Normal Pressure Hydrocephalus in the Older Patient
Normal pressure hydrocephalus (NPH) is remarkable for the simple reason that, if it is diagnosed early, it is one of the few reversible causes of dementia. In 1965, Adams et al1 were the first to describe NPH in a small group of patients who presented with various neurological symptoms, ventricular enlargement, and what looked like normal cerebrospinal fluid (CSF) pressure during lumbar puncture; their symptoms improved after shunting. NPH subsequently became defined as a syndrome consisting of the triad of gait disturbance, urinary incontinence, and progressive dementia in a patient with radiological evidence of communicating hydrocephalus (ie, ventricles dilated out of proportion to any sulcal enlargement and brain tissue shrinkage).1-6
NPH is a relatively uncommon disorder that primarily affects persons older than 60 years of age,7 although a determination of the precise incidence of NPH in the elderly population has been difficult due to the lack of a consensus-based, widely accepted definition or standardized criteria for its diagnosis. It has been estimated to account for approximately 5% or less of all cases of dementia.8-10 The following case illustrates many of the complexities of this disorder.
Case Vignette
Mrs. J was a 70-year-old woman who presented to an outside hospital 1 day after falling and injuring her right shoulder. Her medical history was significant for hypertension, for which she had been noncompliant with medications and follow-up care. Her family reported that she had experienced a gradual decline in her cognition and ability to perform activities of daily living over a period of approximately 18 months. She had been living with her brother, who served as her caregiver. Over a period of 3 weeks before presenting to the hospital, the patient had become increasingly confused, had multiple falls, developed a new gait that was described by her family as “shuffling,” and developed new-onset urinary incontinence. No fevers, nausea, vomiting, headaches, dizziness, weakness, chest pain, or shortness of breath were reported. In addition, Mrs. J had no head trauma or history of losing consciousness during any of her falls. The patient was evaluated at the outside hospital and was found to have a fracture of the right humerus (head and neck). A computed tomography (CT) scan of the head revealed a moderate ventriculomegaly and aneurysms of the left middle cerebral artery (MCA) and left anterior cerebral artery (ACA). She was transferred to an acute hospital for further management.
At the acute hospital, Mrs. J had a Glasgow Coma Scale score of 14, a blood pressure of 200/90 mm Hg, a pulse of 87 beats per minute, and a respiratory rate of 28 breaths per minute. No orthostatic blood pressure changes were noted. The patient’s weight was 140.21 pounds and her height was 5 feet 0.6 inches. Besides the need for her to be placed in a cervical collar, her report of tenderness in the right upper extremity upon palpation, and a limited range of motion of her right arm, the remainder of her physical examination was within normal limits. Her neurological examination was initially limited because she was moderately lethargic, disoriented, and not very cooperative. Cranial nerves II through XII were grossly intact. Her brachioradialis and patellar reflexes were 3+ bilaterally, and her Achilles reflexes were 4+ on the left and 3+ on the right. Mrs. J’s gait, balance, vibration, and positional sense were difficult to assess, as was her Mini-Mental State Examination score. She had 0 out of 3 object recall at 5 minutes during the Mini-Cog Test.
On admission, the patient’s laboratory results were as follows: white blood cell count, 20.4/µL; hemoglobin, 12.3 g/dL; platelet count, 366x103/µL; sodium, 135 mEq/L; potassium, 3.6 mEq/L; blood urea nitrogen, 13 mg/dL; creatinine, 0.92 mg/dL; glucose, 198 mg/dL; total calcium, 8.3 mg/dL; ionized calcium, 1.23 mg/dL; magnesium, 2.1 mEq/L; phosphorus, 2.7 mg/dL; total protein, 5.0 g/dL; and albumin, 2.8 g/dL. A urinalysis was positive for a urinary tract infection (UTI). An admission CT angiogram of the head revealed a left MCA aneurysm measuring 1.5 x 1.5 x 1.4 cm, a distal left ACA aneurysm that was 5 mm in greatest diameter, and a prominent ventricular size. Magnetic resonance imaging (MRI) of the head revealed hydrocephalus, and confirmed the left MCA aneurysm and the left ACA aneurysm. An MRI of the cervical, thoracic, and lumbar spine revealed moderate-to-severe neural foraminal narrowing at C5/C6 and C6/C7; moderate right neural foraminal narrowing was noted at L4/L5. There was no evidence of central canal stenosis or abnormal cord signal.
