Smoking Cessation in the Elderly
At the conclusion of this activity, participants should be able to:
1. Identify the trends in prevalence of tobacco use over the past 50 years.
2. Recognize that smoking cessation is beneficial in the geriatric population.
3. Demonstrate an approach to the smoking geriatric patient who desires to quit.
4. Recommend pharmacologic treatment to the nicotine-dependent geriatric patient.
___________________________________________________________________________________________________________
INTRODUCTION
Cigarette smoking remains the leading preventable cause of death in the United States, contributing to one out of five deaths (438,000 people) every year. It was estimated in 2005 that 22.4% of adults age 45-64 years and 9.1% of adults age 65 years and older are current smokers.1 The decrease in prevalence among the elderly is in part due to the mortality in this group, as smokers die 13-14 years earlier than nonsmokers. The geriatric smoker has typically been smoking for a longer period of time than the younger smoker, and efforts to achieve cessation in this population pose a significant challenge for the practicing clinician. Many of the geriatric smoking population experience symptoms or clinical sequelae from long-term tobacco use. However, cessation of smoking in this age group can still yield beneficial effects on morbidity and mortality.
No clinical trials focusing specifically on smoking cessation methodologies or outcomes in the elderly are available. Numerous intervention trials include elderly patients in their study population and report their results in the different age subgroups. Similarly, studies exist that focus on smoking cessation methodologies and include elderly patients in their study populations. Ideally, the practicing clinician would benefit from age-specific guidelines based on clinical studies conducted on the geriatric age group controlled for comorbidities. In the absence of this database, we will attempt to identify and summarize the evidence in smoking cessation that can be extrapolated to the elderly population. Data from trials that addressed smoking cessation interventions or methodologies and included older smokers will be reviewed with the aim of assisting the practicing clinician in addressing smoking cessation in the elderly.
EPIDEMIOLOGY
Today’s elderly persons achieved adulthood during a time when smoking was socially acceptable and highly prevalent in all age groups. The prevalence of smoking increased markedly after World War II and reached a plateau during the 1970s. Cigarette smoking at that time was widely promoted in the media for its “beneficial” effects on mood, weight control, and social appeal. Smoking was allowed in physician offices and in hospitals. It was not until 1960 that the distribution of free cigarettes was stopped at annual medical and public health meetings; until 1969 elderly patients at nursing homes were still being given free cigarettes on the annual “Respect for the Aged” holiday. The first Surgeon General’s Report linking smoking and lung cancer was issued in 1964. In the same year, however, the American Medical Association supported the tobacco industry’s objection to labeling cigarettes as a health hazard, citing a lack of scientific evidence to support the relationship of smoking to lung cancer.
However, as more research data and epidemiological evidence accumulated, citizens around the world became aware of the hazards of smoking and initiated anti-smoking campaigns. Smoking cessation became a recognized public health effort, and awareness of the addictive properties of tobacco spread. By 1999, 27% of American men and 23% of American women smoked.2 This was a significant decrease from the 1965 prevalence rates of 51.9% and 33.9% among U.S. adult male and female smokers, respectively. This decrease can be attributed to multiple factors. The ban on tobacco advertising in the television and radio media in 1971 had a major impact in the United States. Anti-smoking campaigns were directed at the adolescent, smokingnaïve population. In addition, the increasing cost of cigarettes has had an important effect on the adolescent population; the average cost of cigarettes in 1965 was $1.10 per pack, and it is now over $3.50. Many of those who began smoking in the 1960s, however, continue to smoke at the present time.
The long duration of continuous and cumulative injurious effects of tobacco use has resulted in a disproportionately high incidence of tobacco-related diseases among the elderly.3 The most common causes accounting for excess smoking-related mortality in patients over age 60 are lung cancer, cardiovascular disease, and chronic obstructive pulmonary disease.3 Smoking is implicated in 84% of lung cancers and in the great majority of patients with chronic obstructive lung disease. It is estimated that 50% of long-time smokers will die from a tobacco-related illness.2
Studies comparing mortality rates between smokers and nonsmokers found a dose-related increase in mortality rates among smokers.4 Vollset et al4 conducted a study that followed the survival of smokers and nonsmokers from ages 40 to 70, and demonstrated a significant mortality increase in the smoking group. This difference in mortality rates became larger as the populations aged. This difference reached its maximum at age 70, beyond which patients were not followed (Figure 1).
