A Look at Cardiometabolic Risk
Diabetic Man With Pruritic Pretibial Rash
Alexander K. C. Leung, MD, Maria Fincati, MD, and Henry Schneiderman, MD
A 69-year-old man seen because of a pruritic pretibial rash of 3 months' duration. Started as reddish brown papules that slowly enlarged and changed. Type 2 diabetes mellitus for 10 years, treated with metformin, 500 mg bid; fairly good glycemic control. Nonsmoker; past health otherwise unremarkable. Several well circumscribed, brownish plaques in the left pretibial area. Borders somewhat raised, a bit firm, and more deeply pigmented than centers. Central atrophy with depression below the level of the skin surface in some lesions. Physical examination apart from the skin, normal.
This patient’s shin shows mildly atrophic skin, marked by the paucity of skin lines and an impression that the venous network is unduly prominent beneath the surface, though not dilated. In addition, partial confluence of 2 large circular red-brown lesions is seen, the rightmost “dribbling” down the lateral margin toward the ankle.
Necrobiosis lipoidica diabeticorum (NLD) is characterized at the bedside by ovoid plaques with an irregular, elevated periphery and an atrophic center.1 The condition was first described by Oppenheim in 1929.2 The term “necrobiosis lipoidica diabeticorum” was based on characteristic histologic findings and frequent association with diabetes mellitus.3
Epidemiology. NLD is an uncommon but well-recognized complication of diabetes mellitus, with a prevalence of 0.3% in diabetic adults and 0.06% in diabetic children.4,5 Some three fourths of persons with NLD have diabetes at the time of diagnosis.6 In others, these lesions precede the diagnosis of diabetes by up to several years.7
A family history of diabetes can be obtained in 90% of affected patients.8 The female to male ratio is approximately 4:1. The cause of the sex disparity is unknown.1 NLD usually arises in young or middle adulthood.8,9 Smoking increases the risk of NLD.7 The condition is also more prevalent among those with thyroid disease, Crohn disease, and ulcerative colitis.6,10,11
Pathogenesis. Although the etiology and mechanism of development remain unknown, diabetic microangiopathy might be a major underlying cause.4,8 In this regard, persistent microalbuminuria and retinopathy are more common among patients with diabetes in whom NLD develops than among control patients with diabetes who do not have NLD.12 However, this could merely reflect that persistent microalbuminuria and retinopathy imply long disease duration and poor diabetic control: NLD may also be associated with these factors.12,13
Just to make it more confusing, other groups with severe microvasculopathy—including those with uremic arteriolopathy (calciphylaxis)14—do not characteristically have NLD. Nor is NLD typical of some other vaso-occlusive disorders, such as atheroembolic disease,15 vasculitides,16-18 or the lupus “anticoagulant.” Insights about the pathogenesis of NLD are empiric, limited, and imprecise. One struggles to explain why young diabetics are preferentially affected by NLD, whereas older diabetics—precisely those persons with the worst vasculopathy--constitute a lower-risk population.
Immunoglobulins have been found around blood vessels in affected areas. This could suggest an immune complex vasculitis,19,20 except that vasculitis is not seen in biopsy specimens. Focal degeneration of collagencould also be an initiating event.21-23 In one study, levels of lysyl oxidase (which produces collagen cross-linking by the oxidation of lysine and hydroxylysine) were elevated in patients with NLD.21 In another study, overhydration of collagen was demonstrated in the skin of diabetics, but the relationship between hydration of these fibers and NLD is obscure.22
Histology. The characteristic features include necrobiosis, which is a brightly eosinophilic degeneration of collagen, throughout the skin, and infiltration of collagen bundles by palisading histiocytes—a term that describes how they align with the nuclei in a single parallel axis.1,8 The upper dermis contains fatty deposits.1 Mucin deposits are minimal.8 Although NLD is considered a granulomatous and inflammatory disorder, the significance of the histiocytes is uncertain, as is the paucity of more common inflammatory cells, such as lymphocytes or neutrophils.
Clinical manifestations. This usually begins as erythematous papules. These gradually enlarge, then develop irregular margins, and can coalesce into irregular waxy plaques (Figure 1). The plaques often display a yellowish or reddish brown hue and tend to be sharply demarcated, with raised and sometimes indurated borders that could at least transiently suggest an enlarging skin cancer. Some of the lesions may show central atrophy, telangiectasia, and yellow pigmentation.24 In 85% of cases, the lesion is confined to the pretibial area.4,25 Ankles, feet, calves, and thighs are most often involved, along with the shins, in the remainder. In fewer than 2% of cases, lesions are found in other areas but not the legs.22
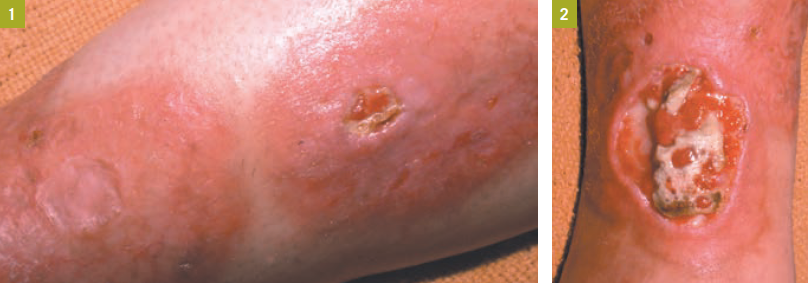
Most NLD lesions are asymptomatic. Some lesions are pruritic (as were those in our patient), painful, or dysesthetic.7 Ulceration develops in one third of cases.26 Ulcers are typically caused by minor trauma; healing is notoriously slow, and not because of any superinfection,26 although terrible-looking infection can occur (Figure 2). Curiously, the ulcerations do not appear to represent simple venous disease superimposed on NLD, although any condition that preferentially affects the shin will make one wonder about venous insufficiency.
