Scrotal Hemangioma in an Infant
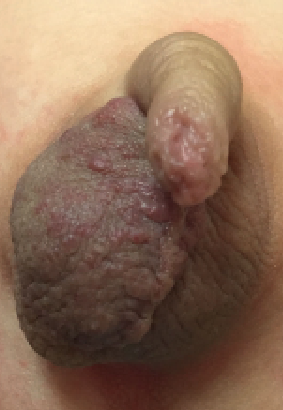
A 24-month-old boy presented to the clinic for a routine health examination. He was a singleton, born at term, and he was an appropriate weight. At birth, his physical examination results were unremarkable except for a reddish-purple hemangioma on the right scrotum (Figure). At 14 months of life, he was referred to a dermatologist, and the family was advised to watch and wait for the lesion to spontaneously resolve. However, the lesion has remained unchanged.
At the clinic, the boy underwent ultrasonography of the scrotum, which revealed a thickened, hypervascular anterior right hemiscrotal wall without extension into surrounding structures. The patient was referred to dermatology and urology for further evaluation.
DISCUSSION
Hemangiomas are proliferations of vascular endothelial cells that typically present at or soon after birth or appear via abnormal vasculogenic stimulation during the first few years of life.1 They are found in up to 10% of infants by 1 year of age, but genital hemangiomas comprise only 2% of the total.2,3
Hemangiomas arise from primitive blood vessels known as angioblasts and are hypothesized to proliferate due to in-utero hypoxia.4,5 Some propose that hypoxia causes vascular proliferation in hemangiomas in order to improve the oxygen content of hypoxic tissues.6 They are more common in infants who experience placental hypoxia, such as in cases of multiple gestations, prematurity, and low birth weight.7,8 Other factors include overexpression of vascular endothelial growth factor (VEGF), fibroblast growth factor 2, and type IV collagenase that promote angiogenesis and endothelial cell proliferation as well as imbalances in VEGF receptor tyrosine kinases.9-12 Although most hemangiomas occur sporadically, familial transmission has been reported.13 The natural history involves an early proliferation phase followed by involution that typically does not require medical intervention; 30% of hemangiomas resolve by 3 years of life, 50% by 5 years of life, and 70% to 90% by 7 years of life.2,14,15
Most hemangiomas are clinically diagnosed.16 However, imaging studies can detect underlying structural abnormalities.17 Ultrasonography results can reveal hyperechoic or hypoechoic areas, depending on the contents of the vascular abnormality.17,18 Computed tomography and magnetic resonance imaging help define the relationship of the hemangioma with adjacent anatomical structures before intervention.17 A typical radiographic finding is a soft-tissue mass containing phleboliths, suggestive of a cavernous hemangioma.17 Finally, if the anatomical abnormality is thought to be malignant, a biopsy may be warranted.19
In contrast to hemangiomas in other locations, treatment for scrotal hemangiomas may help prevent complications.2 Reported complications have included hemorrhage, rectal bleeding, or hematuria through extension of the lesion into the rectum or bladder, ulceration with subsequent infection, possible decreased fertility due to the heat generated by the hemangioma, scrotal hemorrhage after trauma, or decreased testicular size ipsilateral to the hemangioma.20 Propranolol, given orally, is the first-line treatment, with a target dose of 1 to 3 mg/kg/day, divided in 3 daily doses at least 6 hours apart.21 It causes vasoconstriction and inhibition of angiogenesis, which leads to apoptosis of the affected tissue.20 If propranolol fails, surgery with preservation of the scrotum and minimal testicular tissue removal is another option.20 Less invasive procedures such as flash dye pulsed laser, cryotherapy, or sodium chloride 15% infusion can also be useful. Because of the increased risk for complications, referral to dermatology and/or urology should be considered if spontaneous involution does not occur.
Kelly Wilmas, BS, and Hunter P. Nolen, BS, are with the John P. and Kathrine G. McGovern Medical School at University of Texas Health Science Center in Houston.