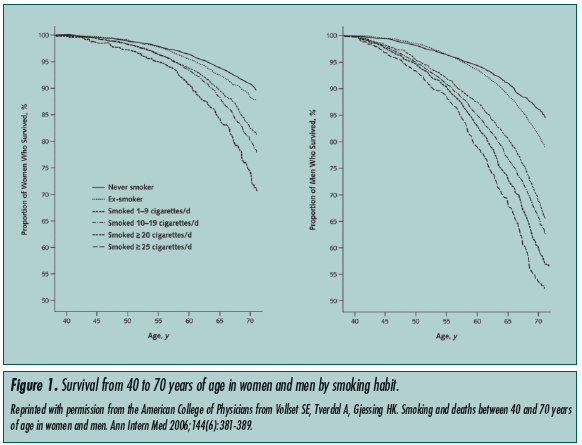
Smoking cessation reduces morbidity and mortality in smokers, even for those who have smoked continuously for more than 30 years. The benefits of cessation are more immediate for cardiovascular disease, and are seen over a longer term for pulmonary disease.5 The degree of improvement depends on the reversibility of the disease processes at the time of cessation. Though the long-term smoking geriatric patient stands to benefit from cessation, insidious irreversible pathological processes may have already taken root. Nonetheless, the 1990 Surgeon General’s Report concluded that smoking cessation is beneficial at all ages.6
INTERVENTIONS
Older smokers are less likely than younger smokers to attempt quitting. The Stages of Change Model,7 which is summarized in Table I, provides a framework for understanding behavior change in adults. Geriatric patients are more likely to reside in the pre-contemplation stage than their younger counterparts. Many smokers who have continued to smoke into their 60s have done so because they are not interested in quitting. However, the older patient who attempts to quit is more likely to be successful.3 This trend is consistent among all smoking cessation interventions, which include behavioral therapy and pharmacotherapy. This phenomenon may result from the personal insights or self-motivation gained through personally experiencing the symptoms or clinical sequelae of long-term tobacco use. Hence, encouraging the older smoker to move from the precontemplation stage to the contemplation or action stage can result in a positive behavioral change, despite decades of continuous smoking.

Counseling
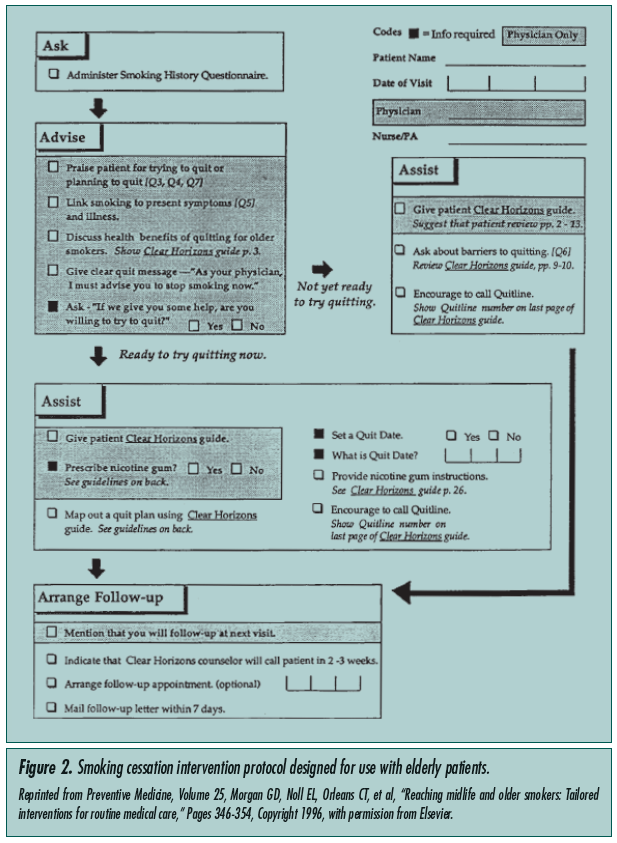
Counseling about smoking cessation is recommended in every visit a patient makes to his/her primary care provider. It is of special importance in older smokers, as they are more likely to smoke due to nicotine dependence rather than behavioral or psychosocial factors.2 In patients age 60 years and over, the rate of smoking cessation was significantly higher in those who received counseling by their primary care physician and a nurse offering advice on smoking cessation in a non-urgent visit when compared to routine primary care (14% vs 9%).2 Morgan et al5 investigated counseling older smokers by trained healthcare workers using an older patient–specific approach during regular visits. A similar increase in abstinence rate was observed comparing an active counseling group to routine care (15.4% vs 8.2%). In the latter study, physicians and healthcare workers were trained for 45-50 minutes on smoking cessation counseling and interventions based on the National Cancer Institute (NCI) manual.8 Patients were given a copy of a smoking cessation guide designed for the elderly titled Clear Horizons. Figure 