review of the literature. Dermatol Surg. 2012;38(4):537-551.
- Larbcharoensub N, Kayankarnnavee J, Sanpaphant S, Kiranantawat K, Wirojtananugoon C, Sirikulchayanonta V. Clinicopathological features of dermatofibrosarcoma protuberans. Oncol Lett. 2016;11(1):661-667.
- Taylor RW. Sarcomatous tumors resembling in some respects keloid. J Cutan Genitourin Dis. 1890;8:384-387.
- Hoffmann E. Über das knollentreibende Fibrosarkom der Haut (Dermatofibrosarkoma protuberans). Dermatol Z. 1925;43:1-28.
- Akram J, Wooler G, Lock-Andersen J. Dermatofibrosarcoma protuberans: clinical series, national Danish incidence data and suggested guidelines. J Plast Surg Hand Surg. 2014;48(1):67-73.
- Rubio GA, Alvarado A, Gerth DJ, Tashiro J, Thaller SR. Incidence and outcomes of dermatofibrosarcoma protuberans in the US pediatric population. J Craniofac Surg. 2017;28(1):182-184.
- Llombart B, Serra-Guillén C, Monteagudo C, López Guerrero JA, Sanmartín O. Dermatofibrosarcoma protuberans: a comprehensive review and update on diagnosis and management. Semin Diagn Pathol. 2013;30(1):13-28.
- Trofymenko O, Zeitouni NC. Association of patient demographic characteristics with dermatofibrosarcoma protuberans tumor size at diagnosis in the U.S. National Cancer Database [published online August 14, 2017]. Br J Dermatol. doi:10.1111/bjd.15357
- Reha J, Katz SC. Dermatofibrosarcoma protuberans. Surg Clin North Am. 2016;96(5):1031-1046.
- Tsai Y-J, Lin P-Y, Chew K-Y, Chiang Y-C. Dermatofibrosarcoma protuberans in children and adolescents: clinical presentation, histology, treatment, and review of the literature. J Plast Reconstr Aesthet Surg. 2014;67(9):1222-1229.
- Han HH, Lim SY, Park YM, Rhie JW. Congenital dermatofibrosarcoma protuberans: a case report and literature review. Ann Dermatol. 2015;27(5):597-600.
- Valdivielso-Ramos M, Hernanz JM. Dermatofibrosarcoma protuberans in childhood. Actas Dermosifiliogr. 2012;103(10):863-873.
- Saiag P, Grob J-J, Lebbe C, et al. Diagnosis and treatment of dermatofibrosarcoma protuberans. European consensus-based interdisciplinary guideline. Eur J Cancer. 2015;51(17):2604-2608.
- Al Barwani AS, Taif S, Al Mazrouai RA, Al Muzahmi KS, Alrawi A. Dermatofibrosarcoma protuberans: insights into a rare soft tissue tumor. J Clin Imaging Sci. 2016;6:16.
- Bernard J, Poulalhon N, Argenziano G, Debarbieux S, Dalle S, Thomas L. Dermoscopy of dermatofibrosarcoma protuberans: a study of 15 cases. Br J Dermatol. 2013;169(1):85-90.
- Piccolo V, Russo T, Staibano S, et al. Dermoscopy of dermatofibrosarcoma protuberans on black skin. J Am Acad Dermatol. 2016;74(6):e119-e120.
- Sung THT, Tam ACW, Khoo JLS. Dermatofibrosarcoma protuberans: a comprehensive review on the spectrum of clinico-radiological presentations. J Med Imaging Radiat Oncol. 2017;61(1):9-17.
- Mullen JT. Dermatofibrosarcoma protuberans: wide local excision versus Mohs micrographic surgery. Surg Oncol Clin N Am. 2016;25(4):827-839.
- Wollina U. Dermatofibrosarcoma protuberans in a 10-year-old child. J Dermatol Case Rep. 2013;7(4):121-124.
- Criscito MC, Martires KJ, Stein JA. Prognostic factors, treatment, and survival in dermatofibrosarcoma protuberans. JAMA Dermatol. 2016;152(12):1365-1371.