Treatment. NLD lesions are usually self-limited.8 However, they may recur.27 Even in those that become ulcerated, infection is rare.28 Skin cancers have been reported in chronically ulcerated NLD lesions as in other kinds of nonhealing ulcers, but this is very rare.
Because of the unsightliness of NLD lesions and concerns about potential infection or neoplasia, diverse treatments have been tried, without consistent benefit.29-31 These include topical, intralesional, or systemic corticosteroids, topical retinoids, the combination of aspirin and the older antianginal/antiplatelet agent dipyridamole), and even cyclosporin. Recently, all patients in a small group of young and middle aged women with NLD benefited from psoralen and ultraviolet A light.32 ■
References:
1. Cunningham FG, Lindheimer MD. Hypertension in pregnancy. N Engl J Med. 1992;326:927-932.
2. Sibai BM. Chronic hypertension in pregnancy. Obstet Gynecol. 2002;100: 369-377.
3. American College of Obstetricians and Gynecologists. Diagnosis and Management of Preeclampsia and Eclampsia. ACOG practice bulletin 33. Washington, DC: American College of Obstetricians and Gynecologists; 2002.
4. National Institutes of Health, National Heart, Lung, and Blood Institutes, National High Blood Pressure Education Program Coordinating Committee. Working group report on high blood pressure in pregnancy. 2000. Accessed February 16, 2005.
5. Lain KY, Roberts JM. Contemporary concepts of the pathogenesis and management of preeclampsia. JAMA. 2002;287:3183-3186.
6. Einarsson JI, Sangi-Haghpeykar H, Gardner MO. Sperm exposure and development of preeclampsia. Am J Obstet Gynecol. 2003;188:1241-1243.
7. Granger JP, Alexander BT, Bennett WA, Khalil RA. Pathophysiology of pregnancy-induced hypertension. Am J Hypertens. 2001;14(6 pt 2):178S-185S.
8. Becker R, Vonk R, Vollert W, Entezami M. Doppler sonography of uterine arteries at 20-23 weeks: risk assessment of adverse pregnancy outcome by quantification of impedance and notch. J Perinat Med. 2002;30:388-394.
9. Livingston JC, Livingston LW, Ramsey R, et al. Magnesium sulfate in women with mild preeclampsia: a randomized controlled trial. Obstet Gynecol. 2003;101: 217-220.
10. Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol. 2003;102:181-192.
11. Sibai BM. Diagnosis, prevention, and management of eclampsia. Obstet Gynecol. 2005;105:402-410.
Mediastinal Lipotomosis
Ashwani Bhardwaj, MD, and Pritam S.Badesha, MD
Van Nuys, California
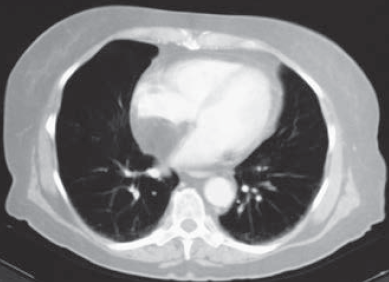 An obese 56-year-old woman was hospitalized after 2 days of chest pain, dyspnea, and palpitations. Physical examination revealed no abnormalities, and serial cardiac enzyme studies and an ECG ruled out myocardial infarction. Suspicion was aroused, however, when transesophageal echocardiography showed a lesion in the mediastinum.
An obese 56-year-old woman was hospitalized after 2 days of chest pain, dyspnea, and palpitations. Physical examination revealed no abnormalities, and serial cardiac enzyme studies and an ECG ruled out myocardial infarction. Suspicion was aroused, however, when transesophageal echocardiography showed a lesion in the mediastinum.
A CT scan of the chest revealed a mass in the mediastinum, and the diagnosis of mediastinal lipomatosis was confirmed by the tumor's characteristic low density (from 220 H to 2100 H). The patient’s chest pain subsided without any intervention. Two years later, the mass showed no change in size, confirming a benign process.
Lipomatosis is often associated with obesity, corticosteroid treatment, or Cushing disease. In most cases, the lesions are benign, although liposarcomas do occur in the mediastinum. ■
Diabetic Dermopathy
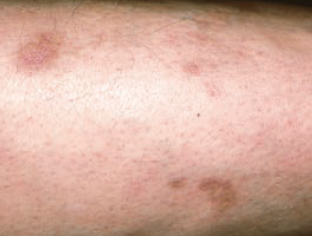 A 52-year-old man with diabetes mellitus presents for evaluation of an asymptomatic rash on his right leg. The lesions have been present for 2 months.
A 52-year-old man with diabetes mellitus presents for evaluation of an asymptomatic rash on his right leg. The lesions have been present for 2 months.
Diabetic dermopathy is thought to be a microangiopathic condition that produces asymptomatic, hyperpigmented macules on the shins of persons with diabetes.
Statis dermatitis is not as discrete and erupts on the lower legs. Lichen planus features purplish plaques. Erythema and scale are characteristic of the less pigmented lesions of dermatophyte infections. Necrobiosis lipoidica diabeticorum plaques are usually atrophic and organish. ■