2 shows the NCI protocol that was followed for each participant, entitled Helping Your Patient Stop Smoking.5 Similar data from comparable studies show that such directed training of healthcare workers roughly doubles the abstinence rates in all age groups studied. In an inpatient study of elderly patients discharged with acute myocardial infarction, those who received smoking cessation counseling prior to discharge had an improved 5-year risk of death.9 Lastly, though the utility of behaviorally oriented group approaches for the older smoker has been well established,5 many smokers prefer personalized and individualized counseling.
Pharmacotherapy
According to the U.S. Clinical Practice Guideline,10 nicotine replacement therapy (NRT) and bupropion SR are reliable first-line pharmacotherapies. The evidence for their effect on smoking cessation is ranked as A-level of evidence by the Agency for Healthcare Research and Quality.10 Clonidine and nortriptyline are listed as second-line options, due to B-level of evidence.
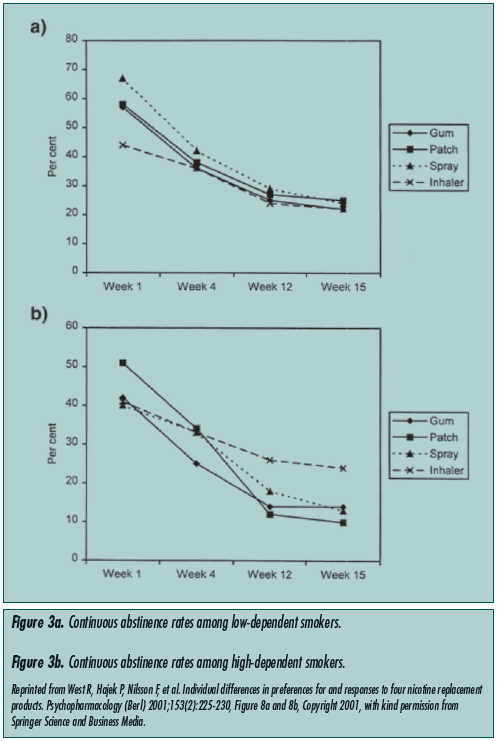
Nicotine replacement therapy. NRT is the most widely used and studied form of pharmacotherapy for managing nicotine dependence and withdrawal.11 These therapies include the transdermal patch, nasal spray, gum, lozenges, and nicotine inhalers. All have been shown to be effective in comparison with placebo.12 These studies, however, all excluded the geriatric population. The U.S. Clinical Practice Guideline endorses first-line NRTs as gum, inhaler, patch, and nasal spray, and excludes nicotine lozenges. West et al12 conducted a robust clinical trial that investigated the preferences and outcomes of four of the available NRTs, namely nicotine patch, inhaler, nasal spray, and gum. The average age of participants in this trial was 40 with a standard deviation (SD) of 10, though advanced age was not an exclusion criterion, and the minimum age for enrollment was 18. The results compared heavy versus light smokers and men versus women. While the degree of generalizability of this study to the elderly is limited, the results showed significant differences in NRT responses between high-dependence and lowdependence patients. High dependence was defined as a score of 7 or higher in the Fagerstrom Test for Nicotine Dependence. In high-dependence smokers, inhalers were found to have the highest long-term (15-week) abstinence rates, followed by gum, nasal spray, and patch. Interestingly, in low-dependence patients, the patch was found to have the highest abstinence rates, followed by spray, with gum and inhaler at approximately the same level (Figure 3). Common side effects of NRT include palpitations, insomnia, headache, and hypertension. The gum can cause mouth or dental irritation, the patch local skin irritation, and the spray nasal irritation.
