Torso Trauma in the Elderly
This article is the third in a continuing series on trauma care and the older adult. The series discusses the growing problem of trauma in the elderly, including its causes and possible ways to prevent it, care in the acute stages, and manifestations and treatment strategies when trauma involves the torso, spine, brain, and hip. Authors include skilled experts in the trauma field representing various specialties at the R Adams Cowley Shock Trauma Center at the University of Maryland Medical Center and the University of Maryland School of Medicine.
Introduction
Elderly persons, defined as persons age 65 years or older, constitute one of the fastest growing segments of the U.S. population. By the year 2030, the number of persons over age 65 will double relative to 2000, representing almost 20% of the nation’s total population.1 Today’s elderly population has an improved quality of life, enabling them to remain physically active and mobile longer.2 Ironically, this active lifestyle has become a risk factor for injury.
Some consider trauma to be exclusively a disease of the young; however, as the population in the United States ages over the next several decades, injuries in older patients will become more apparent. One must realize that trauma is sometimes a different disease in the elderly than in young people, with the elderly responding differently to their injuries.3 Advanced age has been shown to be a risk factor for poor outcomes in trauma patients; however, older patients may benefit from intensive monitoring and aggressive management following injury.4
Falls and motor vehicle collisions (MVCs) are the two most common mechanisms of injury (MOI) in the elderly. Falls tend to be ground-level falls, occurring in 30-40% of those over 65 years of age annually.5 In 2006, more than 177,000 elderly people suffered nonfatal injuries as a result of a MVC.6 Torso trauma represents the second leading cause of mortality in the elderly after brain injury.7 Older persons are at risk for the same thoracic injuries as are younger people. Some injuries, including pneumothorax and hemothorax, require essentially identical treatment, and do not require specific age-associated considerations. Others, including rib fractures, flail chest, and blunt aortic injuries, require specific considerations and will be the focus of this article. Additionally, nonoperative management (NOM) of solid organ injuries and rhabdomyolysis, often exacerbating post-injury course, will be discussed.
Rib Fractures in Older Persons
Rib fractures are the most common injury found in elderly patients with blunt trauma. The two predominant MOI for rib fractures in the elderly are MVCs followed by falls. Rib fractures are a significant cause of increased morbidity and mortality in older patients. This increase can be attributed to several factors including anatomical differences between young and elderly people (osteoporosis, decreased muscle mass, and thinned vertebral bodies), as well as decreased physiological reserve (low cardiopulmonary status and lower immunity). Additionally, the presence of pre-existing comorbidites is a known risk factor for increased morbidity and mortality in this patient population.8
Bulger et al9 demonstrated that in patients older than 65 years of age who had rib fractures, mortality increased 19%, and risk for pneumonia increased 27% for each rib fracture. In addition, elderly patients with rib fractures had significantly worse outcomes than those younger than age 65 with similar injury severity. Rib fractures in older persons have also been associated with intra-abdominal injury. Shweiki et al10 showed that the incidence of liver injuries was 10.7% in their population of patients with rib fractures, and 11.3% had splenic injuries. Close attention to vital signs and blood count is essential in this patient population. Because many elderly patients are prescribed rate-controlling medications (beta blockers), tachycardia may not be present as a sign of ongoing hemorrhage.
Flail chest is a serious, life-threatening chest injury often associated with underlying pulmonary injury. Flail chest is defined as fractures of three or more consecutive ribs in two or more places, and has a prevalence of 5-13% in patients with chest wall trauma.11 It seems logical that the elderly would be vulnerable to flail chest following trauma due to their decreased muscle mass, decreased bone density, and decreased chest wall compliance. Albaugh and colleagues12 demonstrated that for elderly patients with a flail chest, there was a statistically significant increase in the rate of mortality with increased age (58% vs 16%; P = 0.002). For those over age 80 years, there was 86% mortality rate for those with a flail chest.
The diagnosis of rib fractures is often made by clinical exam or on plain chest x-ray (CXR); however, as many as 50% of rib fractures may not be detected by CXR.13 There has been a marked increase in the use of chest computed tomography (CT) for the initial evaluation of patients with blunt chest trauma. It is clear that a CT scan identifies injuries not seen on plain films. Many clinicians question the significance of injury seen on CT scan not seen on CXR, and whether management is altered based on CT findings. However, with the known increase in morbidity and mortality in elderly persons with rib fractures, aggressive evaluation would seem prudent.
