Peer Reviewed
Lumps and Bumps in Adolescents: Benign Tumors, Growths, and Mucosal Lesions
Dr Leung is clinical professor of pediatrics at the University of Calgary and a pediatric consultant at the Alberta Children’s Hospital in Calgary. Dr Wong is clinical lecturer of family medicine at the University of Calgary.
ABSTRACT: Most benign cutaneous tumors and growths in adolescents are asymptomatic and treatment is for cosmetic reasons. Further evaluation for an underlying disorder is required in some instances, such as in patients with multiple cutaneous neurofibromas. Surgical removal of certain growths, eg, dermatofibromas, can result in a scar that may be larger than the original lesion. Lesions with malignant potential, such as common blue nevi, and keloids can be excised but may recur. Hemorrhoids usually return to the rectum spontaneously and respond to adequate fluid intake, high-fiber diets, and stool softeners. For hemorrhoids that fail to respond to medical treatment, banding is by far the most effective option.
Diagnosis of benign cutaneous tumors, growths, and mucosal lesions in adolescents is similar to that in other age groups. It is usually straightforward and can often be made on the basis of the history and physical examination and the lesion’s morphologic characteristics. In the evaluation of any lesion, it is crucial not to overlook the possibility of an underlying disorder, such as neurofibromatosis, or malignancy (eg, malignant blue nevus).
Adolescents, more than other age groups, may seek treatment because of concern about a lesion’s appearance. When deciding on treatment, it is important to balance the likelihood of scarring and recurrence with patient satisfaction.
In this article, we review the clinical features of benign tumors, growths, and mucosal lesions that are common in adolescents and young adults. We also describe the latest techniques in diagnosis and management of these lesions.
NEUROFIBROMAS
Neurofibromas are benign peripheral nerve sheath tumors composed of an extracellular matrix and a heterogeneous mixture of Schwann cells, perineural-like cells, and fibroblasts.1 Cutaneous neurofibromas—the most common type of neurofibroma—often appear during puberty or pregnancy and increase in size and number with age.2
Cutaneous neurofibromas are painless, soft, flesh-colored, nodular or pedunculated lesions that often invaginate into the skin and cause a “buttonholing” sensation when gentle digital pressure is applied to the surface.3 In contrast, subcutaneous neurofibromas, which are usually much harder and more painful, are deeply seated in the dermis and, therefore, less circumscribed.3,4
Plexiform neurofibromas are often present at birth and result from diffuse thickening of nerve trunks.5 They feel like a tangle of thick strands or a ropy mass. Plexiform neurofibromas may be locally invasive and associated with bone erosion and pain; overgrowth of an extremity and malignant transformation are possible.6,7
In the vast majority of cases, cutaneous neurofibromas are solitary and not associated with type 1 neurofibromatosis. However, patients with multiple neurofibromas should be evaluated to see whether they meet any of the 7 diagnostic criteria for type I neurofibromatosis, including8:
•Two or more neurofibromas of any type or 1 plexiform neurofibroma.
•Six or more café au lait spots, 1.5 cm or larger in postpubertal persons and 0.5 cm or larger in prepubertal persons.
•Freckling in the axillary or inguinal regions.
•Optic glioma.
•Two or more Lisch nodules.
•An osseous lesion, such as dysplasia of the sphenoid bone or dysplasia or thinning of the long bone cortex (with or without pseudoarthosis).
•A first-degree relative with type 1 neurofibromatosis, according to the preceding criteria.8
Two or more of the diagnostic criteria have to be met in order to make a diagnosis of type 1 neurofibromatosis.8 The patient described in Case 1 met at least 3 of the criteria described (ie, the presence of multiple neurofibromas and café au-lait spots and a first-degree relative with similar skin findings).
Treatment is mainly symptomatic. Surgical excision of cutaneous neurofibromas is usually not necessary, except for cosmetic purposes.