Bupropion SR. Bupropion is an antidepressant unrelated to selective serotonin reuptake inhibitors or tricyclic antidepressants. This drug’s mechanism of action related to smoking cessation is currently unknown, but it is thought to be mediated by noradrenergic/dopaminergic mechanisms.11 Bupropion SR has proven to be effective in treating smoking withdrawal symptoms and nicotine dependence, doubling the rate of cessation in both men and women.13 In a clinical trial investigating predictors of successful smoking cessation with bupropion SR therapy, advanced age wasreported as one of the predictive factors for successful cessation.14 In the 150-mg per-day and 300-mg per-day dosing groups, smokers age 50 years and over had the highest abstinence rates. Interestingly, the most effective dose for smokers in this age group was only 150 mg per day, while smokers age 20-49 showed the best response with the 300-mg per-day dose (Table II). Finally, in terms of drug tolerability, bupropion SR has been used extensively as an antidepressant in the elderly, and has been found to be well tolerated in this population.15 The most commonly noted side effects associated with bupropion are agitation, dry mouth, insomnia, headache, nausea, constipation, and tremor. Rare side effects include seizures and weight loss.

Clonidine. Clonidine is an α-2 noradrenergic receptor agonist. Though evidence supports its efficacy as a smoking cessation aid, its safety in its use solely for smoking cessation has not been established in the elderly. Clonidine’s side effects include hypotension and rebound hypertension, as well as dry mouth, dizziness, constipation, and agitation. Due to the lack of studies establishing its safety and efficacy in the elderly, it is a second-line agent in this population. Clonidine is not approved by the FDA for smoking cessation.
Nortriptyline. Antidepressants, apart from bupropion, have been studied for their effect on smoking cessation, particularly in their ability to blunt nicotine withdrawal symptoms. In a study population with an average age of 47 years (±14 years) and a range of 18 to 70 years, nortriptyline was found to reduce withdrawal symptoms and improve cessation rates when compared with placebo.16 Nortriptyline in combination with transdermal nicotine was also shown to enhance the cessation rates above the rate seen with transdermal nicotine alone.17 Both trials had similar (41 ± 11) age groups16 in their study populations. No studies were found that addressed antidepressant use for smoking cessation specifically in the elderly. The most common side effects of nortriptyline include palpitations, blurred vision, urinary retention, dry mouth, constipation, weight change, and orthostatic hypotension. Rare side effects include rash, hives, seizures, and hepatitis. Nortriptyline is not approved by the FDA for smoking cessation.