Pain control is crucial in the management of rib fractures. It allows for early ambulation, deep breathing, incentive spirometry, clearance of secretions, and prevention of atelectasis and pneumonia. Pain in younger patients is often well controlled with oral narcotics or nonnarcotic medications. However, pain management in the elderly can be a difficult and challenging issue. Many older individuals are very sensitive to narcotics and can become somnolent quite easily with only a small dose. Resultant decreased mental status and respiratory drive can occur, sometimes requiring intubation and mechanical ventilation. Frequent monitoring of pulse oximetry and the use of arterial blood gas may be beneficial.
Various strategies exist for pain management in the elderly, each carrying its own set of advantages, disadvantages, and complications (Table I). Parenteral narcotic analgesia is often effective but may be limited by central nervous system effects and further depression of respiratory drive. Morphine should not be used in patients with decreased renal function due to the difficulty of managing possible adverse effects related to its metabolites. Hydromorphone, oxycodone, methadone, and fentanyl are the recommended opioids for patients with renal failure. If a patient is receiving dialysis, it is important to note that neither fentanyl nor methadone is dialyzed. Nonnarcotics such as ketorolec, although very effective, may worsen renal function in elderly patients with a pre-existing decreased glomerular filtration rate.
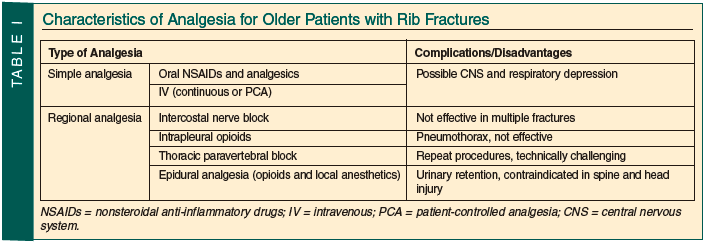
The use of epidural analgesia with agents such as bupivacaine and fentanyl provides superior pain control without the sedating effect of parenteral opioids.14,15 Some reports indicate that epidural analgesia is associated with a decrease in the rate of nosocomial pneumonia and a shorter duration of mechanical ventilation after rib fractures.16 In one large study, epidural use demonstrated a significant reduction in mortality from 16% to 10% at 48 hours post-injury.17 Unfortunately, pre-existing comorbidities such as degenerative spine disease or those requiring the use of aspirin, clopidogrel, or warfarin may preclude the use of epidural analgesia. Complications with epidural catheters do occur. Bulger and colleagues9 demonstrated a 19% complication rate with catheter placement. Most were local infection, transient hypotension, and catheter migration. No epidural hematomas or epidural abscesses were reported as a result of catheter placement.
In high-risk individuals in whom an epidural is contraindicated, paravertebral analgesia may be used.18 Paravertebral anesthesia has been shown to provide effective pain relief in patients with multiple rib fractures and in those who have recently undergone thoracotomies. Pulmonary complications, urinary retention, nausea and vomiting, and hypotension are less common with paravertebral anesthesia than with epidurals.
As a general rule, elderly trauma patients who have more than two isolated rib fractures should be admitted to the hospital for observation. Those with six or more have a high morbidity and mortality, and should be admitted to an Intensive Care Unit (ICU).19 Aggressive pulmonary toilet, airway monitoring, and pain control should be initiated early in the Emergency Department. Epidural anesthesia is recommended as a first-line treatment for persons older than age 65 years with more than four rib fractures.20
Traumatic Aortic Injury
Traumatic aortic injury (TAI) is most often a result of a rapid deceleration injury. It commonly occurs after MVCs or falls of significant height. Injuries due to deceleration are believed to occur at points of strain. In the aorta, this corresponds to the isthmus, the point just distal to the left subclavian artery where the ductus arteriosus and thoracic aorta attach.21
There are three categories of patients with TAI: (1) those who die at the scene (70-80% of the whole); (2) those who present unstable or become unstable (2-5%, with mortality rates of 90-98%); and (3) those who are hemodynamically stable and are diagnosed 4-18 hours after injury (15-25%, with mortality rates of 25% largely due to associated injuries).2
Advanced age is known to be associated with higher mortality rates for blunt thoracic trauma, specifically those with aortic injuries.23 Factors contributing to the increase include the lack of physiologic reserve to compensate for severe multiple injuries, as well as aortic wall age-related changes. Aortic dilation, increased aortic stiffness, and elevated blood pressure are also responsible for the high wall stress generated in the fragile aorta in older persons, making them more susceptible to injury.