Consultations were obtained on hospital days 1 and 2 from the departments of neurosurgery; orthopedics; geriatric medicine; social work; nutrition; and speech, physical, and occupational therapy. Neurosurgery confirmed that the patient had hydrocephalus and recommended ventriculoperitoneal (VP) shunt placement. Orthopedics saw the patient for her fracture of the right humerus and recommended conservative nonoperative management. The physical and occupational therapists recommended that the patient be placed in an acute rehabilitation facility upon discharge from the acute hospital. A speech therapist saw Mrs. J for possible swallowing problems, but found her to have adequate swallowing function that did not require any further intervention with respect to feeding. The geriatric medicine team saw the patient for her multiple geriatric syndromes, and assisted the trauma team in the management of these syndromes and the overall treatment of the patient.
Laboratory results from further workup as a result of the consultations were as follows: thyroid-stimulating hormone, 1.15 mIU/L; free thyroxine, 0.8 ng/dL; folate, 10.3 ng/mL; vitamin B12, 166 pg/mL; 25-hydroxyvitamin D, 15 ng/mL; prealbumin, 14 mg/dL; and total cholesterol, 182 mg/dL.
The patient had multiple medical problems, including some geriatric syndromes:
1. Falls (multifactorial etiology: UTI, vitamin D deficiency, ataxia resulting from hydrocephalus and low vitamin B12, delirium, deconditioning, urinary incontinence)
2. Urinary incontinence (multifactorial etiology: UTI, hydrocephalus, functional)
3. Probable underlying cognitive impairment (multifactorial etiology: vascular dementia from uncontrolled hypertension, vitamin B12 deficiency, hydrocephalus)
4. Acute delirium
5. Vitamin D deficiency
6. Vitamin B12 deficiency
7. Hypertension
8. Hydrocephalus
9. Ataxia
10. UTI
In addition to these multiple medical problems, Mrs. J was also found to have NPH, the topic of this article. As is the case with so many other elderly patients, multiple problems are often identified that may contribute to a specific clinical problem or may merely be coincidental findings. Only by addressing each problem individually and following the clinical outcome can one arrive at a definitive diagnosis.
Pathophysiology and Etiology of NPH
There are both primary (idiopathic) and secondary (known) causes of NPH. Secondary causes frequently occur as a complication following a head trauma, subarachnoid hemorrhage, meningitis, brain tumor, previous neurosurgical procedures, or irradiation, and have a much better prognosis than their idiopathic counterparts. Approximately one-half of NPH cases are idiopathic and one-half result from a known event.11-13
Physiologically, CSF is produced by specialized capillaries known as the choroid plexus. CSF flows from the two lateral ventricles through their respective foramina of Monro into the singular third ventricle, and from there passes through the aqueduct of Sylvius into the fourth ventricle, which is located in the posterior fossa. CSF then makes its way into the subarachnoid space through small openings in the ventricular system and covers the brain and spine within the subarachnoid space, serving as a cushion between the brain and its coverings and protecting the brain from shock. From the subarachnoid space, CSF is absorbed by arachnoid villi, or arachnoid granulations, situated near the top of the brain and eventually drains into the venous system through the superior sagittal sinus3,9 (Figure 1).

