Paroxysmal Atrial Fibrillation Following Electronic Cigarette Use in an Elderly Woman
Since the release of the 1964 US Surgeon General’s report on the health hazards of smoking, the negative effects of tobacco use on health have been well documented. Smoking is now known to be a significant risk factor for cardiovascular, cerebrovascular, and lung diseases, as well as for a variety of malignancies.1,2 It is also the leading cause of preventable illness in the United States, responsible for more than 443,000 deaths annually.3 Smoking cessation is a huge challenge, however, for both patients and physicians. Nearly 70% of US smokers would like to quit smoking and about 40% attempt cessation in any given year,4,5 but most of these attempts are unsuccessful. Even with the use of smoking cessation interventions that have shown efficacy, complete cessation is difficult.6,7
To improve cessation rates, alternative products are being marketed. One example is the electronic cigarette (e-cigarette), a device that looks and feels like a real cigarette but does not burn tobacco. Instead, these battery-powered plastic tube devices simulate tobacco cigarettes by vaporizing nicotine, other chemicals, and flavors (eg, tobacco, chocolate, fruit, mint, coffee) so that they become inhalable. Some have a light-emitting diode, which glows to resemble the burning end of a cigarette.3,8 The e-cigarette is often marketed as a healthier option than a tobacco cigarette,3 which is a notion many users buy into.8
The sale of e-cigarettes in the United States and other countries continues to rise, despite limited information on their efficacy, safety, and side effects.8 A preliminary analysis of sample e-cigarettes by the US Food and Drug Administration (FDA) revealed toxic chemicals in several devices, the presence of nicotine in products labeled as having none, and potentially lethal levels of nicotine in refill bottles. Based on its findings, the FDA declared the devices potentially addictive.9
We present a case of e-cigarette use by an elderly hospitalized patient who developed a temporally associated tachyarrhythmia. Cigarette smoking is a known risk factor for cardiovascular and cerebrovascular disease, even at the lowest levels of exposure, and a few cases of atrial fibrillation (AF) following use of nicotine gum have been reported10,11; however, our case appears to be the first to document the development of cardiac arrhythmias in an e-cigarette user. We also provide a brief overview of the available data on e-cigarettes and explore the plausible link between e-cigarette use and arrhythmia development.

Case Report
Our patient was a 70-year-old white woman with a medical history of hypertension, hyperlipidemia, osteoarthritis, and allergic rhinitis, as well as a remote history of breast adenocarcinoma. She had an approximately 40-pack-year history of tobacco use, but had been actively trying to quit smoking over the preceding 5 to 6 months. She recently sustained a right hip fracture and had undergone total hip arthroplasty. The perioperative period was uneventful. She was discharged to continue physical therapy. Three weeks later, the patient developed an infected hematoma, which required rehospitalization for incision and drainage.
On the day of her hematoma drainage, the patient developed an asymptomatic acute episode of AF with rapid ventricular response (RVR; Figure 1). She was transferred to the intensive care unit for arrhythmia control with a diltiazem drip, which was later discontinued because of hypotension. Overnight, the patient’s AF spontaneously converted to normal sinus rhythm (Figure 2).
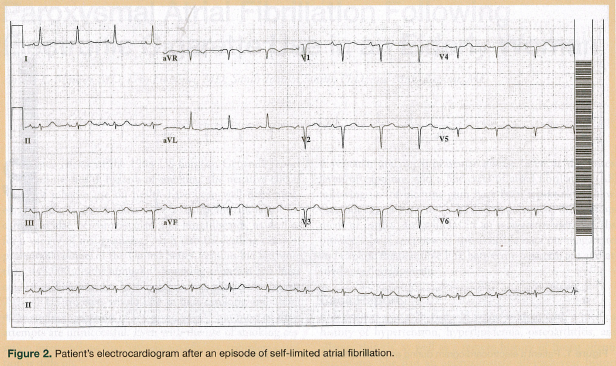
She was then transferred to the acute care for the elderly unit for further medical management, rehabilitation, and monitoring on telemetry. On postoperative day 3, she developed a second episode of AF with RVR, as reported by telemetry. Metoprolol was ordered, but the patient refused to take it because of concerns about side effects experienced during prior use. A cardiologist was again consulted. One dose of diltiazem was given and the patient again returned to normal sinus rhythm. Her hip pain was reportedly under good control.
