“Male Menopause”: Just the Facts
The sole purpose of sexual activity in virtually all species is reproduction. Although sexual activity has, at least until recently, been a necessary prerequisite for the maintenance of the human species, humans have also practiced it for nonreproductive purposes for thousands of years. This partial dissociation of reproduction from sexual functions is more evident among older men. Most men have fulfilled their reproductive role by age 50, but it is abundantly evident that the vast majority of men ≥50 years of age wish to continue functioning sexually. It is clear that sexual activity in humans serves personal and societal needs for stability by promoting pair-bonding and stable family formation.1
Sexual functioning has been associated with the gonads since antiquity. In Historia Animalium, Aristotle observed, “If castration occurred in roosters after their growth was completed...the comb would turn yellow, they would cease to crow and no longer desire sexual intercourse.”2 For centuries, people believed that gonadal failure was responsible for many of the symptoms of old age. In 1889, renowned French physiologist Brown-Séquard prepared a dog testicle extract and injected it into himself.3 He reported that the injections improved his vigor and capacity for work. We now know that these aqueous extracts contained minimal quantities of testosterone, a steroid that is not soluble in water. Today, although it is clear that the aging process cannot be attributed to a simple lack of sex steroids, the general public perceives impotence or reduced libido in men as a definite sign of aging, regardless of whether the problem is due to hormonal changes.4
We call the end of the reproductive phase of life in women menopause, a term derived from the Greek words menas (month) and pausis (cessation). It is defined as the cessation of menstrual bleeding, and the clinical diagnosis can be made retrospectively, following at least 1 year of amenorrhea.
In the vast majority of women, menopause occurs between 50 and 53 years of age as a result of a critical decline in the oocyte pool that leads to a 10- to 20-fold rise in levels of serum follicle-stimulating hormone (FSH) and a 3-fold increase in luteinizing hormone (LH) levels. Serum estrogen (estradiol) levels drop until nearly undetectable, and progesterone production permanently ceases. In most women, these hormonal changes are associated with a range of clinical manifestations. Immediate consequences include hot flashes, insomnia, anxiety, irritability, poor concentration, mood fluctuations, and, less frequently, reduction in sexual activity and libido. Intermediate symptoms develop several months later and include dyspareunia (painful intercourse); vaginal dryness; and urinary problems, such as dysuria and urgent or frequent urination. Long-term clinical complications associated with estrogen deprivation include osteoporosis, increased risk of myocardial infarction and stroke, and possibly accelerated development of various forms of dementia.
Some of these problems can also be detected in aging men, and many clinical scientists have proposed the existence of a “male menopause” syndrome. One of the more common proposed definitions of male menopause is the constellation of symptoms, both physical and emotional, experienced by men of advancing age that can be attributed to endocrine changes in the body; namely, the gradual decline of serum testosterone levels and/or the relative target tissue/organ insensitivity to testosterone and its metabolites.5
In contrast to the clear, concise definition of menopause in women, the proposed definitions for male menopause invoke subjective judgments (eg, What is the exact constellation of symptoms required to make the diagnosis? Are all symptoms of equal importance? Is there a minimum number or combination of symptoms required?) and largely unmeasurable or undefined parameters (eg, What are the critical diagnostic levels of testosterone? How do we estimate target tissue insensitivity? What tissues/organs are we talking about?). Furthermore, there is not even consensus on the terminology to use in describing this event. In addition to the term male menopause, the following terms have been proposed:
• PADAM (Partial Androgen Deficiency in the Aging Male)
• ADAM (Androgen Decline in Aging Males)
• Male climacteric
• Viropause
• Relative hypogonadism
• Hypoandrogenemia
• “Manopause”
• Andropause
All of these terms are currently used in the literature to describe what is essentially the same phenomenon or combination of symptoms and laboratory findings.