The exact pathophysiology of idiopathic NPH has been considered over several decades and remains unclear. Many theories have been postulated to explain the pathophysiology of idiopathic NPH, including: (1) scarring of the arachnoid granulations responsible for CSF absorption, leading to blockage of CSF circulation out of the ventricles or altered CSF absorption; (2) overproduction of CSF; and (3) increased transvenular resistance in the territory of the superior sagittal sinus, impeding CSF outflow into the venous system.14 The increased venous resistance is thought to be a sequela of deep white matter ischemia because patients with NPH have been found to have a higher incidence of periventricular hyperintensities (ie, small vessel ischemic changes) than age-matched control subjects.15-21 There appears to be general agreement that CSF resorption in NPH is definitely abnormal22-2; this results in the development of a pressure gradient between the fluid in the subarachnoid space and the ventricular system. This, in turn, leads to decreased CSF outflow and a higher, yet still normal, baseline pressure development within the ventricles. These intermittent high CSF pressures are referred to as B waves.8,25-28
The name normal pressure hydrocephalus is used despite the presence of B waves. While the cerebral ventricles are enlarged, the pressure, which is measured through a lumbar puncture (spinal tap), is usually found to be within normal limits. NPH is a poor name for this syndrome because it implies that the pressure is always normal, which is not the case.
It is thought that increased compliance of brain tissue with aging enables an enlargement of the ventricles to compensate for the increased yet normal baseline pressure.29 The progressive ventriculomegaly causes stretching of neighboring nerve fibers and compression of periventricular parenchyma, including arterioles and capillaries, and eventually leads to ischemia.
Clinical Features of NPH
The clinical manifestations of NPH can be described on the basis of both mechanical and ischemic factors, and they relate to which structures or parts of the brain are compressed or affected by the enlarging ventricles. For instance, pressure on the frontal lobes and their interconnections have been associated with dementia and cognitive impairment; pressure on the cortical center responsible for bladder and bowel control in the paracentral lobes may lead to incontinence; and pressure on the corticospinal tract, whose fibers supply motor function to the legs, may lead to gait disturbance.17-19,30 For these reasons, NPH has been associated with the aforementioned triad symptom complex of gait abnormality, dementia, and urinary incontinence.1-6
Gait abnormality is usually the most predominant symptom and the first symptom to manifest in patients with NPH. It is also the first symptom to improve after VP shunting. This is because the fibers of the corticospinal tract, which, as stated previously, supply motor function to the legs, pass closest to the lateral ventricles in the corona radiata.31 The gait disturbance can be described as a shuffling, a magnetic or broad-based type of gait, or a combination of these. Steps appear slower and shortened in the shuffling gait, feet appear “glued to the floor” with decreased step height in the magnetic gait, and there is a wide-based stance in the broad-based gait.1,32 The wide stance is usually a result of trying to compensate for a feeling of imbalance when walking. Freezing during walking and difficulty with turning or initiating walking are suggestive of disease progression. Frequent falls requiring the use of an assistive device such as a cane or a walker become a common result of these gait abnormalities. The gait disturbance is also referred to as gait apraxia because it appears as if the person has forgotten how to walk and has difficulty with basic components of walking, such as heel strike, mid-stance, and swing phase.
The short stride, slow gait, and difficulty with turning present challenging similarities between the gait of NPH and that of other conditions such as Parkinson’s disease and cerebellar ataxia. Hints to help identify NPH include the absence of extrapyramidal signs such as cogwheel rigidity, no resting tremor, and no response to levodopa. Unlike patients with NPH, patients with Parkinson’s disease respond to visual and acoustic cues.32 Cerebellar ataxia has other features such as dysarthria, gaze-evoked nystagmus, and appendicular dysmetria, none of which are present in NPH.
The urinary disorder associated with NPH begins with urinary frequency or urgency and then progresses to complete loss of bladder control (incontinence) in the later stages of the disease. The initial presentation is thought to be a result of the involvement of the sacral fibers of the corticospinal tract and the later features are attributed to the development of dementia.3 There is also the possibility of functional incontinence in persons with NPH as a result of the slow gait delaying the time for reaching the bathroom. Other causes of urinary frequency, urgency, and incontinence such as enlarged prostate and UTI will need to be ruled out so that one can be certain that these symptoms are purely attributed to NPH.