Rimonabant. Rimonabant is a new promising agent for smoking cessation, although its exact mechanism is unknown. It is not yet available in the United States. It is a cannabinoid receptor type 1 blocker and has been shown to have beneficial effects on weight loss, cholesterol, and diabetes. The STudies with Rimonabant And Tobacco USe (STRATUS) trials examine its potential value in smoking cessation.18
STRATUS-US is a randomized, double-blind, placebo-controlled trial conducted in the United States that enrolled smokers who used an average of 23 cigarettes per day. Patients were randomly assigned to receive rimonabant 20 mg per day, 5 mg per day, or placebo. At the end of the study, the rate of abstinence was found to be significantly higher in the high-dose rimonabant group as compared with placebo (36.2% vs 20.6%; P < 0.001), but not in the low-dose rimonabant group (20.2%). Among subjects with prolonged abstinence, those in the placebo group gained an average of 3.7 kg of body weight, compared with 0.6 kg in the high-dose rimonabant group (P < 0.001), representing an impressive 84% reduction in weight gain for rimonabant over placebo. The STRATUS-EU (Europe) has a protocol identical to STRATUS-US, and enrolled 789 subjects in Europe; STRATUS-WW (Worldwide) is a large 1-year maintenance study that was conducted among 5055 subjects across 54 sites worldwide. Results from these latter two trials are expected within 1 year. Biographic data, including age and comorbidities, were not stated in the trial’s reports. Hence, the efficacy of this drug in the elderly smoker remains to be established, and this effort may require pooling of data from the three studies.
Varenicline. Varenicline is a recently FDA-approved acetylcholine nicotinic receptor partial agonist. It has specificity for the receptor subtype α4-β2, the receptor responsible for the reinforcing effects of nicotine.11 In a randomized, controlled trial conducted at multiple medical clinics in seven countries, varenicline was found by Tonstad et al19 to be effective when compared with placebo over 1 year. The abstinence rates compared to placebo during weeks 13 to 24 (70.5% vs 49.6%; odds ratio [OR], 2.48; 95% confidence interval [CI], 1.95-3.16; P < 0.001) and during weeks 13 to 52 (43.6% vs 36.9%; OR, 1.34; 95% CI, 1.06-1.69; P = 0.02) demonstrated that the effect of this medication did not extinguish over time. No differences in side effects were noted between the varenicline and placebo groups. Patients in this trial were of ages 18-75, with a mean age of 45.4 (±10.4), 45.3 (±10.4), and 44.2 years (±10.7) for the varenicline, placebo, and open-label varenicline groups, respectively.
Two randomized, placebo-controlled trials compared abstinence rates with use of varenicline, bupropion SR, and placebo.20,21 With strikingly similar results, these trials concluded that varenicline and bupropion SR both were more effective than placebo over 1 year. The continuous abstinence rate for weeks 9 through 24 of this 52-week trial demonstrated superiority of varenicline over bupropion SR (20.7% vs 10.5%; OR, 1.63; 95% CI, 1.14-2.33; P = 0.007). However, varenicline was no longer significant when compared with bupropion SR for weeks 9 through 52 of the study (16.1% vs 8.4%; OR, 1.46; 95% CI, 0.99-2.17; P = 0.057). The mean ages for the three intervention groups ranged from 42.0 to 44.6, with SD 11.1-11.9. For both of these studies, the age range of participants was 18 to 75, with no age-specific results reported. The most common side effects of varenicline include nausea, abnormal dreams, constipation, vomiting, sleep disturbance, and flatulence.
CONCLUSION
Assisting the elderly smoker to quit can be particularly challenging for the practicing clinician. That patient may have been smoking for many years and may seem refractory to cessation efforts. However, counseling the patient at every visit and pointing out the effects that ongoing tobacco use has already exacted may serve to motivate the patient to attempt to quit. These health-related effects might be minor in comparison to cardiovascular disease or emphysema: esophageal reflux disease, erectile dysfunction, wrinkles, osteoporosis, and rhinitis are some examples. Pharmacotherapy remains an important option and adjunct to counseling. Inferences regarding its use in the elderly can be extrapolated from data on smoking cessation trials. Nonetheless, the lack of controlled trials focusing on age as a determinant of intervention success/failure limits the utility of these inferences and demands individual clinical judgment to dictate intervention preferences among physicians today.
1. Doolan DM, Froelicher ES. Efficacy of smoking cessation intervention among special populations: Review of the literature from 2000 to 2005. Nurs Res 2006;55(4 suppl 1):S29-S37.