Effects of Aging on Male Sexual Activity
The fact that libido and sexual performance may decline with advancing age is widely accepted and has been at least since the golden era of Athens, nearly 25 centuries ago; Aristophanes’ comedies include several pertinent remarks. On the other hand, the Bible contains multiple passages with reports of patriarchs fathering children at a very old age.6
The first scientifically documented quantitative data, however, appeared in a report by Kinsey and colleagues7 in 1948. They measured sexual activity in men according to the number of ejaculations over a defined period of time. Data showed that sexual activity peaks when men are in their mid- to late teens and starts declining when they are in their late 20s or early 30s, becoming almost nil by the time they are in their late 80s and 90s. More recently, in follow-up studies, Martin8,9 confirmed a steady decrease in the frequency of orgasmic events starting when men are in their early 30s. In addition, he found that while erectile dysfunction (impotence) was rare among men in their 20s and 30s, it gradually increased, with a prevalence of 8% in men 55 years of age, 20% in men 65 years of age, and 40% in men 75 years of age. It is important to note, however, that in these studies the majority of men, even those who were 75 years of age, were able to achieve erections and orgasms. Many subsequent studies have reported similar findings.10,11
With normal aging, the general character of the sexual response cycle, as defined by Masters and Johnson,12,13 is maintained, although there are measurable changes when compared with the norms for younger men. The earliest age-related change that men experience in sexual function is a decline in their capacity for repeated orgasmic events within a short period of time. For men, multiorgasmic capacity peaks in the middle teens and declines steadily thereafter. Masters and Johnson attributed this phenomenon to prolongation of the refractory period after ejaculation with advancing age. In contrast, most women maintain their multiorgasmic capacity well into their late 30s to early 40s. Masters and Johnson also identified an age-related increase in the stimulation time required for men to achieve a full erection. In addition, they reported that vasomotor responses are attenuated and detumescence following orgasm is more rapid with advancing age. Nocturnal penile tumescence shows a decline in total duration among men in their 20s and early 30s, with additional, smaller declines occurring steadily in later years.12,13 From a clinical perspective, it is very important to note that psychological and sociological factors, as well as the general health status of the patient and his partner, have been found to be among the strongest determinants of sexual performance for older men.6,8
Hormonal Changes in the Aging Man
The two main functions of the testicles are spermatogenesis, which involves the seminiferous tubules, and the secretion of testosterone, which is produced by the Leydig cells. Spermatogenesis is under the control of testosterone and the anterior pituitary hormone FSH. Testosterone production is under the control of the pituitary hormone LH, which in turn is regulated by the hypothalamic hormone luteinizing hormone–releasing hormone (LHRH). LHRH secretion is regulated in part by testosterone through a negative feedback mechanism.14 LHRH secretion is also modulated by many other hormones and neurotransmitters, such as estrogens, prolactin, norepinephrine, dopamine, beta-endorphins, and enkephalins.6
Circulating testosterone is partially tightly bound to the testosterone-binding globulin (TeBG). The testosterone that is available to the cells and tissues of the entire body (also known as bioavailable testosterone) is the non–TeBG-bound testosterone, which consists of the free and albumin-bound fractions.15,16
In men, a clear circadian rhythm of serum total and bioavailable testosterone levels has been documented, with evidence of higher levels in the morning. This indicates that in clinical practice, testosterone should be measured at approximately the same time each day, preferably before 9:00 am.
We will now review the effects of aging on the male hormonal milieu, sexual behavior, and other suspected results of androgen deficiency. The strength of the evidence for these assumptions will also be discussed.
Testosterone Changes With Age
The earliest reports on the relationship between advancing age and testosterone levels appeared in 1948 and 1958, issued by investigators who used various bioassay techniques to identify an age-associated decline in circulating testosterone levels.17,18 In the years after the radioimmunoassay for measuring testosterone was introduced in the late 1960s, a large number of studies were conducted, reporting results that were conflicting and confusing. Some early studies of outpatient clinic patients found a substantial age-related decline in testosterone levels.19 Shortly thereafter, studies conducted in subgroups of the Baltimore Longitudinal Study of Aging20 and the Boston Veterans Affairs NAS (Normative Aging Study)21 that closely screened participants for the absence of acute or chronic disease did not detect any significant decrease in total and/or bioavailable levels of testosterone associated with advancing age. Several other investigators in Europe and Asia reported similar findings.22 The majority of studies, however, including an analysis of data for the entire male population of the Baltimore Longitudinal Study of Aging,23 have documented significant age-related reductions in serum total, free, and/or bioavailable testosterone levels.19,24-26
The inconsistencies among the results reported by the various studies are probably due to differences in characteristics of the study subjects, such as variation in socioeconomic background, health status, testosterone binding and/or metabolism, and habits and medication use that might affect testosterone production.27-30 Serum testosterone levels are affected by smoking, which some studies associate with higher testosterone levels, and ethanol use, which almost all studies associate with lower testosterone levels. Chronic calorie restriction leads to lower serum testosterone, but obese men have also been found to have decreased total and free levels of testosterone. Sleep apnea causes a decrease in total and free testosterone levels that is independent of age and obesity.