 DERMATOFIBROMA
DERMATOFIBROMA
Dermatofibroma (benign fibrous histiocytoma) is a common, benign, dermal lesion composed primarily of fibroblasts and histiocytes, with excessive deposition of collagen in the dermis.9,10 Prolonged exposure to the sun, chronic infection, and trauma have been suggested as possible causes.10 Typically, the lesion presents as an asymptomatic round or ovoid, firm nodule usually less than 1 cm in diameter.9 The lesion can have variable colors, ranging from pink to tan to brown. Although the lesion can develop anywhere on the body, it is most commonly found on the extremities, and particularly on the lower extremities of young adults (see Case 2).9 There is a slight female predominance.9,11
The lesion has a smooth surface. It is attached to the skin but not to the underlying structure. Pinching the lesion often leads to depression, or dimpling, of the nodule (dimple sign, bullet sign, or Fitzpatrick sign). Dermatofibromas tend to reach a certain size and then remain stable. The lesions are often solitary; multiple lesions are found in approximately 10% of cases.9 The classic dermoscopic pattern is the central white scar-like patch and peripheral delicate pigment network.11
Treatment is usually unnecessary, as surgical removal can result in a scar that may be larger than the original lesion.
BLUE NEVUS
Blue nevi are a subset of melanocytic proliferation that contain cells reminiscent of the embryonal neural crest-derived dendritic melanocytic precursors.12 These nevi are benign, usually acquired, and asymptomatic; they occur in 0.5% to 4% of healthy white adults.13 Blue nevi are a heterogenous group of disorders that includes established entities, such as common (dendritic) blue nevus and cellular blue nevus.
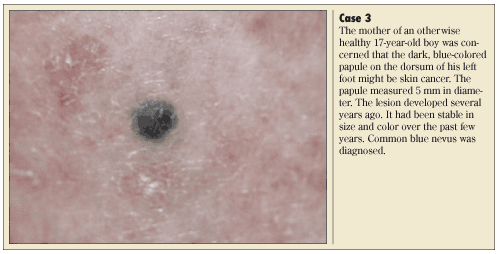 A common blue nevus typically presents as a well-circumscribed, solitary, dome-shaped, slate-blue or blue-black papule or nodule, as is illustrated in Case 3.14 The lesion is usually smaller than 1 cm in diameter and is generally located on the dorsa of the hands and feet.14 The onset is most common in childhood and adolescence. The condition is more common in females.12 Histologically, the lesion is composed predominately of pigmented, spindle-shaped, dendritic melanocytes.14
A common blue nevus typically presents as a well-circumscribed, solitary, dome-shaped, slate-blue or blue-black papule or nodule, as is illustrated in Case 3.14 The lesion is usually smaller than 1 cm in diameter and is generally located on the dorsa of the hands and feet.14 The onset is most common in childhood and adolescence. The condition is more common in females.12 Histologically, the lesion is composed predominately of pigmented, spindle-shaped, dendritic melanocytes.14
A cellular blue nevus typically presents as a firm, bluish gray to bluish black nodule, measuring 1 to 3 cm in diameter.12,14 Lesions larger than 10 cm have also been described.12 Sites of predilection include the buttocks and the lower back, followed by the scalp.12,14 Although cellular blue nevi can present at any age, adults younger than 40 years are most commonly affected.12,14 Histologically, the lesion is characterized by densely packed strands or nests of scarcely pigmented ovol to plump spindle cells, intermingled with heavily pigmented dendritic melanocytes and variable amounts of melanophages.14,15 Sclerosis is often prominent.
Malignant blue nevus is a rare, aggressive dermal melanocytic neoplasm that often metastasizes to the regional lymph nodes.12 Most malignant blue nevi arise de novo from a preexisting common blue nevus or cellular blue nevus.12
Surgical excision of a blue nevus is appropriate when malignancy is suspected and can be considered for cosmetic reasons.
 HEMORRHOIDS
HEMORRHOIDS
Hemorrhoids are fibrovascular cushions that contain arteriovenous communications located in the subepithelial space of the anal canal.16 Chronic excessive straining with defecation, which can occur with constipation, may result in the sliding down of the internal cushions along the anal canal with the formation of internal hemorrhoids.16 It is estimated that approximately 5% of the general population and 50% of adults older than 50 years have experienced hemorrhoid problems.17 The sex ratio is equal.16
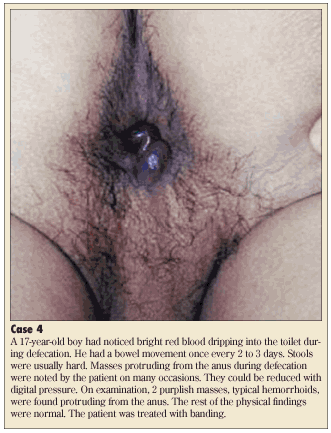
Typically, affected patients pre-sent with painless rectal bleeding, anal masses and protrusion, mucous discharge, or a feeling of incomplete evacuation (see Case 4).17 Hemorrhoids usually return to the rectum spontaneously, but sometimes they need to be pushed back. If the prolapsed hemorrhoid cannot be reduced, it may lead to thrombosis, ulceration, strangulation, infraction, and gangrene.