Diagnosis of aortic rupture is challenging and requires a high index of suspicion. Signs and symptoms are often nonspecific and unreliable. Some patients state that lying down causes severe back pain and prefer to be upright, while others present pale and clammy. On exam, some patients may be hoarse or exhibit a difference in blood pressure between the upper and lower extremities or between the right and left upper arm. However, as many as 50% of patients may have a normal physical exam.24
Work-up for patients suspected of having a TAI most often begins with a plain chest radiograph. Widening of the mediastinum correlating to mediastinal hematoma is the classic first indicator seen on CXR.25 However, a normal CXR does not rule out the diagnosis. Many elderly trauma patients have ectatic and tortuous aortas obscuring the normal contour, thus making it difficult to rule out an injury. Those with a MOI involving deceleration and a widened mediastinum on CXR require further imaging. To assume an underlying ectatic aorta would be unwise. Retrograde aortography was traditionally used for definitive diagnosis. It has 96% sensitivity and 98% specificity26; however, it is an invasive procedure and has complication rates of up to 1%.
Currently, CT is the modality of choice for the diagnosis of TAI. Helical CT angiography appears to be more sensitive than angiography in detecting small intimal injuries that are still significant.2 It has been shown to have 100% sensitivity, 99.7% specificity, an 89% positive predictive value, and a 100% negative predictive value.27 In addition, current 3-dimensional reconstruction gives valuable data with which to plan operative or endovascular approaches. Although CT represents a less invasive diagnostic modality than aortography with similar results, it still places patients, specifically the elderly, at risk for contrast-induced nephropathy.
Once initial stabilization is completed and ongoing hemorrhage is controlled, all TAI should be initially treated using a medical management approach. The goal is to prevent progression of the aortic tear to a catastrophic rupture leading to exsanguination and death. Similar to the pharmacologic treatment of an aortic dissection, change in aortic pressure is best controlled by decreasing the force of left ventricular ejection in systole with beta blocker therapy (negative inotrope). The mean arterial pressure of 60-70 mm Hg maintains perfusion to kidneys and brain and is used as a target point.
Medical treatment should begin with beta blocker therapy unless contraindicated, adding vasodilators as needed. Preferred beta blocker agents include esmolol and labetalol. Esmolol is an ultra-short-acting intravenous cardioselective beta antagonist. It has an extremely short half-life (mean, 9 min; range, 4-16 min). A quick onset of action (60 sec) and full recovery from beta blockade is observed 18-30 minutes after termination of the infusion. A loading dose of 0.5 to 1 mg/kg may be initiated, followed by an infusion at 50 to 300 μg/kg/min. Esmolol is easily titrated to response, and adverse effects such as hypotension or bradycardia are rapidly managed by termination of the infusion. Labetalol is a combined alpha and beta blocker (1:7 ratio). Its onset of action occurs within 2-5 minutes, with peak effect in 5-15 minutes. Duration of action is 2-4 hours. Labetalol may be administered as 20-mg intravenous boluses (up to 80 mg) or as a continuous infusion starting at 1-2 mg/min to a total of 1-2 mg/kg. When beta blocker therapy is contraindicated, a vasodilator may be used. Nitroprusside is the most common vasodilator used to manage blood pressure in patients with TAI. Nitroprusside is a combined arterial and venous vasodilator. It is very potent, acting within seconds, with duration of action of 1-2 minutes. It is administrated as an infusion starting at 0.3-0.5 μg/kg/min and titrated to desired effect. It may cause a reflex tachycardia, which may necessitate addition of a beta blocker. Other disadvantages of nitroprusside include decreased cerebral blood flow and coronary steal.
The surgical treatment for TAI has undergone change in the last few decades. The concept that all aortic injuries required immediate operative repair has undergone significant modification. Delayed operative repair is feasible and can allow time to correct associated injuries that would preclude or significantly increase the risk of operation.28 Furthermore, traditional open thoracotomy using either a “clamp and sew” technique or repair with bypass is no longer the considered first-line treatment. Most high-volume trauma centers are now using endovascular stent grafts (SGs) for repair of TAI. Additionally, conservative management, with blood pressure and heart rate control, may be a reasonable alternative to surgical repair in a high-risk patient population.