Cognitive impairment and dementia of NPH have been associated with pressure on the frontal lobes and their interconnections, including structures such as the limbic system, by the enlarging ventricles. The deficits are subcortical in origin and include problems with attention and concentration, latency in recall, and apathy, which presents as an absence of spontaneity and initiative (also referred to as abulic traits). The lack of cortical features such as apraxia (difficulty performing simple tasks or movements), agnosia (inability to interpret stimuli), and aphasia (word-finding difficulty) in NPH help differentiate the dementia of NPH from that of Alzheimer’s disease.33 Behavioral disturbances such as agitation and depression, as well as hypersomnia, may also be noted in a person with NPH.
Differential Diagnosis of NPH
There are many other disease processes that can manifest similar clinical characteristics as NPH, either by themselves or in combination. These include Alzheimer’s disease, Parkinson’s disease, dementia with Lewy bodies, multi-infarct (vascular) dementia, frontotemporal dementia, chronic alcoholism, carcinomatous meningitis, subdural hematoma, intracranial infections and tumors, and endocrine disorders such as hypothyroidism and Addison’s disease.
There is the possibility of multiple pathologies coexisting and presenting with features that are suggestive of NPH. For instance, gait-associated conditions such as cervical stenosis and spinal stenosis, cognition-associated conditions such as vascular dementia and subdural hematoma, and abnormal urination–associated conditions such as benign prostatic hypertrophy may be present at the same time in a patient and will manifest similar to NPH.
Diagnosis of NPH
Diagnosing NPH is not always straightforward, and as a result, NPH is often misdiagnosed. Diagnosis usually involves combining a patient’s clinical history (signs and symptoms), physical examination findings, and various imaging and CSF studies. It is important to note that the diagnostic sensitivity for NPH is reduced by the variability that occurs in its clinical presentation and course. In addition, the diagnostic specificity decreases as a result of the common occurrence of other conditions in the elderly who have symptoms that closely resemble those of NPH. Since the history and physical examination findings of NPH have already been discussed, we will now examine some of the imaging and CSF evaluation findings associated with NPH.
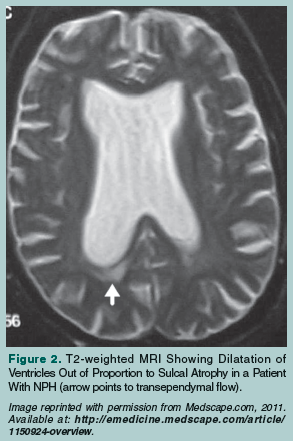 Features on CT scans that characterize NPH are the presence of bigger temporal horns, ventriculomegaly that is out of proportion to sulcal enlargement, and brain tissue shrinkage. Findings on MRI that are suggestive of NPH are transependymal resorption (Figure 2),34 T2-weighted increased intensities in brain parenchyma next to the ventricles, and the preservation of hippocampal tissue. (The presence of hippocampal tissue loss is associated with Alzheimer’s disease.) Other MRI findings that are suggestive of NPH are a higher level of CSF flow through the sylvian aqueduct and a forceful nature of CSF flow through the foramina and channels within the cerebrum.3,8,14,35-37
Features on CT scans that characterize NPH are the presence of bigger temporal horns, ventriculomegaly that is out of proportion to sulcal enlargement, and brain tissue shrinkage. Findings on MRI that are suggestive of NPH are transependymal resorption (Figure 2),34 T2-weighted increased intensities in brain parenchyma next to the ventricles, and the preservation of hippocampal tissue. (The presence of hippocampal tissue loss is associated with Alzheimer’s disease.) Other MRI findings that are suggestive of NPH are a higher level of CSF flow through the sylvian aqueduct and a forceful nature of CSF flow through the foramina and channels within the cerebrum.3,8,14,35-37
Cisternography using a radionuclide isotope introduced into the subarachnoid space via a lumbar puncture (spinal tap) has been one of the most used tests for diagnosing NPH.29 The test detects early and continual reflux of the radionuclide material within the ventricles and the reflux’s slow clearance, suggesting increased resistance to CSF absorption via the arachnoid granulations at the convexity, as seen in NPH. However, it is important to remember that a normal cisternogram does not rule out NPH.3 Also, a positive cisternogram together with a nuclear or xenon-enhanced CT scan showing normal cerebral blood flow is a better predictor of positive shunt response than cisternography alone.38
There are six CSF-related tests that can be done to help ascertain the presence of NPH and also to help determine the likely response to CSF shunting. These include standard spinal tap, large-volume spinal tap, temporary external lumbar drainage, extended intracranial monitoring, CSF outflow assessment, and measurement of aqueductal CSF flow.