On postoperative day 5, the patient was improving significantly with rehabilitation. The primary care team decided to continue rehabilitation at a skilled nursing facility; however, before discharge, she developed a third episode of AF with RVR reported by telemetry. The patient reported experiencing no symptoms, such as chest pain, shortness of breath, palpitations, or dizziness, during this or any of her previous episodes. Cardiac enzyme levels were evaluated to rule out acute coronary syndrome. This third episode of AF was again self-limited without requirement for acute intervention. The geriatric service was consulted for medical management of the patient’s paroxysmal AF after she refused to be transferred to the intensive care unit.
During the initial interview with the attending geriatrician, the patient reported no concerns except that she was somewhat worried about her arrhythmias. She reported normal appetite, oral intake, bowel movements, and sleep. The patient’s medications at the time were as follows: rifampin, 600 mg daily; intravenous vancomycin, 1000 mg daily; aspirin, 324 mg daily; montelukast, 10 mg daily; cetirizine, 10 mg daily; albuterol nebulizer, every 6 hours as needed; ipratropium nebulizer, every 6 hours as needed; cyclobenzaprine 5-mg tablet, three times daily as needed; hydrocodone-acetaminophen 5-325-mg tablet, one to two every 4 to 6 hours as needed; morphine injection, 2 mg every 4 hours as needed for severe pain; acetaminophen 650 mg tablet, every 6 hours as needed; multivitamin, one tablet daily; zolpidem, 5 mg nightly as needed; famotidine, 20 mg daily; ondansetron, 4 mg every 8 hours as needed; bisacodyl, 5 mg daily as needed; and diltiazem, 30 mg three times daily.
The patient’s vital signs were as follows: blood pressure, 129/88 mm Hg; pulse, 113 beats per minute; temperature, 96.1°F; respiratory rate, 16 breaths per minute; and oxygen saturation level, 93% on room air. Telemetry indicated normal sinus rhythm with rates ranging between 90 and 115 beats per minute. Her pain score was well controlled at 2 to 3 on a 10-point scale. In general, the patient was alert, oriented, and in no apparent distress. On head and neck examination, her pupils were equal, round, and reactive to light; her oropharynx was clear; and her mucous membranes were moist. Cardiovascular examination revealed regular rhythm with mild tachycardia and no murmurs. Pulmonary examination revealed clear lungs to auscultation bilaterally, and extremity examination revealed mild bilateral ankle edema with bilateral thromboembolic deterrent hose in place. The surgical wound of the right hip was clean and dry with no evidence of infection or active bleeding.
Laboratory studies disclosed the following values, with our hospital’s normal range in parenthesis: white blood cell count, 11,600/µL (normal, 4000-10,000/µL); hemoglobin, 11.9 g/dL (normal, 11.5-15.5 g/dL); hematocrit, 37.2% (34-45%); mean corpuscular volume, 94.9 µm3 (normal, 80-96 µm3); platelet count, 471 x103/ µL (normal, 150-400 x103/µL); sodium, 137 mEq/L (normal, 135-145 mEq/L); potassium, 3.9 mEq/L (normal, 3.5-5.0 mEq/L); chloride, 102 mEq/L (98-108 mEq/L); bicarbonate, 28 mEq/L (23-31 mEq/L); blood urea nitrogen, 15 mg/dL (7-23 mg/dL); glucose, 82 mg/dL (70-110 mg/dL); creatinine, 0.40 mg/dL (0.60-1.25 mg/dL); and total calcium, 9.2 mg/dL (8.6-10.6 mg/dL). The results of three sets of cardiac enzymes were negative for evidence of ischemia. Thyroid function tests were within normal limits. An electrocardiogram revealed sinus tachycardia, with no ischemic changes. A transthoracic echocardiogram showed an ejection fraction of 55% to 65% with mild diastolic dysfunction, but no valvular abnormalities or focal wall motion changes. A ventilation-perfusion scan, also known as a VQ scan, was undertaken because of the patient’s tachycardia, recent surgery, and immobility. The scan showed a low probability for pulmonary embolism.