The diagnosis is almost always a clinical one. Anoscopy and proctoscopy should be performed to look for evidence of thrombosis and concomitant anorectal pathology.16,18
Most hemorrhoids respond to adequate fluid intake, high-fiber diets, and stool softeners.18 Sitz baths may be used for anal hygiene and to relax the spastic pelvic floor muscles.16 Hemorrhoids that fail to respond to medical treatment may be treated with elastic band ligation, sclerotherapy, infrared photocoagulation, hemorrhoidopexy, and hemorrhoidec-tomy.17,18 Hemorrhoid banding is by far the most effective option.18
 KELOID
KELOID
The term “keloid” originates from the Greek word chele, meaning crab claw and has been used to describe extensions of scar tissue into surrounding skin.19 A keloid represents an excessive response to cutaneous injury. It is a benign hyper-proliferative growth of dense fibrous tissue and overabundant deposition of disorganized, thick, hyalinized collagen.20
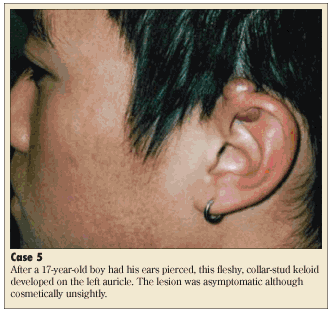
Common examples of cutaneous injury include varicella infection, ear piercing (as in Case 5), burn injury, laceration, and surgical incision. The condition is more common in dark-skinned individuals.20 There is also a familial predisposition to keloid formation.19 The peak incidence is between 10 and 30 years of age.19-21
Clinically, keloids appear as skin-colored firm nodules that extend beyond the margins of the original wound. This is in contrast to a hypertrophic scar, which tends to stay within the margins of the original wound.20 Some keloids have a pinkish appearance with notable telangiectasias.
The condition can be anatomically disfiguring and emotionally traumatic.20 Some affected patients may experience pruritus and pain.20 Keloids can be complicated by secondary infections.20
Small keloids may not need to be treated. Large keloids or keloids in an exposed area can be treated with intralesional injections of triamcinolone, alone or in combination with surgical excision. Other treatment options include silicone gel sheeting, carbon dioxide laser radiation, and pulsed dye laser.19,20 Postoperative pressure therapy with a custom-made methyl methacrylate stent is efficacious, safe, and usable for keloids of both the helix and the earlobe.22
 LIPOMA
LIPOMA
Lipoma is a benign subcutaneous tumor composed of mature adipocytes; it is, by far, the most common mesenchymal neoplasm.23 The cause is unknown.
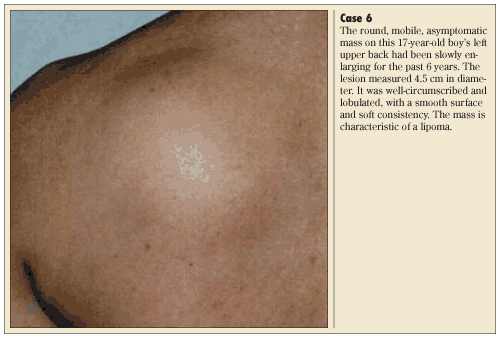
Clinically, a lipoma presents as a slow-growing, soft, well-circumscribed, lobulated mass, which is not attached to the overlying skin or underlying structures (Case 6). A “slipping sign” elicited by gently sliding the fingers off the edge of the tumor is typical. Sites of predilection include the back, shoulders, upper arm, upper thigh, and neck.23 The condition is more common in adults than in children.
Most lipomas are solitary and asymptomatic. Rarely, the condition may be associated with compression of a peripheral nerve, hereditary multiple lipomatosis, adiposis dolorosa, Madelung disease, or Gardner syndrome.24 A lipoma may be complicated by fatty necrosis, calcification, and xanthomatous change.25 Liposarcoma usually arises de novo and not from a benign lesion.
Treatment may be considered for cosmetic reasons and for painful or rapidly growing lipomas. Management options include excision, corticosteroid injections, and liposuction. n