The standard spinal tap test involves collecting approximately 20 to 50 mL of CSF, measuring the opening pressure, and conducting CSF fluid analysis. The presence of a normal white blood cell count, CSF protein level, and glucose level, as well as an opening pressure within the range of 60 to 240 mm of water, is highly suggestive of NPH. The patient may also show signs of improvement of symptoms—especially gait—within 1 to 2 days after the standard spinal tap, and then worsen to almost the predrainage symptom severity by 1 to 2 weeks after the standard spinal tap.29
Large-volume spinal tap involves collecting approximately 60 mL of CSF and comparing the patient’s symptoms before and after fluid drainage. A positive symptom response to the drainage is suggestive of a likelihood of obtaining good results if the patient undergoes shunting.39
Temporary external lumbar drainage involves 3 to 5 days of CSF drainage through intermittent or constant pump-controlled removal of CSF, combined with close clinical monitoring of the patient. Each day, approximately 100 to 150 mL of CSF is removed, and the patient’s temperature and pulse rate are measured. Daily laboratory and microbiological controls of CSF samples are done to detect preclinical evidence of intrathecal bacterial contamination. Temporary external lumbar drainage requires hospitalization, and has been shown to have a more accurate indication of response to shunting than the other tests because it closely mimics shunting.40-42
Extended intracranial pressure monitoring is performed with the aim of identifying the intermittent waves of high pressure (B waves), which are thought to characterize NPH. The presence of B waves during at least 50% of the intracranial pressure recording time has been reported to be predictive of a good response to shunting.8
CSF outflow assessment can also be performed during lumbar puncture via introducing a sterile fluid while measuring and monitoring intracranial pressure during the introduction of the fluid and the release of CSF. It is thought that an increased resistance to outflow is indicative of a good response to shunting,35,37,43 but a review of several studies of NPH cohorts showed inconsistent response.44
Increased aqueductal CSF flow, which can be determined indirectly by measuring aqueductal CSF stroke volume using phase-contrast MRI, has been shown to be associated with a favorable response to shunting.35,37,43,45
Treatment of NPH
VP shunting involves placing a catheter into one of the lateral ventricles, attaching a cap and valve, and tunneling tubing subcutaneously from the valve into the peritoneal cavity of the abdomen. It has been found to be the most effective available treatment for NPH. It is thought that shunting helps improve symptoms by relieving the pressure off of the parenchymal absorption pathway at the areas of CSF production within the ventricles, reducing interstitial swelling and interstitial pressure, which, in turn, improves perfusion and reduces ischemia.46
As previously discussed, several indicators of good shunting outcome include early diagnosis, when gait is the predominant presentation, when there is a known cause of the hydrocephalus, the presence of mild or lack of white matter disease, positive symptom response to large-volume CSF drainage, presence of B waves during more than 50% of continuous intracranial monitoring, CSF outflow resistance more than 18 mm Hg/mL per minute, and increased aqueductal CSF flow. Patients with significant cognitive impairment, severe white matter disease, or severe atrophy on imaging are less likely to have a good response to shunting.8 The mean positive response to shunting has been estimated to be between 30% and 40% for idiopathic NPH and 50% and 70% for NPH with a known cause.8-10 Shunting-related complications include subdural and intracerebral hematomas, seizures, shunt malfunctioning, anesthesia-associated complications, severe residual deficit, and death. Statistically, the rate of complications ranges from 20% to 40%.8,9