On further questioning, the patient reported that she had been trying to quit smoking for the past 5 to 6 months by using an e-cigarette bought at a local store. After her third episode of paroxysmal AF, she recalled that her use of the e-cigarette had preceded each episode. We did not perform a challenge test for ethical reasons.
After the work-up identified no clear triggers, the patient was asked to cease all further use of her e-cigarette, and she experienced no further episodes of paroxysmal AF during the remainder of her hospitalization. Before discharge, the patient’s diltiazem dose was increased to 60 mg three times daily, and she was instructed to follow up with her primary care physician and outpatient cardiologist. She was discharged to a rehabilitation facility soon thereafter. At follow-up, the patient had no reported episodes of paroxysmal AF, and she was found to have a regular sinus rate and rhythm.
Discussion
Tobacco use remains the most preventable cause of morbidity and mortality in industrialized countries.5,12 Despite extensive documentation of its significant health risks, the prevalence of cigarette smoking remains high, and about 20% of adults older than 18 years13 and 10% of those older than 65 years are smokers. By 2050, older adults will account for about 20.2% of the US population,14 and tobacco-related diseases will likely remain the leading cause of death in the segment that smokes.
Even in the elderly, smoking cessation decreases the incidence of cardiovascular diseases, cancer, and overall morbidity and mortality, and can lead to improved quality of life.15,16 One study showed that smoking cessation after diagnosis of early-stage lung cancer improves prognosis.15 Cardiovascular risk may also decrease within the first 2 years of smoking cessation.17
Nicotine is the primary substance responsible for addiction in smokers, an effect that may be enhanced by the thousands of other chemicals in tobacco smoke.18 Another challenging obstacle to smoking cessation is the association of smoking with many everyday activities (eg, driving, drinking coffee, socializing, stress relief), and this obstacle may be the most challenging to overcome.
Interventions developed to assist with smoking cessation include nicotine replacement therapy, medications, and psychotherapy or counseling. Abstinence rates with these therapies vary significantly.6 A major impediment to quitting is the development of withdrawal symptoms, including irritability, depressed mood, anxiety, difficulty concentrating, insomnia, and tobacco cravings.18 Counseling alone may lead to response in only 10% to 20% of patients, whereas pharmacological interventions such as bupropion, varenicline, and nicotine replacement therapy (gum, patch, inhaler, nasal spray, sublingual tablet) may lead to response in 20% to 30% of patients.6,17 The Public Health Service and the US Preventive Services Task Force recommend the combined use of behavioral counseling and pharmacotherapy to help cigarette smokers quit, as this approach has shown the greatest efficacy thus far, with successful cessation in 38% of patients.6,12,19 Regardless of which approach is used, it is common for smokers to undertake many cessation attempts before achieving lifelong abstinence.18
Given the harmful effects of tobacco and the continued prevalence of its abuse, the need is urgent for more successful smoking cessation treatment options. E-cigarettes have been marketed by their manufacturers as one such option, and with their striking resemblance to real cigarettes, these devices are an attractive option for many individuals. E-cigarettes administer nicotine into lung tissue via an inhaled vapor. The small nicotine particles contained in the vapor permit rapid nicotine delivery into the bloodstream, and, depending on the nicotine concentration in the device, may quickly relieve cravings and tobacco withdrawal symptoms.1,6,8 Despite their potential of being a viable smoking cessation method, the FDA has not approved e-cigarettes for this purpose. The side effects and extent of nicotine uptake through the lungs have not been well established for these devices,1,8,20 and the FDA has noted several safety concerns regarding the use of e-cigarettes.9
The E-Cigarette Dilemma