SG was initially recommended for patients with multiple injuries or those with comorbid diseases, such as the elderly.29 An endovascular approach to aortic pathology is now accepted practice in trauma care. It does not require a thoracotomy, single-lung ventilation, aortic cross-clamping, or systemic heparinization. As a result, endovascular repair serves as an effective, minimally invasive treatment alternative, especially in patients with severe concomitant injuries, which may be prohibitive to open surgical repair. In 2008, Demetriades and colleagues30 compared results of SG versus traditional operative approach. The authors demonstrated that the elderly, specifically those over the age of 70, were more likely to have had a SG placed than open operation. Neschis et al31 reviewed the world’s medical literature and identified 23 reports that included five or more patients. Among 220 patients undergoing endovascular repair, there were 15 deaths (a rate of 6.8%). In this series of 39 consecutive patients, there were five deaths (12.8%) and no cases of paraplegia.31 None of the deaths were related to the SG. Patel et al32 compared open repair versus endovascular repair in patients older than 75 years of age. The authors demonstrated a reduced early mortality in the SG group, as well as a shorter hospital stay.
The long-term disability of aortic SG for trauma has not been well described. Still, there has been noted a 20% device-related complication rate and a 14.4% endoleak rate, one-third of which required subsequent open repair.30 It is clear that long-term follow-up is required for all patients. CT scan should be performed three times in the first year, and then yearly as long as no problems are identified.
Solid Organ Injuries
The diagnosis of blunt abdominal trauma is suspected based on the MOI, the presence of associated injuries (eg, right or left lower rib fractures), and physical exam findings. The presence of obvious peritonitis or hemodynamic instability is a strong indication for emergency laparotomy. However, in patients with multitrauma with impaired level of consciousness, physical examination has significant limitations and may be misleading. Evaluation of older patients who have sustained blunt abdominal trauma may create a significant diagnostic challenge. Absence of hypotension or tachycardia does not always mean hemodynamic stability. Elderly patients with chronic hypertension or those taking beta blockers may not show the classic clinical signs of hemodynamic instability.
Ultrasound or the focused assessment with sonography for trauma (FAST) is often the initial diagnostic test. Patients who are hemodynamically unstable and have evidence of hemorrhage on FAST go emergently to the Operating Room for an exploratory laparotomy. Stable patients with or without evidence of hemorrhage who require additional imaging have a CT performed. CT is the main diagnostic tool for the evaluation of solid organ injury after trauma. Findings of Esses et al33 demonstrate the ability of CT to change important clinical decisions (need for admission, surgery, antibiotics, most likely diagnosis) in elderly patients in the Emergency Department with abdominal pain not related to trauma.
NOM of solid organ injuries due to blunt trauma has become the standard of care in hemodynamically stable patients. The management of blunt abdominal trauma and solid organ injury in older patients is not a simple decision-making process. Historically, surgeons have considered advanced age as a prohibitive risk for consideration of NOM of blunt solid organ injury.34 Increased splenic fragility and decreased physiologic reserves associated with aging explain why older patients are twice as likely to be managed operatively.35
Early studies suggested a 60-90% failure rate for NOM of blunt splenic injuries in patients older than age 55 years.34 In 2002, Albrecht et al36 reported a nonoperative failure rate of 33% in geriatric patients. Studies indicate that in properly selected elderly patients with blunt trauma, NOM has success rates of 62-85%.37,38
Ninety percent of hepatic trauma is managed nonoperatively, with failure rates ranging from 5-15% across all age groups.39 Improvements in diagnostic imaging and ICU monitoring have permitted safe observation of patients previously thought to require operative intervention. While there are no studies specifically addressing radiologic findings in elderly patients that could be predictive of failure of NOM, many authors recommend NOM in all hemodynamically stable patients without regard to age and grade of injury.40,41
Predictors of NOM failure include hypotension on admission, high CT grades of injury, active contrast extravasation on CT, and the need for blood transfusion.42,43 The importance of other factors such as advanced age or neurologic injury has been debated. If the CT scan shows a low degree of splenic injury and a patient is hemodynamically stable, the presence of a head injury does not dictate a need for operation. Those who failed NOM were noted to have higher grades of splenic injury and free intraperitoneal fluid levels.43
Complications of NOM occur in approximately 3-11% of adult patients. Operative reports suggest a 1-3% rate of missed bowel injuries, delayed hemorrhage, and delayed infections.44 Most elderly patients who fail NOM do so within the first 48-72 hours regardless of embolization after injury, and those patients who fail NOM seem to have worse clinical outcomes than younger patients. Consideration of operative intervention should occur with unstable hematocrit, increasing abdominal pain, persistent unexplained tachycardia, or hemodynamic decompensation or instability. Markers of occult hypoperfusion, such as lactate and base excess, may play an important role in identifying the older trauma patient who has a solid organ injury and impending hemodynamic collapse. A repeat CT scan in patients with solid organ injuries grades 3 and more within 48-72 hours may detect pseudoaneurysm unrecognized at previous study. This policy is accepted by most trauma centers.