Progress Report of the Case Patient
The case patient had exhibited multiple medical problems, including falls, urinary incontinence, cognitive impairment, acute delirium, vitamin D deficiency, vitamin B12 deficiency, hypertension, hydrocephalus, ataxia, and UTI. She received a multidisciplinary assessment and management, and received appropriate treatment for all of the above-mentioned problems, including VP shunting performed by neurosurgery on hospital day 3. Mrs. J was discharged to a rehabilitation facility 5 days after VP shunt placement, with a significant improvement in her gait and cognition. She no longer had urinary incontinence and was no longer confused; she was oriented to time, place, person, and self. She was back to her reported baseline, was eating, and was working with physical and occupational therapists while in the rehabilitation facility. Although NPH was present in this patient and she had all of the symptoms of the triad of NPH, her cognitive decline, lethargy, incontinence, and even her gait disturbance could have been at least partially attributed to her other problems, such as her UTI. She had her VP shunt placement at the same time that she was treated for her UTI, making it difficult to determine which of these problems was the major contributor to her presentation. Nevertheless, the cause of the patient’s change in cognitive function on presentation was likely multifactorial, and her improvement was a result of addressing all of the underlying contributing factors.
Conclusion
NPH is a relatively uncommon condition that is found in the elderly population. It is characterized by a common clinical triad of gait disturbance, urinary incontinence, and dementia. There is a need for additional research and understanding of the pathophysiology of the disease to enhance early diagnosis, better patient selection of persons who will benefit from VP shunt placement, and treatment.
The author reports no relevant financial relationships.
Dr. Osei-Boamah is Assistant Professor of Medicine, Division of Gerontology & Geriatric Medicine, University of Maryland School of Medicine, Baltimore.
References
1. Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH. Symptomatic occult hydrocephalus with “normal” cerebrospinal pressure. A treatable syndrome. N Engl J Med. 1965;273(3):117-126.
2. McHugh PR. Occult hydrocephalus. Q J Med. 1964;33(2):297-308.
3. Gleason PL, Black PM, Matsumae M. The neurobiology of normal pressure hydrocephalus. Neurosurg Clin N Am. 1993;4(4):667-675.
4. Hakim CA, Hakim R, Hakim S. Normal-pressure hydrocephalus. Neurosurg Clin N Am. 2001;12(4):761-773, ix.
5. Krauss JK, Faist M, Schubert M, Borremans JJ, Lücking CH, Berger W. Evaluation of gait in normal pressure hydrocephalus before and after shunting. Adv Neurol. 2001;87:301-310.
6. Stolze H, Kuhtz-Buschbeck JP, Drücke H, et al. Gait analysis in idiopathic normal pressure hydrocephalus—which parameters respond to the CSF tap test? Clin Neurophysiol. 2000;111(9):1678-1686.
7. Woodworth GF, McGirt MJ, Williams MA, Rigamonti D. Cerebrospinal fluid drainage and dynamics in the diagnosis of normal pressure hydrocephalus. Neurosurgery. 2009;64(5):919-925.
8. Vanneste JA. Diagnosis and management of normal-pressure hydrocephalus. J Neurol. 2000;247(1):5-14.
9. Vanneste JA, Augustijn P, Dirven C, Tan WF, Goedhart ZD. Shunting normal-pressure hydrocephalus: do the benefits outweigh the risks? A multicenter study and literature review. Neurology. 1992;42(1):54-59.
10. Vanneste J, Augustijn P, Tan WF, Dirven C. Shunting normal pressure hydrocephalus: the predictive value of combined clinical and CT data. J Neurol Neurosurg Psychiatry. 1993;56(3):251-256.