In 2009, the FDA Center for Drug Evaluation and Research, Division of Pharmaceutical Analysis (DPA), conducted laboratory testing on samples of e-cigarettes and their components from two leading manufacturers, revealing “that quality control processes used to manufacture these products are inconsistent or non-existent.”9 Testing identified the presence of diethylene glycol, an ingredient used in antifreeze, in one cartridge; nitrosamines, which are known to be potent carcinogens in humans, in 50% of samples; and tobacco-specific impurities (TSIs), such as anabasine, which is used as an insecticide in some countries, in the majority of samples. The DPA also found that concentrations of nicotine not only varied between manufacturers, which sold cartridges containing from 0 mg to 18 mg of nicotine, but that the advertised amount did not always reflect what was actually in or vaporized by the cartridge, leading to considerable variability between cartridges with the same advertised strengths and in the amount of nicotine emitted by a single cartridge between puffs, with nicotine levels per puff ranging from 26.8 to 43.2 µg nicotine/100 mL.9 It also found that most e-cigarette cartridges labeled as containing no nicotine actually contained low levels of nicotine. Instead of purchasing new prefilled cartridges, users can elect to more cheaply refill their empty cartridges with bottled nicotine, also referred to as e-liquid, liquid nicotine, or e-cig juice. These refill bottles have been found to contain up to 1000 mg of nicotine, potentially adding health risks, as a dose of 30 to 60 mg has been reported to be fatal for adults, and it is possible to inadvertently receive this dose by spilling the nicotine on one’s skin.21
As a result of its findings, the FDA sought to classify e-cigarettes as drug delivery devices, thereby preventing their marketing in the United States until their safety and efficacy could be established in clinical trials. This classification was challenged in court, and it was ruled in 2010 that “e-cigarettes and other nicotine-containing products are not drugs or devices unless they are marketed for therapeutic purposes, but that other nicotine-containing products can be regulated as ‘tobacco products’ under the Federal Food, Drug, and Cosmetic Act.”22 As a result of this ruling, the FDA is working to develop regulations for e-cigarettes and it has continued to publicize the dangers of these devices. Although such efforts by the FDA have been supported by various medical organizations, including the American Cancer Society and the American Heart Association, they have also received criticism, including by some clinicians.
In a 2011 report published in the Harm Reduction Journal, it is noted that the FDA’s testing of the e-cigarettes and their cartridges was not performed in a scientific and systematic manner.23 The author points out several flaws: (1) the FDA only tested a few of the e-cigarette samples it had at its disposal, leading to questions as to how it decided which samples to test; (2) the 10-mg Nicotrol Inhaler cartridge, an FDA-regulated pharmaceutical nicotine product used as a control for some testing methods, was not tested for nitrosamines, despite knowledge that nicotine medications (eg, nicotine gum, patch) also contain these compounds; and (3) the actual nitrosamine levels in the e-cigarette products were not reported, but it appears the FDA’s testing method detected these compounds at concentrations about one million times lower than those known to have an ill effect on health. In the article, the author concludes “Although laboratory studies have detected trace concentrations of some contaminants, this appears to be a small problem that is amenable to improvements in quality control and manufacturing that are likely with FDA regulation as tobacco products.”
Although it is currently unlawful to market e-cigarettes as smoking cessation devices, many people use them for this purpose and some success has been reported in the literature. In 2010, a survey of 81 European and Canadian e-cigarette users revealed that 79% of these users thought that these devices had helped them quit smoking.8 The respondents also reported numerous other benefits, including less coughing, fewer breathing problems, an absence of unpleasant odors or bad breath, and the pleasurable sensation provided upon inhalation of the vapors. In 2011, Caponnetto and colleagues24 reported that use of an e-cigarette enabled three smokers with a documented history of recurring relapses to quit smoking and to remain abstinent for at least 6 months. Although some have viewed e-cigarettes as a viable harm-reduction strategy for tobacco control, significant questions over its potential benefits and risks remain, and more research is required in this area.