Angiographic embolization (AE) plays an important role in the NOM of solid organ injuries in the elderly. In a series of 150 adult patients, Sclafani and colleagues45 demonstrated a 98.5% salvage rate with splenic AE on hemodynamically stable trauma patients. Other studies report failure rates closer to 10-13.5% with a 20% complication rate (eg, recurrent hemorrhage, missed injuries, infection).46 No reports specifically address the use of AE in older persons. However, angiography is a reasonable option for the hemodynamically stable elderly trauma patient who has solid organ injury and a blush or extravasation on CT. Complications occurred in more than 50% of patients undergoing angioembolization for solid organ injuries (Table II). Serious organ-related complications are more common after liver embolization.47,48 In addition, patients are exposed to high volumes of contrast, with 5.1% chance of contrast-induced nephropathy in general populations.49 Elderly persons, especially those with impaired renal function, are at highest risk—more than seven times that of their young counterparts. This kidney dysfunction may persist and require long-term dialysis.
Currently, age is no longer considered an exclusionary criterion for the NOM of solid organ injuries. As experience with NOM of blunt trauma increases, all patients, regardless of age, who sustain blunt splenic and hepatic injuries, and whose conditions are hemodynamically stable, are candidates for NOM of their injuries.

Rhabdomyolysis
Rhabdomyolysis occurs in up to 85% of patients with traumatic injuries. It is a potentially life-threatening condition that must be suspected in all trauma patients with a MOI consistent with potential skeletal muscle damage. Close attention is warranted for patients who have a crush injury, even without significant external signs of trauma. In addition, alcoholics and elderly patients with a fall or stroke who have prolonged muscle compression require evaluation for rhabdomyolysis. Those who develop subsequent renal failure have a mortality of approximately 20%.50
Rhabdomyolysis and crush syndrome are common results of natural disasters such as earthquakes. Age has been shown to be the only independent predictor of outcome.5 Rhabdomyolysis in patients with head trauma might be explained by muscle damage caused by blunt trauma to the torso and immobilization. It has rarely been reported when a surgical procedure is performed in an improper position or following the prolonged use of a tourniquet.52,5 Among hospitalized patients, nontraumatic causes of rhabdomyolysis occur five times more frequently than traumatic causes.54
Muscle injury, regardless of mechanism, results in a cascade of events that leads to leakage of large quantities of potassium, phosphate, myoglobin, and creatine kinase (CK) into the circulation. Myoglobin in the renal glomerular filtrate can precipitate and cause renal tubular obstruction, leading to renal damage.55 An elevated serum CK is the most sensitive and reliable indicator of muscle injury. Myoglobin elevation occurs before CK levels rise following injury; however, it is rapidly cleared through renal excretion and metabolism to bilirubin. Myoglobinuria, a urinalysis positive for hemoglobin without red blood cells on microscopic examination, is a hallmark for the diagnosis of rhabdomyolysis. Visual inspection of the urine will often demonstrate a dark tea–colored urine.
The classic triad of symptoms of muscle pain, weakness, and dark urine has been seen in less than 10% of patients. However, many patients who develop rhabdomyolysis are critically ill, on a ventilator, and unable to articulate specific symptoms. Thus, the care provider is left with clinical evaluation that includes physical exam and laboratory values.
Complications of rhabdomyolysis include acute renal failure, metabolic derangements, and disseminated intravascular coagulation. Close monitoring of renal function including blood urea nitrogen (BUN) and creatinine levels is imperative. Hypocalcemia, the most common electrolyte abnormality, occurs early and is usually asymptomatic. Hyperkalemia can become life-threatening and must be treated emergently. Treating the underlying cause is crucial. However, traditional treatment of hyperkalemia and replacement of calcium may be necessary.
Patients with obvious soft-tissue injury do not pose a diagnostic challenge for clinicians. However, those with a normal external physical exam may still develop rhabdomyolysis. Patients with extremity fractures, especially lower leg, need to be monitored closely for compartment syndrome that can lead to rhabdomyolysis. Frequent exams for calf fullness or measurement of compartment pressure are warranted. In addition, patients who have been exposed to propofol can develop propofol infusion syndrome (PIS). PIS should be suspected in patients taking propofol who have an unexplained acidosis and increased CK and myoglobin.