Normal aging is characterized by a rise in TeBG and, according to most studies, a decline in the level of non–TeBG-bound (bioavailable) testosterone. It remains unclear whether this decrease is solely attributable to the effects of aging or is at least partially due to age-related changes in lifestyle, diet, and/or chronic physical activity limitations, all of which could lead to significant changes in body composition.23,26
Overall, it appears that there is a moderately strong negative correlation between testosterone and age, with a somewhat stronger negative correlation between bioavailable testosterone and age. The variation in serum testosterone levels among older men is very high, and some individuals in their 70s and 80s have values in the high normal range for young adult men.31
Pituitary Hormone Changes With Age
The normal response of the pituitary gland to decreasing circulating testosterone levels and failing spermatogenesis would be to increase the secretion of LH and FSH. Indeed, most published studies have reported increased LH and FSH levels in older men.20,32-34 On the other hand, a significant number of studies have not found elevated LH and/or FSH levels in this population.35-37
The finding of unaltered or decreasing serum testosterone levels corresponding with increasing LH levels is compatible with the development of an age-related testicular failure related to the Leydig cells that is comparable to the ovarian failure seen in menopausal women. Finding normal or low LH levels, however, points to a hypothalamic/pituitary failure. We cannot be sure yet whether these aging men are experiencing a testicular gland failure, a pituitary gland failure, or some degree of failure in both glands.
Testosterone and Sexual Activity
 Although many studies have reported decreased sexual activity with advancing age and many others have documented some decline in serum testosterone levels in older men, the association between these two variables in the same population has not been studied adequately.
Although many studies have reported decreased sexual activity with advancing age and many others have documented some decline in serum testosterone levels in older men, the association between these two variables in the same population has not been studied adequately.
The possibility that these are associated was first studied in the early 1980s in a population of 183 healthy married men, ages 60 to 80 years, enrolled in the Baltimore Longitudinal Study of Aging.31 A comparison of testosterone levels with age-adjusted sexual activity levels—defined as orgasmic events per month—suggested that older men with high testosterone levels were more likely to exhibit higher levels of sexual activity when compared with men who had low testosterone levels. Although the finding was statistically significant, there was tremendous overlap of serum testosterone values among the groups. These data, which are frequently misquoted by advocates of “testosterone replacement,” do not justify the assertion that decreased testosterone levels cause diminished sexual activity in older men. In fact, only a small fraction of older men in this study population who reported low levels of sexual activity had an abnormally low testosterone level. The major finding of this study was that the association between testosterone level and sexual activity accounted for a relatively modest fraction of the variance; a much stronger association had been expected based on previous assumptions. A study by Davidson et al38 reported similar findings. The investigators found a somewhat stronger association between testosterone level and libido, however.
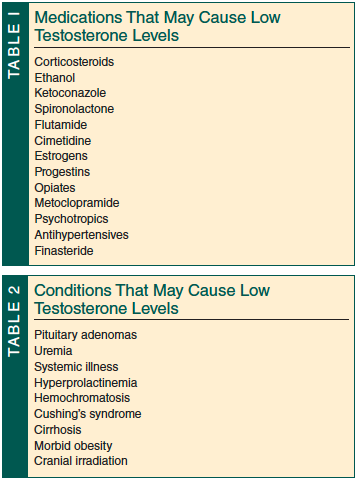
Finally, it is important to point out that many diseases and medications frequently used in older patients can cause hypogonadism and/or erectile dysfunction39,40 (Tables 1-3).