Our Patient’s Case
We could find no previously published data to link e-cigarette use to any particular disease process, and we can draw no direct conclusions from our patient’s case; however, the temporal relationship between her e-cigarette use and AF episodes suggests a causal relationship. Each episode occurred immediately following her use of the e-cigarette, and she had no further episodes after she ceased using it.
Aside from a temporal relationship, a plausible dose-response relationship may exist between the nicotine content in our patient’s e-cigarette and the development of her arrhythmia. The patient was using a full-strength nicotine replacement device. She also admitted to not refilling the cartridge correctly, as her refill bottle did not last as long as expected. We suspect that the patient was exposed to a high dose of nicotine via direct lung delivery and rapid tissue absorption, which may have contributed to the development of her AF.
Chronic cigarette smoking has been associated with chronic exposure to high levels of nicotine, which can have a proarrhythmic effect.2,10,11 By increasing atherosclerosis of coronary arteries or vascular spasms, cigarette smoking is well known to induce cardiac ischemia, which in turn leads to necrosis and replacement fibrosis. Nicotine itself can stimulate collagen expression in cardiac myocytes, which may further lead to fibrotic change. Nicotine’s arrhythmogenic potential has been associated with the development of AF,2 and two cases have been reported in which use of nicotine gum was linked to AF onset.10,11
Conclusion
Smoking cessation can decrease morbidity and mortality and improve quality of life, including in elders. Cessation poses a challenge for patients and their physicians, however, often requiring multiple attempts with different aids. Although e-cigarettes are an attractive cessation option because they maintain the physiopsychological aspects of smoking, more research is needed to establish their safety and appropriate use. Our case suggests a potential link between e-cigarette use and the development of a dangerous cardiovascular arrhythmia in an elderly patient. Physicians and providers should be cognizant of the growing use of e-cigarettes worldwide and encourage their patients to use psychosocial interventions and pharmacotherapies that have been rigorously tested. They should also ensure that their patients are made aware of all available resources that can help them quit smoking. Until research shows e-cigarettes to be a safe and effective smoking cessation device, caution regarding their use should be exercised, and the FDA encourages healthcare professionals and consumers to report any associated potential adverse events through the MedWatch program.25
The authors report no relevant financial relationships.
References
1. Centers for Disease Control and Prevention. Cigarette smoking among adults—United States, 2007 [published correction appears in MMWR Morb Mortal Wkly Rep. 2008;57(47):1281]. MMWR Morb Mortal Wkly Rep. 2008;57(45):1221-1226.
2. Goette A, Lendeckel U, Kuchenbecker A, et al. Cigarette smoking induces atrial fibrosis in humans via nicotine. Heart. 2007;93(9):1056-1063.
3. Kuschner WG, Reddy S, Mehrotra N, Paintal HS. Electronic cigarette and third hand tobacco smoke: two emerging health care challenges for the primary care provider.
Int J Gen Med. 2011;4:115-120.
4. Breitling LP, Rothenbacher D, Stegmaier C, Raum E, Brenner H. Older smokers’ motivation and attempts to quit smoking: epidemiological insight into the question of lifestyle versus addiction. Dtsch Arztebl Int. 2009;106(27):451-455.
5. Office on Smoking and Health, National Center for Chronic Disease Prevention and Health Promotion. 2010 Surgeon General’s Report—How tobacco smoke causes disease: the biology and behavioral basis for smoking-attributable disease. Atlanta, GA: US Department of Health and Human Services, 2010. www.cdc.gov/tobacco/data_statistics/sgr/2010/index.htm. Accessed February 7, 2012.