11. Corkill RG, Cadoux-Hudson TA. Normal pressure hydrocephalus: developments in determining surgical prognosis. Curr Opin Neurol. 1999;12(6):671-677.
12. Ojemann RG, Fisher CM, Adams RD, Sweet WH, New PF. Further experience with the syndrome of “normal” pressure hydrocephalus. J Neurosurg. 1969;31(3):279-294.
13. Huckman MS. Normal pressure hydrocephalus: evaluation of diagnostic and prognostic tests. AJNR Am J Neuroradiol. 1981;2(5):385-395.
14. Bradley WG. Normal pressure hydrocephalus: new concepts on etiology and diagnosis. AJNR Am J Neuroradiol. 2000;21(9):1586-1590.
15. Koto A, Rosenberg G, Zingesser LH, Horoupian D, Katzman R. Syndrome of normal pressure hydrocephalus: possible relation to hypertensive and arteriosclerotic vasculopathy. J Neurol Neurosurg Psychiatry. 1977;40(1):73-79.
16. Graff-Radford NR, Godersky JC. Idiopathic normal pressure hydrocephalus and systemic hypertension. Neurology. 1987;37(5):868-871.
17. Casmiro M, D’Alessandro R, Cacciatore FM, Daidone R, Calbucci F, Lugaresi E. Risk factors for the syndrome of ventricular enlargement with gait apraxia (idiopathic normal pressure hydrocephalus): a case-control study. J Neurol Neurosurg Psychiatry. 1989;52(7):847-852.
18. Bradley WG Jr, Whittemore AR, Watanabe AS, Davis SJ, Teresi LM, Homyak M. Association of deep white matter infarction with chronic communicating hydrocephalus: implications regarding the possible origin of normal pressure hydrocephalus. AJNR Am J Neuroradiol. 1991;12(1):31-39.
19. Krauss JK, Regel JP, Vach W, Droste DW, Borremans JJ, Mergner T. Vascular risk factors and arteriosclerotic disease in idiopathic normal pressure hydrocephalus of the elderly. Stroke. 1996;27(1):24-29.
20. Krauss JK, Regel JP, Vach W, et al. White matter lesions in patients with idiopathic normal pressure hydrocephalus and in an age-matched control group: a comparative study. Neurosurgery. 1997;40(3):491-496.
21. Krauss JK, Droste DW, Vach W, et al. Cerebrospinal fluid shunting in idiopathic normal-pressure hydrocephalus of the elderly: effect of periventricular and deep white matter lesions. Neurosurgery. 1996;39(2):292-300.
22. Boon AJ, Tans JT, Delwel EJ, et al. Does CSF outflow resistance predict the response to shunting in patients with normal pressure hydrocephalus? Acta Neurochir Suppl. 1998;71(1):331-333.
23. Castro ME, Portnoy HD, Maesaka J. Elevated cortical venous pressure in hydrocephalus. Neurosurgery. 1991;29(2):232-238.
24. Greitz D, Greitz T, Hindmarsh T. A new view on the CSF-circulation with the potential for pharmacological treatment of childhood hydrocephalus. Acta Paediatr. 1997;86(2):125-132.
25. Savolainen S, Hurskainen H, Paljärvi L, Alafuzoff I, Vapalahti M. Five-year outcome of normal pressure hydrocephalus with or without a shunt: predictive value of the clinical signs, neuropsychological evaluation and infusion test. Acta Neurochir (Wien). 2002;144(6):515-523.
26. Pisani R, Mazzone P, Cocito L. Continuous lumbar cerebrospinal fluid pressure monitoring in idiopathic normal-pressure hydrocephalus: predictive value in the selection for shunt surgery. Clin Neurol Neurosurg. 1998;100(2):160-162.
27. Symon L, Dorsch NW, Stephens RJ. Pressure waves in so-called low-pressure hydrocephalus. Lancet. 1972;2(7790):1291-1292.
28. Symon L, Dorsch NW. Use of long-term intracranial pressure measurement to assess hydrocephalic patients prior to shunt surgery. J Neurosurg. 1975;42(3):258-273.