Testosterone and Other Pathophysiologic Changes in Aging Men
 In aging men, androgen deficiency may have unfavorable consequences on body composition, bone mineral density, muscle strength, insulin sensitivity, and lipid profile.41 Several studies have documented an age-related loss of lean body mass with a corresponding increase in body fat.41,42 Some investigators have observed decreases in muscle strength, aerobic capacity (VO2max), and insulin sensitivity.41 Data suggest that men in their 80s have a 20% to 25% reduction in bone mineral density compared with men in their 20s, although it should be noted that bone density loss for women this age is 40% to 50%.42 In men, it is assumed that there is a cause-and-effect relationship between decreasing androgen levels and bone loss, but this has not been proven.
In aging men, androgen deficiency may have unfavorable consequences on body composition, bone mineral density, muscle strength, insulin sensitivity, and lipid profile.41 Several studies have documented an age-related loss of lean body mass with a corresponding increase in body fat.41,42 Some investigators have observed decreases in muscle strength, aerobic capacity (VO2max), and insulin sensitivity.41 Data suggest that men in their 80s have a 20% to 25% reduction in bone mineral density compared with men in their 20s, although it should be noted that bone density loss for women this age is 40% to 50%.42 In men, it is assumed that there is a cause-and-effect relationship between decreasing androgen levels and bone loss, but this has not been proven.
Testosterone Supplementation Studies
All of the observations discussed above in this review have led many investigators to conduct testosterone supplementation studies. The detailed findings of these studies were discussed in a previous article in Clinical Geriatrics.43
In summary, the results from investigations into testosterone supplementation have been inconclusive and rather disappointing overall. It appears that testosterone supplementation produces a modest increase in lean body and muscle mass with a corresponding decrease of the fat mass, and libido usually improves.44-46 In contrast, researchers have not been able to verify conclusively the anticipated effects of testosterone supplementation on bone mineral density, muscle strength, aerobic capacity,41 physical function, sexual activity levels,44,47,48 quality of life, cognition, and depression.49 Potential adverse outcomes associated with long-term testosterone administration have yet to be defined.
Summary
In contrast with the well-defined phenomenon of menopause in women, it has not been determined with certainty whether aging men experience its equivalent, sometimes referred to as male menopause or by one of the near-synonymous terms previously mentioned.
Menopause is universal among women, occurring in the vast majority of them within a narrow age range (50-53 years of age). The changes presumed to be components of what has been called male menopause are far from universal and can appear at any age after 50 to 55 years. In many men, these changes are not evident until they reach their late 70s to 80s, and even then they vary greatly between individuals. Results of testosterone supplementation studies in androgen-deficient older men have not been conclusive. It is left to the reader to judge whether a syndrome called male menopause or by any of the synonyms mentioned earlier exists and deserves further clinical recognition and attention.
The author reports no relevant financial relationships.
Dr. Tsitouras is Clinical Director, Kronos Longevity Research Institute, Phoenix, AZ.
References
1. Harman SM. Clinical aspects of aging in the male reproductive system. In: Schneider EL, ed: The Aging Reproductive System. New York, NY: Raven Press; 1978:29.
2. Tsitouras PD, Alvarez RR. Etiology and management of sexual dysfunction in elderly men. Psychiatr Med. 1984;2(1):43-55.
3. Brown-Séquard CE. Note on the effects produced on man by subcutaneous injections of a liquid obtained from the testicles of animals. Lancet. 1889;2:105-107.
4. Murad F, Haynes RC Jr. Androgens and anabolic steroids. In: Goodman LS, Gilman A, eds: The Pharmacological Basis of Therapeutics. 6th ed. New York, NY: MacMillan; 1980:1448.
5. Demers LM. Andropause: an androgen deficiency state in the ageing male. Expert Opin Pharmacother. 2003;4(2):183-190.
6. Tsitouras PD. Effects of age on testicular function. Endocrinol Metab Clin North Am. 1987;16(4):1045-1059.
7. Kinsey AC, Pomeroy WB, Martin CE. Sexual Behavior in the Human Male. Philadelphia, PA: WB Saunders; 1948.
8. Martin CE. Marital and sexual factors in relation to age, disease, and longevity. In: Wirt RD, Winokur G, Roff M, eds. Life History Research in Psychopathology. Vol 4. Minneapolis: University of Minnesota Press; 1975:326.