All patients with rhabdomyolysis should be admitted for intravenous hydration, serial laboratory evaluation, and management of potential complications. A monitored bed may be most appropriate for the first 24-48 hours, particularly for older patients, severely injured patients, and those with cardiac or renal comorbidities, as these patients tend to develop hyperkalemia and cardiac dysrhythmias. Higher levels of CK do not always indicate a higher risk for complications. However, elderly debilitated patients with lower elevations in total CK levels could progress to renal failure, and therefore would represent a greater clinical concern.56
The treatment of rhabdomyolysis includes initial stabilization and resuscitation of the trauma patient while concomitantly attempting to preserve renal function. Retrospective analysis demonstrates that early aggressive fluid replacement with saline is beneficial in minimizing the occurrence of renal failure. This may be challenging in the elderly who have underlying cardiac dysfunction. Large-volume resuscitation can precipitate pulmonary edema and require mechanical intubation. Those patients deemed to require such a large resuscitation should be in an ICU for closer monitoring.
It has been demonstrated that the longer it takes for rehydration to be initiated, the more likely it is that renal failure will develop.53,54 The goal should be to ensure adequate urine output (200-300 mL/hr). While randomized controlled trials are lacking, the available evidence suggests that mannitol and bicarbonate have no benefit over the aggressive fluid resuscitation.57 If rhabdomyolysis leads to acute renal failure, renal replacement therapy may be indicated. Conventional hemodialysis does not effectively remove myoglobin, due to its large protein size. Continuous venovenous hemofiltration has shown some efficacy in removing myoglobin, principally with the use of super high-flux filters and high volumes of ultrafiltration.58 Plasmapheresis has not been shown to have any effect on outcomes or on the myoglobin clearance.59
Conclusion
There continues to be an increasing number of older persons sustaining torso trauma. This article attempts to summarize the most important aspects of torso trauma affecting the elderly and presents specific treatment options. Treatment of geriatric trauma patients follows the same pattern as that for younger patients. Caution and a high index of suspicion to injuries specific for this age group are required to optimize treatment. Understanding the physiologic changes of aging may help explain the increased morbidity and mortality associated with torso trauma in the geriatric population.
Primary care providers should enhance their understanding of the complex issues of geriatric trauma to facilitate prevention and to assist the patient’s recovery to normal function. Implementation of geriatric trauma programs is mandatory in order to further improve outcomes of geriatric trauma victims. Areas that need further study include specific aspects of trauma prevention, trauma system care, pre-hospital triage guidelines, and trauma interventions that improve both short- and long-term outcomes in the elderly.
The authors report no relevant financial relationships.
Drs. Bala and Menaker are from the Department of Surgery, Division of Surgical Critical Care, University of Maryland School of Medicine, R Adams Cowley Shock Trauma Center, Baltimore. Dr. Gambert is Professor of Medicine and Co-Director, Division of Gerontology and Geriatric Medicine, Department of Medicine, University of Maryland School of Medicine, Director of Geriatric Medicine, University of Maryland Medical Center and R Adams Cowley Shock Trauma Center, and Professor of Medicine, Division of Gerontology and Geriatric Medicine, Johns Hopkins University School of Medicine, Baltimore. Dr. Stein is Chief of Critical Care, R Adams Cowley Shock Trauma Center, and Associate Professor of Surgery, Department of Surgery, University of Maryland School of Medicine.
References
1. He W, Sengupta M, Velkoff VA, DeBarro KA. U.S. Census Bureau. 65+ in the United States: 2005. Current population reports. U. S. Government Printing Office. Issued December 2005. http://www.census.gov/prod/2006pubs/p23-209.pdf. Accessed February 4, 2010.
2. Schwab CW, Kauder DR. Trauma in the geriatric patient. Arch Surg 1992;127:701-706.
3. Osler T, Hales K, Baack B, et al. Trauma in the elderly. Am J Surg 1988;156:537-543.
4. Demetriades D, Karaiskakis M, Velmahos G, et al. Effect on outcome of early intensive management of geriatric trauma patients. Br J Surg 2002;89:1319-1322.
5. King MB, Tinetti ME. A multifactorial approach to reducing injurious falls. Clin Geriatr Med 1996;12:745-759. p> 6. WISQARS™ (Web-based Injury Statistics Query and Reporting System). 2006. Centers for Disease Control and Prevention. www.cdc.gov/injury/wisqars/index.html. Accessed February 4, 2010.
7. Wang SC, Schneider L. An aging population: Fragile, handle with care. Proceedings of the National Highway Traffic Safety Administration; 1999.
8. Osler T, Hales K, Baack B, et al. Trauma in the elderly. Am J Surg 1988;156:537-543. 9. Bulger EM, Arneson MA, Mock CN, Jurkovich CJ. Rib fractures in the elderly. J Trauma 2000;48(6):1040-1047.