6. Fiore MC, Jaen CR, Baker TB, et al; Tobacco Use and Dependence Guideline Panel. Treating tobacco use and dependence: 2008 update. Rockville, MD: US Department of Health and Human Services, 2008. www.ahrq.gov/path/tobacco.htm. Accessed February 7, 2012.
7. Kralikova E, Kozak JT, Rasmussen T, Gustavsson G, Le Houezec J. Smoking cessation or reduction with nicotine replacement therapy: a placebo-controlled double blind trial with nicotine gum and inhaler. BMC Public Health. 2009;9:433.
8. Etter JF. Electronic cigarettes: a survey of users. BMC Public Health. 2010;10:231.
9. US Food and Drug Administration. Summary of results: laboratory analysis of electronic cigarettes conducted by the FDA. 2009. www.fda.gov/NewsEvents/PublicHealthFocus/ucm173146.htm. Accessed February 7, 2012.
10.Rigotti NA, Eagle KA. Atrial fibrillation while chewing nicotine gum. JAMA. 1986;255(8):1018.
11. Choragudi NL, Aronow WS, DeLuca AJ. Nicotine gum-induced atrial fibrillation. Heart Dis. 2003;5(2):100-101.
12. Ezzati M, Lopez AD, Rodgers A, et al. Selected major risk factors and global and regional burden of disease. Lancet. 2002;360(9343):1347-1360.
13. Centers for Disease Control and Prevention. Prevalence of current smoking among adults aged 18 years and over: United States, 1997-2009. www.cdc.gov/nchs/data/nhis/earlyrelease/201006_08.pdf. Accessed February 7, 2012.
14. U.S. Census Bureau. US population projections. Percent distribution of the projected population by selected age groups and sex for the United States: 2010 to 2050, based on Census 2000. Released 2008. www.census.gov/population/www/projections/summarytables.html. Accessed March 7, 2012.
15. Parsons A, Daley A, Begh R, Aveyard P. Influence of smoking cessation after diagnosis of early stage lung cancer on prognosis: systemic review of observational studies with meta-analysis. BMJ. 2010;340:b5569.
16. Cataldo JK, Malone RE. False promises: the tobacco industry, “low-tar” cigarettes, and older smokers. J Am Geriatr Soc. 2008;56(9):1716-1723.
17. Erhardt L. Cigarette smoking: an untreated risk factor for cardiovascular disease. Atherosclerosis. 2009;205(1):23-32.
18. Mannino DM. Why won’t our patients stop smoking? The power of nicotine addiction. Diabetes Care. 2009;32(suppl 2):S426-S428.
19. McClave A, Rock V, Thorne S, Malarcher M. State-specific prevalence of cigarette smoking and smokeless tobacco use among adults, United States, 2009. MMWR Morb Mortal Wkly Rep. 2010;59(43):1400-1406.
20. WHO Study Group on Tobacco Product Regulation. The scientific basis of tobacco product regulation. Geneva, Switzerland: World Health Organization, 2010. www.who.int/tobacco/global_interaction/tobreg/who_tsr.pdf. Accessed February 7, 2012.
21. International Programme on Chemical Safety (IPCS) INCHEM. Nicotine. www.inchem.org/documents/pims/chemical/nicotine.htm. Accessed March 1, 2012.
22. US Food and Drug Administration. Electronic cigarettes. www.fda.gov/newsevents/publichealthfocus/ucm172906.htm. Accessed March 1, 2012.
23. Rodu B. The scientific foundation for tobacco harm reduction, 2006-2011. Harm Reduct J. 2011;8:19.
24. Caponnetto P, Polosa R, Russo C, Leotta C, Campagna D. Successful smoking cessation with electronic cigarettes in smokers with a documented history of recurring relapses: a case series. J Med Case Reports. 2011;5(1):585.
25. US Food and Drug Administration. MedWatch: the FDA safety information and adverse event reporting program. 2012. www.fda.gov/safety/medwatch/default.htm. Accessed February 7, 2012.