9. Martin CE. Factor affecting sexual functioning in 60-79-year-old married males. Arch Sex Behav. 1981;10(5):399-420.
10. Edwards JN, Booth A. The cessation of marital intercourse. Am J Psychiatry. 1976;33(11):1333-1336.
11. Pfeiffer E. Sexuality in the aging individual. J Am Geriatr Soc. 1974;22(11):481-484.
12. Masters WH, Johnson VE. Sex and the aging process. J Am Geriatr Soc. 1981;
29(9):385-390.
13. Masters WH, Johnson VE. Human Sexual Response. Boston, MA: Little, Brown & Co;
1966:221.
14. Kastin AJ, Schally AV, Gual C, Arimura A. Release of LH and FSH after administration of synthetic LH-releasing hormone. J Clin Endocrinol Metab. 1972;34(4):753-756.
15. Nankin HR, Murono E, Lin T, Osterman J. Morning and evening human Leydig cell responses to hCG. Acta Endocrinol (Copenh). 1980;95(4):560-565.
16. Harman SM, Metter EJ, Tobin JD, Pearson J, Blackman MR; Baltimore Longitudinal Study of Aging. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab. 2001;86(2):724-731.
17. Dabbs JM Jr. Age and seasonal variation in serum testosterone concentration among men. Chronobiol Int. 1990;7(3):245-249.
18. Gandy HM, Peterson RE. Measurement of testosterone and 17-ketosteroids in plasma by the double isotope dilution derivative technique. J Clin Endocrinol Metab. 1968;28(7):949-977.
19. Vermeulen A, Verdonck L. Some studies on the biological significance of free testosterone. J Steroid Biochem. 1972;3(3):421-426.
20. Harman SM, Tsitouras PD. Reproductive hormones in aging men. I. Measurement of sex steroids, basal luteinizing hormone, and Leydig cell response to human chorionic gonadotropin. J Clin Endocrinol Metab. 1980;51(1):35-40.
21. Sparrow D, Bosse R, Rowe JW. The influence of age, alcohol consumption, and body build on gonadal function in men. J Clin Endocrinol Metab. 1980;51(3):508-512.
22. Coiro V, Passeri M, Volpi R, et al. Different effects of aging on the opioid mechanisms controlling gonadotropin and cortisol secretion in man. Horm Res. 1989;32(4):119-123.
23. Rodriguez A, Muller DC, Metter EJ, et al. Aging, androgens, and the metabolic syndrome in a longitudinal study of aging. J Clin Endocrinol Metab. 2007;92(9):3568-3572.
24. Ceda GP, Denti L, Ceresini G, Torsiglieri W, Hoffman AR, Valenti G. The effects of aging on the secretion of the common alpha-subunit of the glycoprotein hormones in men. J Am Geriatr Soc. 1991;39(4):353-358.
25. Fingscheidt U, Nieschlag E. The response of inhibin to human chorionic gonadotrophin is decreased in senescent men compared with young men. J Endocrinol. 1989;123(2):R9-R11.
26. Veldhuis JD, Urban RJ, Lizarralde G, Johnson ML, Iranmanesh A. Attenuation of luteinizing hormone secretory burst amplitude as a proximate basis for the hypoandrogenism of healthy aging in men. J Clin Endocrinol Metab. 1992;75(3):707-713.
27. Cangemi R, Friedmann AJ, Holloszy JO, Fontana L. Long-term effects of calorie restriction on serum sex-hormone concentrations in men. Aging Cell. 2010;9(2):236-342.
28. Ponholzer A, Plas E, Schatzl G, et al. Relationship between testosterone serum levels and lifestyle in aging men. Aging Male. 2005;8:190-193.
29. Kaplan SA, Meehan AG, Shah A. The age related decrease in testosterone is significantly exacerbated in obese men with the metabolic syndrome. What are the implications for the relatively high incidence of erectile dysfunction observed in these men? J Urol. 2006;176(4):1524-1527.