Lynnette Mazur, MD, MPH, is a professor of pediatrics at the John P. and Kathrine G. McGovern Medical School at University of Texas Health Science Center in Houston.
References
1. Avallone AN, Avallone MA, Share S, Rubin BP. Epithelioid hemangioma—a rare scrotal tumor of childhood. Urology. 2012;80(3):707-709.
2. Dinehart SM, Kincannon J, Geronemus R. Hemangiomas: evaluation and treatment. Dermatol Surg. 2001;27(45):475-485.
3. Biswamay R, Clark SS. Hemangioma of scrotum. Urology. 1976;8(5):502-505.
4. Walter JW, North PE, Waner M, et al. Somatic mutation of vascular endothelial growth factor receptors in juvenile hemangioma. Genes Chromosomes Cancer. 2002;33(3):295-303.
5. Colonna V, Resta L, Napoli A, Bonifazi E. Placental hypoxia and neonatal haemangioma: clinical and histological observations. Br J Dermatol. 2010;162(1):208-209.
6. Drolet BA, Frieden IJ. Characteristics of infantile hemangiomas as clues to pathogenesis: does hypoxia connect the dots? Arch Dermatol. 2010;146(11):1295-1299.
7. Hemangioma Investigator Group; Haggstrom AN, Drolet BA, et al. Prospective study of infantile hemangiomas: demographic, prenatal, and perinatal characteristics. J Pediatr. 2007;150(3):291-294.
8. Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170(4):907-913.
9. Takahashi K, Mulliken JB, Kozakewich HP, Rogers RA, Folkman J, Ezekowitz RA. Cellular markers that distinguish the phases of hemangioma during infancy and childhood. J Clin Invest. 1994;93(6):2357-2364.
10. Tan ST, Velickovic M, Ruger BM, Davis PF. Cellular and extracellular markers of hemangioma. Plast Reconstr Surg. 2000;106(3):529-538.
11. Jinnin M, Medici D, Park L, et al. Suppressed NFAT-dependent VEGFR1 expression and constitutive VEGFR2 signaling in infantile hemangioma. Nat Med. 2008;14(11):1236-1246.
12. Claesson-Welsh L. Healing hemangiomas. Nat Med. 2008;14(11):1147-1148.
13. Blei F, Walter J, Orlow SJ, Marchuk DA. Familial segregation of hemangiomas and vascular malformations as an autosomal dominant trait. Arch Dermatol. 1998;134(6):718-722.
14. Tonini G, Intagliata S, Cagli B, et al. Recurrent scrotal hemangiomas during treatment with sunitinib. J Clin Oncol. 2010;10;28(35):e737-e738.
15. Chang LC, Haggstrom AN, Drolet BA, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics. 2008;122(2):360-367.
16. Finn MC, Glowacki J, Mulliken JB. Congenital vascular lesions: clinical application of a new classification. J Pediatr Surg. 1983;18(6):894-900.
17. Chavan D, Javalgi AP. Scrotal hemangioma: a case report. J Clin Diagn Res. 2014;8(12):ND03-ND04.
18. Akbar SA, Sayyed TA, Jafri SZH, Hasteh F, Neill JSA. Multimodality imaging of paratesticular neoplasms and their rare mimics. RadioGraphics. 2003;23(6):1461-1476.
19. Frieden IJ, Eichenfield LF, Esterly NB, Geronemus R, Mallory SB. Guidelines of care for hemangiomas of infancy. American Academy of Dermatology Guidelines/Outcomes Committee. J Am Acad Dermatol. 1997;37(4):631-637.
20. Patoulias J, Farmakis K, Kaselas C, Patoulias D. Ulcerated scrotal hemangioma in an 18-month-old male patient: a case report and review of the literature. Case Rep Urol. 2016;2016:9236719.
21. Biesbroeck L, Brandling-Bennett, HA. Propranolol for infantile haemangiomas: review of report of a consensus conference. Arch Dis Child Educ Pract Ed. 2014;99(3):95-97.