2. Appel DW, Aldrich TK. Smoking cessation in the elderly. Clin Geriatr Med 2003;19(1):77-100.
3. Burns DM. Cigarette smoking among the elderly: Disease consequences and the benefits of cessation. Am J Health Promot 2000; 14(6):357-361.
4. Vollset SE, Tverdal A, Gjessing HK. Smoking and deaths between 40 and 70 years of age in women and men. Ann Intern Med 2006;144(6):381-389.
5. Morgan GD, Noll EL, Orleans CT, et al. Reaching midlife and older smokers: Tailored interventions for routine medical care. Prev Med 1996;25(3):346-354.
6. U.S. Department of Health and Human Services. The Health Benefits of Smoking Cessation: A Report of the Surgeon General (DHHS). Washington, DC: U.S. Government Printing Office; 1990: Publication No (CDC) 90:8416. Available at: www.cdc.gov/tobacco/sgr/index.htm. Accessed January 4, 2007.
7. Okuyemi KS, Nollen NL, Ahluwalia JS. Interventions to facilitate smoking cessation. Am Fam Physician 2006;74:262-271.
8. Glynn TJ, Manley MW. How to Help Your Patients Stop Smoking: A National Cancer Institute Manual for Physicians. Washington, DC: USDHHS; 1989: NIH Publication No. 89:3064.
9. Brown D, Croft J, Schenck AP, et al. Inpatient smoking-cessation counseling and all-cause mortality among the elderly. Am J Prev Med 2004;26(2):112-118.
10. Fiore MC, Bailey WC, Cohen SJ, et al. Treating Tobacco Use and Dependence. Clinical Practice Guideline. Rockville, MD: US Department of Health and Human Services, Public Health Service; 2000.
11. Henningfield JE, Fant RV, Buchhalter AR, Stitzer ML. Pharmacotherapy for nicotine dependence. CA Cancer J Clin 2005;55(5):281-299.
12. West R, Hajek P, Nilsson F, et al. Individual differences in preferences for and responses to four nicotine replacement products. Psychopharmacology (Berl) 2001;153(2):225-230.
13. Scharf D, Shiffman S. Are there gender differences in smoking cessation, with and without bupropion? Pooled- and meta-analyses of clinical trials of bupropion SR. Addiction 2004;99:1462-1469.
14. Dale LC, Glover ED, Sachs DP, et al. Bupropion for smoking cessation: Predictors of successful outcome. Chest 2001;119:1357-1364.
15. DasGupta K. Treatment of depression in elderly patients: Recent advances. Arch Fam Med 1998;7(3):274-280.
16. Prochazka AV, Weaver MJ, Keller RT, et al. A randomized trial of nortriptyline for smoking cessation. Arch Intern Med 1998;158(18):2035-2039.
17. Prochazka AV, Kick S, Steinbrunn C, et al. A randomized trial of nortriptyline combined with transdermal nicotine for smoking cessation. Arch Intern Med 2004;164:2229-2233.
18. Gelfand EV, Cannon CP. Rimonabant: A cannabinoid receptor type 1 blocker for management of multiple cardiometabolic risk factors. J Am Coll Cardiol 2006;47(10):1919-1926. Epub 2006 Apr 24.
19. Tonstad S, Tonnesen P, Hajek P, et al; the Varenicline Phase 3 Study Group. Effect of maintenance therapy with varenicline on smoking cessation: A randomized controlled trial. JAMA 2006;296:64-71.
20. Jorenby DE, Hays JT, Rigotti NA, et al; the Varenicline Phase 3 Study Group. Efficacy of varenicline, an α4β2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: A randomized controlled trial. JAMA 2006;296:56-63.
21. Gonzales D, Rennard SI, Nides M, et al, the Varenicline Phase 3 Study Group. Varenicline, an α4β2 nicotinic acetylcholine receptor partial agonist, vs sustained-release bupropion and placebo for smoking cessation: A randomized controlled trial. JAMA 2006; 296:47-55.