29. Bejjani GK, Hammer MD. Normal-pressure hydrocephalus: another treatable dementia, part I. Contemporary Neurosurgery. 2005;27(16):1-4.
30. Hakim S, Vengas JG, Burton JD. The physics of the cranial cavity, hydrocephalus and normal pressure hydrocephalus: mechanical interpretation and mathematical model. Surg Neurol. 1970;5(3):187-210.
Arch Neurol. 1986;43(9):940-942.
32. Stolze H, Kuhtz-Buschbeck JP, Drücke H, Jöhnk K, Illert M, Deuschl G. Comparative analysis of the gait disorder of normal pressure hydrocephalus and Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2001;70(3):289-297.
33. Bret P, Guyotat J, Chazal J. Is normal pressure hydrocephalus a valid concept in 2002? A reappraisal in five questions and proposal for a new designation of the syndrome as “chronic hydrocephalus.” J Neurol Neurosurg Psychiatry. 2002;73(1):9-12.
34. Dalvi A, Premkumar A. Normal pressure hydrocephalus. Medscape Reference Journal [serial online]. 2011. http://emedicine.medscape.com/article/1150924-overview. 35. Bradley WG Jr, Scalzo D, Queralt J, Nitz WN, Atkinson DJ, Wong P. Normal-pressure hydrocephalus: evaluation with cerebrospinal fluid flow measurements at MR imaging. Radiology. 1996;198(2):523-529.
36. Hurley RA, Bradley WG Jr, Latifi HT, Taber KH. Normal pressure hydrocephalus: significance of MRI in a potentially treatable dementia. J Neuropsychiatry Clin Neurosci. 1999;11(3):297-300.
37. Mase M, Yamada K, Banno T, Miyachi T, Ohara S, Matsumoto T. Quantitative analysis of CSF flow dynamics using MRI in normal pressure hydrocephalus. Acta Neurochir Suppl. 1998;71:350-353.
38. Chang CC, Kuwana N, Ito S, Ikegami T. Prediction of effectiveness of shunting in patients with normal pressure hydrocephalus by cerebral blood flow measurement and computed tomography cisternography. Neurol Med Chir (Tokyo). 1999;39(12):841-846.
39. Mori K. Management of idiopathic normal-pressure hydrocephalus: a multiinstitutional study conducted in Japan. J Neurosurg. 2001;95(6):970-973.
40. Chen IH, Huang CI, Liu HC, Chen KK. Effectiveness of shunting in patients with normal pressure hydrocephalus predicted by temporary, controlled-resistance, continuous lumbar drainage: a pilot study. J Neurol Neurosurg Psychiatry. 1994;57(11):1430-1432.
41. Haan J, Thomeer RT. Predictive value of temporary external lumbar drainage in normal pressure hydrocephalus. Neurosurgery. 1988;22(2):388-391.
42. Marmarou A, Young HF, Aygok GA, et al. Diagnosis and management of idiopathic normal-pressure hydrocephalus: a prospective study in 151 patients. J Neurosurg. 2005;102(6):987-997.
43. Kim DS, Choi JU, Huh R, Yun PH, Kim DI. Quantitative assessment of cerebrospinal fluid hydrodynamics using a phase-contrast cine MR image in hydrocephalus. Childs Nerv Syst. 1999;15(9):461-467.
44. Hebb AO, Cusimano MD. Idiopathic normal pressure hydrocephalus: a systematic review of diagnosis and outcome. Neurosurgery. 2001;49(5):1166-1186. 45. Egeler-Peerdeman SM, Barkhof F, Walchenbach R, Valk J. Cine phase-contrast MR imaging in normal pressure hydrocephalus patients: relation to surgical outcome. Acta Neurochir Suppl. 1998;71:340-342.
46. Mitchell P, Mathew B. Third ventriculostomy in normal pressure hydrocephalus. Brit J Neurosurg. 1999;13:382-385.