30. Travison TG, Araujo AB, Kupelian V, O’Donnell AB, McKinlay JB. The relative contributions of aging, health, and lifestyle factors to serum testosterone decline in men. J Clin Endocrinol Metab. 2007;92(2):549-555.
31. Tsitouras PD, Martin CE, Harman SM. Relationship of serum testosterone to sexual activity in healthy elderly men. J Gerontol. 1982;37(3):288-293.
32. Harman SM, Tsitouras PD, Costa PT, Blackman MR. Reproductive hormones in aging men. II. Basal pituitary gonadotropins and gonadotropin responses to luteinizing hormone-releasing hormone. J Clin Endocrinol Metab. 1982;54(3):547-551.
33. Nieschlag E, Kley KH, Wiegelmann W. Age dependence of the endocrine testicular function in adult men. Acta Endocrinol (Kbh). 177(suppl):122, 1973.
34. Rubens R, Dhont M, Vermeulen A. Further studies on Leydig cell function in old age. J Clin Endocrinol Metab. 1974;39(1):40-45.
35. Giusti M, Marini G, Traverso L, Cavagnaro P, Granziera L, Giordano G. Effect of pulsatile luteinizing hormone-releasing hormone administration on pituitary-gonadal function in elderly man. J Endocrinol Invest. 1990;13(2):127-132.
36. Korenman SG, Morley JE, Mooradian AD, et al. Secondary hypogonadism in older men: its relation to impotence. J Clin Endocrinol Metab. 1990;71(4):963-969.
37. Veldhuis JD, Urban RJ, Beitins IZ, et al. Pathophysiological features of the pulsatile secretion of biologically active luteinizing hormone in man. J Steroid Biochem. 1989;33(4B):739-749.
38. Davidson JM, Kwan M, Greenleaf WJ. Hormonal replacement and sexuality in men. Clin Endocrinol Metab. 1982;11(3):599-623.
39. Tsitouras PD, Bulat T. The aging male reproductive system. Endocrinol Metab Clin North Am. 1995;24(2):297-315.
40. Isidori AM, Giannetta E, Gianfrilli D, et al. Effects of testosterone on sexual function in men: results of a meta-analysis. Clin Endocrinol (Oxf). 2005;63(4):381-394.
41. Blackman MR, Sorkin JD, Munzer T, et al. Growth hormone and sex steroid administration in healthy aged women and men: a randomized controlled trial. JAMA. 2002;288(18):2282-2292.
42. Snyder PJ, Peachey H, Hannoush P, et al. Effect of testosterone treatment on bone mineral density in men over 65 years of age. J Clin Endocrinol Metab. 1999;84(6):1966-1972.
43. Tsitouras PD. Counterpoint: testosterone therapy in older men: not ready for prime time yet. Clinical Geriatrics. 2009;17(7):25-27.
44. Boloña ER, Uraga MV, Haddad RM, et al. Testosterone use in men with sexual dysfunction: a systematic review and meta-analysis of randomized placebo-controlled trials. Mayo Clin Proc. 2007;82(1):20-28.
45. Steidle C, Schwartz S, Jacoby K,Sebree T, Smith T, Bachand R; North American AA2500 T Gel Study Group. AA2500 testosterone gel normalizes androgen levels in aging males with improvements in body composition and sexual function. J Clin Endocrinol Metab. 2003;88(6):2673-2681.
46. Cavallini G, Caracciolo S, Vitali G, Modenini F, Biagiotti G. Carnitine versus androgen administration in the treatment of sexual dysfunction, depressed mood, and fatigue associated with male aging. Urology. 2004;63(4):641-646.
47. O’Carroll R, Bancroft J. Testosterone therapy for low sexual interest and erectile dysfunction in men: a controlled study. Br J Psychiatry. 1984;145:146-151.
48. Schiavi RC, White D, Mandeli J, Levine AC. Effect of testosterone administration on sexual behavior and mood in men with erectile dysfunction. Arch Sex Behav. 1997;26(3):231–241.
49. Bhasin S, Cunningham GR, Hayes FJ, et al; Task Force, Endocrine Society. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95(6):2536-2559.