10. Shweiki E, Klena J, Wood GC, Indeck M. Assessing the true risk of abdominal solid organ injury in hospitalized rib fracture patients. J Trauma 2001;50:684-688.
11. LoCicero J 3rd, Mattox KL. Epidemiology of chest trauma. Surg Clin North Am 1989;69:15-19.
12. Albaugh G, Kann B, Puc MM, et al. Age-adjusted outcomes in traumatic flail chest injuries in the elderly. Am Surg 2000;66:978-981.
13. Blaisdell FW, Trunkey DD, eds. Cervicothoracic Trauma (Trauma Management). Volume III. New York, NY: Thieme; 1986.
14. Bergeron E, Lavoie A, Clas D, et al. Elderly trauma patients with rib fractures are at greater risk of death and pneumonia. J Trauma 2003;54:478-485.
15. Wu CL, Jani ND, Perkins FM, Barquist E. Thoracic epidural analgesia versus intravenous patient-controlled analgesia for treatment of rib fracture pain after motor vehicle crash. J Trauma 1999;47:564-567.
16. Barnea Y, Kashtan H, Skornick Y, Werbin N. Isolated rib fractures in elderly patients: Mortality and morbidity. Can J Surg 2002;45:43-46.
17. Bakhos C, O’Connor J, Kyriadides T, et al. Vital capacity as a predictor of outcome in elderly patients with rib fractures. J Trauma 2006;61:131-134.
18. Joshi GP, Bonnet F, Shah R, et al. A systematic review of randomized trials evaluating regional techniques for postthoracotomy analgesia. Anesth Analg 2008;107(3):1026-1040.
19. Kieninger AN, Bair HA, Bendick PJ, Howells GA. Epidural versus intravenous pain control in elderly patients with rib fractures. Am J Surg 2005;180:327-330.
20. Simon BJ, Cushman J, Barraco R, et al; EAST Practice Management Guidelines Work Group. Pain management guidelines for blunt thoracic trauma. J Trauma 2005;59:1256-1267.
21. Neschis DG, Scalea TM, Flinn WR, Griffith BP. Blunt aortic injury. N Engl J Med 2008;359:1708-1716.
22. Mattox KL, Wall MJ Jr. Historical review of blunt injury to the thoracic aorta. Chest Surg Clin N Am 2000;10:167-182.
23. Sturm JT, Billiar TR, Dorsey JS, et al. Risk factors for survival.following surgical treatment of traumatic aortic rupture. Ann Thorac Surg 1985;39:418-421.
24. Kram HB, Appel PL, Wohlmuth DA, Shoemaker WC. Diagnosis of traumatic thoracic aortic rupture: A 10-year retrospective analysis. Ann Thorac Surg 1989;47:282-286.
25. Marnocha KE, Maglinte DD. Plain-film criteria for excluding aortic rupture in blunt chest trauma. AJR Am J Roentgenol 1985;144:19-21.
26. Dyer DS, Moore EE, Ilke DN, et al. Thoracic aortic injury: how predictive is mechanism and is chest computed tomography a reliable screening tool? A prospective study of 1,561 patients. J Trauma 2000;48:673-683.
27. Mirvis SE, Shanmuganathan K, Buell J, Rodriguez A. Use of spiral computed tomography for the assessment of blunt trauma patients with potential aortic injury. J Trauma 1998;45:922-930.
28. Pate JW, Gavant ML, Weiman DS, Fabian TC. Traumatic rupture of the aortic isthmus: Program of selective management. World J Surg 1999;23:59-63.
29. Cook J, Salerno C, Krishnadasan B, et al. The effect of changing presentation and management on the outcome of blunt rupture of the thoracic aorta. J Thorac Cardiovasc Surg 2006;131:594-600.
30. Demetriades D, Velmahos GC, Scalea TM, et al; American Association for the Surgery Trauma Thoracic Aortic Injury Study Group. Operative repair or endovascular stent graft in blunt traumatic thoracic aortic injuries: Results of an American Association for the Surgery of Trauma Multicenter Study. J Trauma 2008;64:561-571.
31. Neschis DG, Moaine S, Gutta R, et al. Twenty consecutive cases of endograft repair of traumatic aortic disruption: Lessons learned. J Vasc Surg 2007;45:487-492. Published Online: January 24, 2007.
32. Patel HJ, Williams DM, Upchurch GR, et al. A comparison of open and endovascular descending thoracic aortic repair in patients older than 75 years of age. Ann Thorac Surg 2008;85:1597-1604.
33. Esses D, Birnbaum A, Bijur P, et al. Ability of CT to alter decision making in elderly patients with acute abdominal pain. Am J Emerg Med 2004;22:270-272.
34. Godley CD, Warren RL, Sheridan RL, McCabe CJ. Nonoperative management of blunt splenic injury in adults: Age over 55 years as a powerful indicator for failure. J Am Coll Surg 1996;183(2):133-139.
35. Tsugawa K, Koyanagi N, Hashizume M, et al. New insight for management of blunt splenic trauma: Significant differences between young and elderly. Hepatogastroenterology 2002;49:1144-1149.
36. Albrecht RM, Schermer CR, Morris A. Nonoperative management of blunt splenic injuries: Factors influencing success in age > 55 years. Am Surg 2002;68(3):227-231.
37. Barone JE, Burns G, Svehlak SA, et al. Management of blunt splenic trauma in patients older than 55 years. Southern Connecticut Regional Trauma Quality Assurance Committee. J Trauma 1999;46:87-90.
38. Falimirski ME, Provost D. Nonsurgical management of solid abdominal organ injury in patients over 55 years of age. Am Surg 2000;7:631-635.
39. Richardson JD. Changes in the management of injuries to the liver and spleen. J Am Coll Surg 2005;200(5):648-669.
40. Knudson MM, Maull KI. Nonoperative management of solid organ injuries. Past, present, and future. Surg Clin North Am 1999;79:1357-1371.
41. Maull KI. Current status of nonoperative management of liver injuries. World J Surg 2001;25:1403-1404.
42. Velmahos GC, Chan LS, Kamel E, et al. Nonoperative management of splenic injuries: Have we gone too far? Arch Surg 2000;135:674-681.
43. Ochsner MG. Factors of failure for nonoperative management of blunt liver and splenic injuries. World J Surg 2001;25:1393-1396.
44. Shapiro MB, Nance ML, Schiller HJ, et al. Nonoperative management of solid abdominal organ injuries from blunt trauma: impact of neurologic impairment. Am Surg 2001;67:793-796.
45. Sclafani SJ, Shaftan GW, Scalea TM, et al. Nonoperative salvage of computed tomography-diagnosed splenic injuries: Utilization of angiography for triage and embolization for hemostasis. J Trauma 1995;39(5):818-827.
46. Haan JM, Bochicchio GV, Kramer N, Scalea TM. Nonoperative management of blunt splenic injury: A 5-year experience. J Trauma 2005;58(3):492-428.
47. Dabbs DN, Stein DM, Scalea TM. Major hepatic necrosis: A common complication after angioembolization for treatment of high-grade liver injuries. J Trauma 2009;66:621-629.
48. Stein DM, Scalea TM. Nonoperative management of spleen and liver injuries. J Intensive Care Med 2006;21:296-304.
49. Hipp A, Desai S, Lopez C, Sinert R. The incidence of contrast-induced nephropathy in trauma patients. Eur J Emerg Med 2008;15(3):134-139.
50. Bagley WH, Yang H, Shah KH. Rhabdomyolysis. Intern Emerg Med 2007;2:210-218. Published Online: October 1, 2007.
51. Sever MS, Erek E, Vanholder R, et al; Marmara Earthquake Study Group. The Marmara earthquake: Epidemiological analysis of the victims with nephrological problems. Kidney Int 2001;60:1114-1123.
52. Biswas S, Gnanasekaran I, Ivatury R, et al. Exaggerated lithotomy position-related rhabdomyolysis. Am Surg 1997;63:361-364.
53. Odeh M. The role of reperfusion-induced injury in the pathogenesis of the crush syndrome. N Engl J Med 1991;324:1417-1422.
54. Visweswaran P, Guntupalli J. Rhabdomyolysis. Crit Care Clin 1999;15:415-428, ix-x.
55. Knochel JP. Mechanisms of rhabdomyolysis. Curr Opin Rheumatol 1993;5:725-731.
56. Ye X, Rastogi A, Nissenson AR. Renal replacement therapy in the elderly. Clin Geriatr Med 2009;25:529-542.
57. Huerta-Alardin AL, Varon J, Marik PE. Bench-to-bedside review: Rhabdomyolysis—An overview for clinicians. Crit Care 2005;9:158-169.
58. Ronco C, Bellomo R, Homel P et al. Effects of different doses in continuous veno-venous hemofiltration on outcomes of acute renal failure: A prospective randomized trial. Lancet 2000;356:26-30.
59. Conger J. Interventions in clinical acute renal failure: Where are the data? Am J Kidney Dis 1995;26:565-576.


